Diasorin and Fisher Scientific Enter into US Distribution Agreement for Molecular POC Platform
Posted on 29 Jan 2026
Diasorin (Saluggia, Italy) has entered into an exclusive distribution agreement with Fisher Scientific, part of Thermo Fisher Scientific (Waltham, MA, USA), for the LIAISON NES molecular point-of-care (POC) platform in the U.S. hospital market. This follows the recent 510(k) clearance and CLIA-waiver received by Diasorin from the U.S. Food and Drug Administration (FDA) for its first assay—the FLU A/B, RSV & COVID-19 Panel for use on the LIAISON NES platform.
The LIAISON NES is a next-generation molecular POC system designed to expand access to high-quality diagnostic testing in decentralized settings. Its fully automated workflow requires only a few seconds of hands-on time and delivers results in approximately 15 minutes, enabling timely clinical decisions while reducing dependence on specialized laboratory personnel.
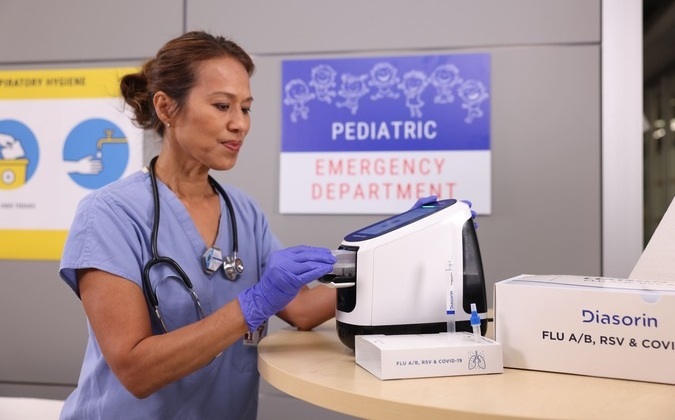
The U.S. represents one of the largest global markets for molecular respiratory testing, with over 7,000 hospitals and approximately 130,000 physician office laboratories (POLs) performing such diagnostics. Fisher Scientific’s strong footprint and established relationships across the hospital channel make it a strategic partner for the introduction and scale-up of the LIAISON NES platform and its respiratory panel.
The agreement strengthens Diasorin’s commercial capabilities in the hospital segment, complementing the company’s existing U.S. commercial organization, which today promotes its molecular solutions, including LIAISON MDX and LIAISON PLEX, as well as the recently expanded sales force dedicated to the LIAISON NES program.
“This agreement will accelerate our entry into the hospital segment and support access to major U.S. Integrated Delivery Networks (IDNs),” said Carlo Rosa, CEO of Diasorin. “We are also working to finalize additional distribution agreements with others in the United States aimed at providing full coverage of the non-acute market segment, which includes more than 130,000 POLs, ensuring a comprehensive and efficient go-to-market strategy across all key customer categories.”
Related Links:
Diasorin
Thermo Fisher Scientific














