Global COVID-19 Saliva-Based Screening Market to Continue Growing Due to Ease of Use and Shorter Test-to-Result
|
By LabMedica International staff writers Posted on 07 Sep 2021 |
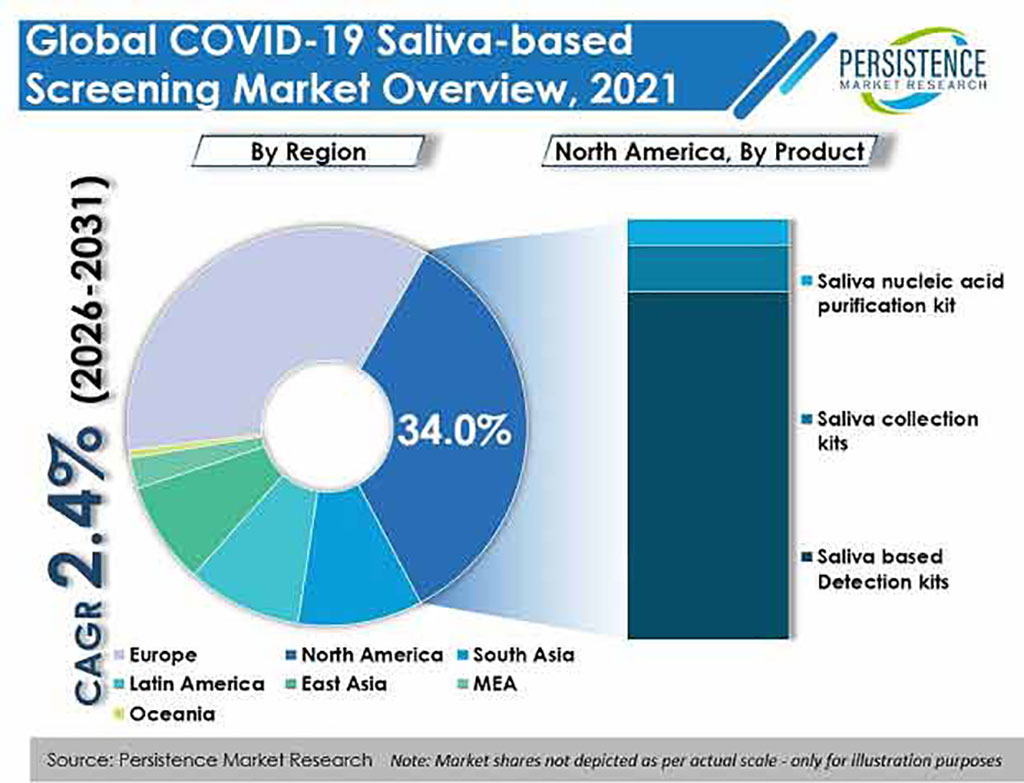
Image: Global COVID-19 Saliva-Based Screening Market to Continue Growing due to Ease of Use and Shorter Test-To-Result (Photo courtesy of Persistence Market Research)
The global COVID-19 saliva-based screening market was valued at over USD 1.7 billion in 2020 and is gaining traction, as it is patient-friendly, easy to use, and has a shorter test-to-result timeline.
These are the latest findings of Persistence Market Research (New York City, NY, USA), a market research company.
COVID-19 saliva-based screening plays a dynamic role in screening asymptomatic persons, thus sustaining market growth. This is mainly owing to numerous advantages offered by saliva-based tests, growing product approvals by FDA, and a shift toward point-of-care testing. With the onset of the COVID-19 pandemic, several key players have entered the saliva-based screening market. Furthermore, manufacturers are investing extensively in research & development activities to develop novel techniques that can allow rapid testing and accurate results.
Additionally, the ageing population is more susceptible to viral infections due to patient comorbidities, which is expected to boost the demand for COVID-19 saliva-based screening. New COVID-19 saliva tests make testing easier for all ages since the saliva-based method enables a simpler and quicker collection of samples. The current market is still powered by other specimen testing kits that use samples apart from saliva, such as blood and nasopharyngeal swabs, although many new players are expected to launch saliva-based kits. With the rising adoption of saliva-based screening, saliva as a sample is expected to rule the specimen type.
