Eurofins Launches New Multiplex PCR Assay for Rapid Detection of B.1.617 Kappa SARS-CoV-2 Variant
|
By LabMedica International staff writers Posted on 04 Jun 2021 |

Image: GSD NovaType III SARS-CoV-2 RT-PCR assay (Photo courtesy of Eurofins Technologies)
Eurofins Technologies (Ebersberg, Germany) has launched its GSD NovaType III SARS-CoV-2 RT-PCR assay, developed for the rapid detection of SARS-CoV-2 Variants of Concern including B.1.617 (India), B.1.427/B.1.429 (California/USA), B.1.351 (South Africa) or P.1 (Brazil).
The assay facilitates the identification of the relevant mutations E484Q, E484K and L452R in one reaction combined with the simultaneous discrimination from the S gene E484 wildtype variant. These mutations have been associated with reports of potential decreased efficacy of certain vaccines and an increase in the transmissibility of the virus.
GSD NovaType III SARS-CoV-2 is the latest addition to the GSD NovaType product line, a continuously updated panel of single and multiplex assays for the screening and detection of emerging Variants of Concern (VOC) and of Interest (VOI) on positive samples. These assays may play an important role in the fight against COVID-19 by enabling the rapid identification of new variants that may endanger the success of global vaccination campaigns and the implementation of enhanced isolation, testing and vaccination measures in affected areas. GSD NovaType III SARS-CoV-2 will be available as a research-use-only kit by the June 9. A CE marked version is expected by the second half of June.
The assay facilitates the identification of the relevant mutations E484Q, E484K and L452R in one reaction combined with the simultaneous discrimination from the S gene E484 wildtype variant. These mutations have been associated with reports of potential decreased efficacy of certain vaccines and an increase in the transmissibility of the virus.
GSD NovaType III SARS-CoV-2 is the latest addition to the GSD NovaType product line, a continuously updated panel of single and multiplex assays for the screening and detection of emerging Variants of Concern (VOC) and of Interest (VOI) on positive samples. These assays may play an important role in the fight against COVID-19 by enabling the rapid identification of new variants that may endanger the success of global vaccination campaigns and the implementation of enhanced isolation, testing and vaccination measures in affected areas. GSD NovaType III SARS-CoV-2 will be available as a research-use-only kit by the June 9. A CE marked version is expected by the second half of June.
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel
AI-Based Blood Test Diagnose Multiple Brain Disorders from Blood Sample
Diagnosing the cause of age-related cognitive symptoms remains challenging because clinical presentations of neurodegenerative diseases often overlap, and multiple pathologies can co-occur... Read more
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read moreMolecular Diagnostics
view channel
RNA Profiling Uncovers Therapeutic Targets in Solid Tumors
Many patients with advanced solid tumors exhaust broad DNA panel testing yet still lack biomarkers that match guideline-recommended therapies, limiting access to targeted options. Expanding molecular profiling... Read more
Whole Genome Sequencing in Routine Care Expands Rare Disease Detection
Rare diseases often involve prolonged diagnostic journeys that delay clinical decision-making and complicate family planning. As phenotypes become more heterogeneous, sequencing-based methods are increasingly... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
New Bacterial Target Identified for Early Detection of Noma
Noma is a rapidly progressing orofacial infection that begins as gingivitis and can destroy oral and facial tissues, primarily affecting young children living in extreme poverty. Without treatment, it... Read more
Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
Streptococcus dysgalactiae subspecies equisimilis (SDSE) infections are increasing worldwide and include variants that may lead to severe disease. Researchers now report that whole-genome sequencing of... Read morePathology
view channel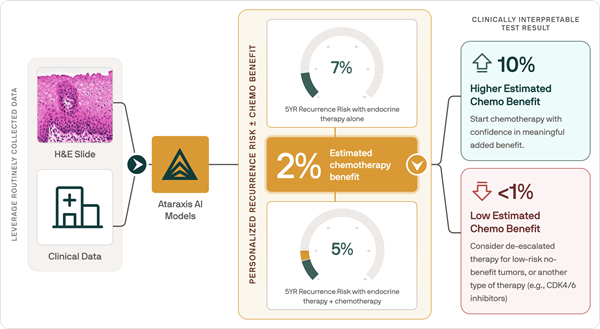
AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer is typically guided by recurrence risk and population-level averages rather than patient-specific benefit. However, existing clinicopathologic... Read more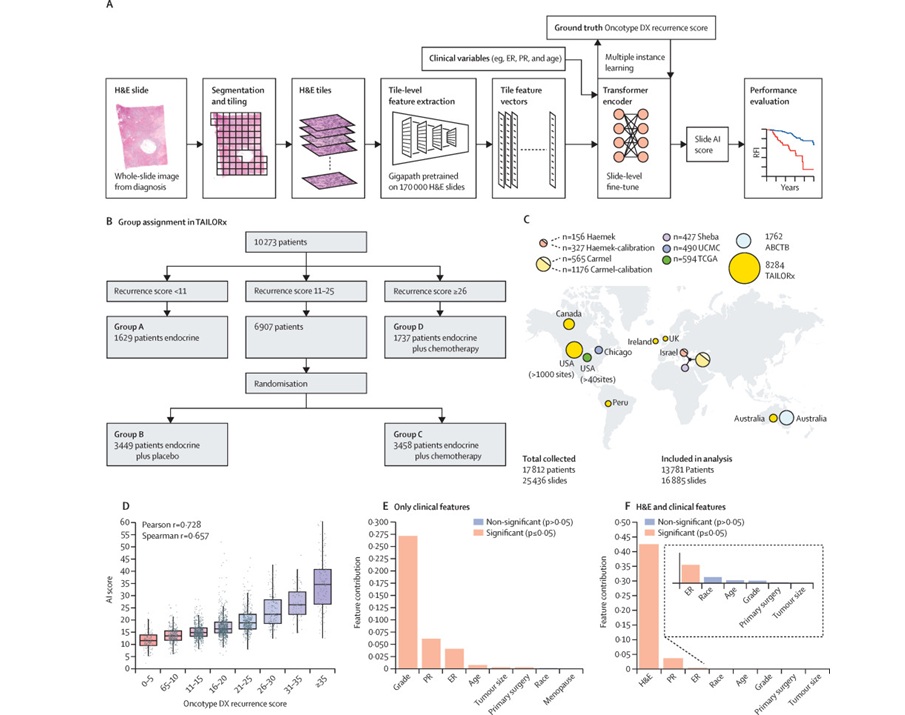
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Takara Bio USA and Hamilton Partner Partner to Automate NGS Library Preparation
Takara Bio USA, Inc. (San Jose, CA, USA), a wholly owned subsidiary of Takara Bio Inc., and Hamilton Company (Reno, NV, USA) announced a development and co-marketing agreement to deliver integrated, automated... Read more