Moreover, the outbreak of the novel coronavirus and its rapid spread has forced countries to impose compulsory guidelines for COVID-19 diagnostics and other activities such as travelling. Many countries have made it mandatory to issue a negative COVID-19 test to travel internationally as well as domestically. Also, to break the chain, government guidelines have also included testing of individuals who may have come in contact with infected persons. A rise in the number of tests due to government norms and guidelines offers considerable growth opportunities for the COVID-19 saliva-based screening market.
However, the market for COVID-19 saliva-based screening products is expected to gradually decline over the years due to increasing focus on vaccination across countries, support from developing countries to supply vaccinations to underdeveloped countries, and the population attaining herd immunity. Nevertheless, with the occurrence of the second wave in countries in the last two quarters and the anticipated outbreak of the second and third waves across different countries, the demand for COVID-19 saliva-based screening will remain high. Hence, COVID-19 saliva-based screening is expected to experience further growth during the forecast period due to the pandemic.
Based on product, saliva-based detection kits are leading the COVID-19 saliva-based screening market with a share of over 83%. The inclination towards saliva-based tests can be due to escalating demand for home-based COVID-19 testing using saliva-based detection kits. By technology, RT-qPCR technology holds a share of around 49% of the global COVID-19 saliva-based screening market, primarily due to increasing focus on rapid testing and growing research activities. The segment’s high share can also be attributed to continued demand in areas such as gene expression analysis, genetic variation analysis, and genotyping, along with increasing focus on the development of RT-qPCR technology. Based on application, COVID-19 diagnostics held a large share of 63% in 2021. The rising prevalence of COVID-19, need for rapid diagnostics, and increasing demand for home-based COVID-19 testing are contributing to the growth of COVID-19 diagnostics. Diagnostic laboratories dominate the market among end-users with a share of 27%. Diagnostic laboratories will provide lucrative opportunities for market growth owing to growing incidence of COVID-19 and increased virus mutation, which has led to an increase in the need for rapid detection with more accuracy.
Geographically, Europe is set to dominate the global COVID-19 saliva-based screening market with a value share of around 36%. Russia dominates the European market for COVID-19 saliva-based screening with a share of 21%. North America is slated to be the second-largest leading market for COVID-19 saliva-based screening with a value share of 34% through 2031. The US dominated the North American market with a total share of 91% in 2021, and is projected to continue experiencing low to moderate growth throughout the forecast period. Moreover, increasing incidence rate of COVID-19 in the US is also boosting demand for COVID-19 saliva-based screening.
Related Links:
Persistence Market Research
These are the latest findings of Persistence Market Research (New York City, NY, USA), a market research company.
COVID-19 saliva-based screening plays a dynamic role in screening asymptomatic persons, thus sustaining market growth. This is mainly owing to numerous advantages offered by saliva-based tests, growing product approvals by FDA, and a shift toward point-of-care testing. With the onset of the COVID-19 pandemic, several key players have entered the saliva-based screening market. Furthermore, manufacturers are investing extensively in research & development activities to develop novel techniques that can allow rapid testing and accurate results.
Additionally, the ageing population is more susceptible to viral infections due to patient comorbidities, which is expected to boost the demand for COVID-19 saliva-based screening. New COVID-19 saliva tests make testing easier for all ages since the saliva-based method enables a simpler and quicker collection of samples. The current market is still powered by other specimen testing kits that use samples apart from saliva, such as blood and nasopharyngeal swabs, although many new players are expected to launch saliva-based kits. With the rising adoption of saliva-based screening, saliva as a sample is expected to rule the specimen type.
Moreover, the outbreak of the novel coronavirus and its rapid spread has forced countries to impose compulsory guidelines for COVID-19 diagnostics and other activities such as travelling. Many countries have made it mandatory to issue a negative COVID-19 test to travel internationally as well as domestically. Also, to break the chain, government guidelines have also included testing of individuals who may have come in contact with infected persons. A rise in the number of tests due to government norms and guidelines offers considerable growth opportunities for the COVID-19 saliva-based screening market.
However, the market for COVID-19 saliva-based screening products is expected to gradually decline over the years due to increasing focus on vaccination across countries, support from developing countries to supply vaccinations to underdeveloped countries, and the population attaining herd immunity. Nevertheless, with the occurrence of the second wave in countries in the last two quarters and the anticipated outbreak of the second and third waves across different countries, the demand for COVID-19 saliva-based screening will remain high. Hence, COVID-19 saliva-based screening is expected to experience further growth during the forecast period due to the pandemic.
Based on product, saliva-based detection kits are leading the COVID-19 saliva-based screening market with a share of over 83%. The inclination towards saliva-based tests can be due to escalating demand for home-based COVID-19 testing using saliva-based detection kits. By technology, RT-qPCR technology holds a share of around 49% of the global COVID-19 saliva-based screening market, primarily due to increasing focus on rapid testing and growing research activities. The segment’s high share can also be attributed to continued demand in areas such as gene expression analysis, genetic variation analysis, and genotyping, along with increasing focus on the development of RT-qPCR technology. Based on application, COVID-19 diagnostics held a large share of 63% in 2021. The rising prevalence of COVID-19, need for rapid diagnostics, and increasing demand for home-based COVID-19 testing are contributing to the growth of COVID-19 diagnostics. Diagnostic laboratories dominate the market among end-users with a share of 27%. Diagnostic laboratories will provide lucrative opportunities for market growth owing to growing incidence of COVID-19 and increased virus mutation, which has led to an increase in the need for rapid detection with more accuracy.
Geographically, Europe is set to dominate the global COVID-19 saliva-based screening market with a value share of around 36%. Russia dominates the European market for COVID-19 saliva-based screening with a share of 21%. North America is slated to be the second-largest leading market for COVID-19 saliva-based screening with a value share of 34% through 2031. The US dominated the North American market with a total share of 91% in 2021, and is projected to continue experiencing low to moderate growth throughout the forecast period. Moreover, increasing incidence rate of COVID-19 in the US is also boosting demand for COVID-19 saliva-based screening.
Related Links:
Persistence Market Research
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel
Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
Colorectal cancer (CRC) is a leading cause of cancer-related death worldwide, with more than 60% of cases still diagnosed at a late stage. Uptake of existing screening tools remains suboptimal,... Read more
Automated NfL Assay Supports Monitoring of Neurological Disorders
Neuroaxonal injury occurs across a wide range of neurological disorders and remains difficult to monitor noninvasively over time. Blood-based measurement of neurofilament light chain (NfL) provides a biologically... Read moreMolecular Diagnostics
view channel
New Respiratory Panel Expands Pathogen Detection to 25 Targets
Respiratory infections often present with overlapping symptoms, complicating differential diagnosis in acute and community settings. The stakes are higher for older adults, young children, and people with... Read more
Simple Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
Alzheimer’s disease affects millions worldwide but remains difficult to detect at its earliest, pre-symptomatic stage. Clinicians need tools that can identify biological changes before cognitive symptoms... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read more
Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
Early identification of treatment response and relapse remains a major challenge in solid tumors, where minimal residual disease is difficult to detect with routine imaging and blood tests.... Read moreMicrobiology
view channel
Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Accurate and rapid identification of bacterial infections remains challenging in acute care, where delays can hinder timely, targeted therapy. Infectious diseases are a major cause of mortality worldwide,... Read more
Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
Direct-to-consumer gut microbiome kits promise personalized insights by profiling fecal bacteria and generating health readouts, but their analytical accuracy remains uncertain. A new study shows that... Read more
WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
Tuberculosis (TB) remains one of the world’s leading infectious disease killers, yet millions of cases go undiagnosed or are detected too late. Barriers such as reliance on sputum samples, limited laboratory... Read morePathology
view channel
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read more
AI-Powered Tool to Transform Dermatopathology Workflow
Skin cancer accounts for the largest number of cancer diagnoses in the United States, placing sustained pressure on pathology services. Diagnostic interpretation can be variable for challenging melanocytic... Read moreTechnology
view channel
Online Tool Supports Family Screening for Inherited Cancer Risk
Genetic test results in oncology often have implications for relatives who may share inherited cancer risk. Many health systems lack structured processes to help patients alert family members, limiting... Read more
Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
Pneumonia is commonly confirmed with chest X-rays or laboratory assays that can take hours, delaying clinical decisions in acute and outpatient settings. Breath-based diagnostics promise faster answers... Read moreIndustry
view channel
Integrated DNA Technologies Expands into Clinical Diagnostics
Integrated DNA Technologies (IDT; Coralville, Iowa, USA) has announced the launch of Archer FUSIONPlex-HT Dx and VARIANTPlex-HT Dx. This launch marks the company’s first in vitro diagnostic (IVD) offerings... Read more