Fujirebio Europe Partners with Centogene to Provide Rapid and High-Quality Preventive SARS-CoV-2 Antigen Testing
|
By LabMedica International staff writers Posted on 10 Nov 2020 |
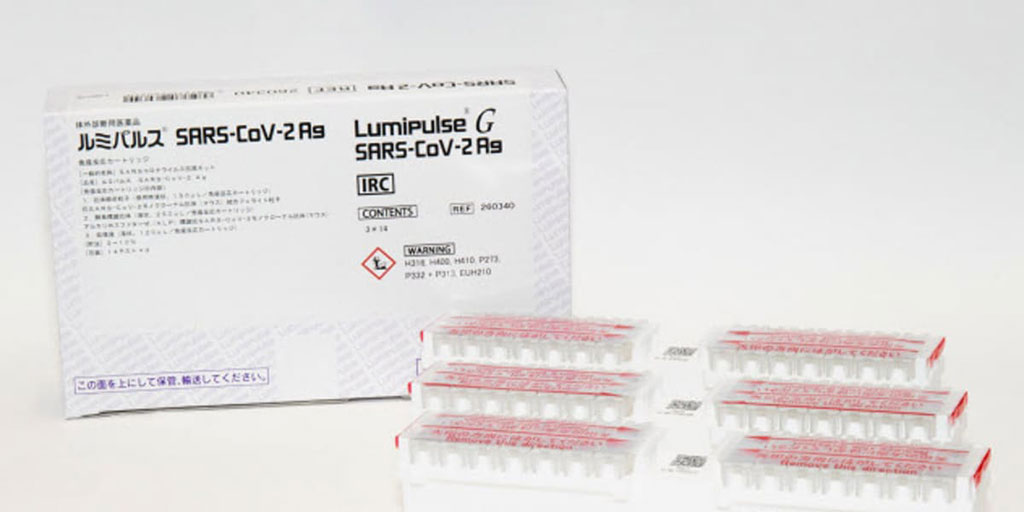
Image: Lumipulse G SARS-CoV-2 Ag (Photo courtesy of Fujirebio)
Fujirebio Europe (Ghent, Belgium) and Centogene N.V. (Rostock, Belgium) have entered into a partnership to leverage LUMIPULSE G technology for large-scale COVID-19 antigen testing with further deployment in German airports.
Lumipulse G SARS-CoV-2 Ag was the first high-sensitive antigen assay launched on a fully automated chemiluminescent platform. It is used on a LUMIPULSE G system for quantitative measurement of SARS-CoV-2 antigen in human nasopharyngeal swab fluid or saliva. It is used as an aid in the diagnosis of a SARS-CoV-2 infection and utilizes proven CLEIA (chemiluminescent enzyme immunoassay) technology. The assay generates fast results (available in up to 40 minutes from sample receipt in the laboratory) using different sample types, including less invasive saliva samples.
Lumipulse G SARS-CoV-2 Ag has been used by Japanese authorities since August this year for quarantine screening of arriving travelers in major international Japanese airports. Centogene has an exclusive cooperation with Lufthansa. As part of the agreement, Fujirebio will supply Lumipulse G SARS-CoV-2 Ag assays running on fully automated CLEIA-based LUMIPULSE G1200 instruments. Sample collection is to be performed by Centogene via the usual oropharyngeal swabs, with the analysis of the samples performed at Centogene test laboratories, and the LUMIPULSE G1200 then supporting by delivering high quality results in up to only 40 minutes from sample receipt in the laboratory. These instruments will be an integrated part of the existing sophisticated automated Centogene workflow that ensures fast and direct delivery of results to customers on their smartphones or digital devices.
“We are very pleased that Centogene has chosen Fujirebio and the Lumipulse G platform to step up their current RT-PCR based COVID-19 testing,” said Christiaan De Wilde, CEO of Fujirebio Europe. “We are certain that the speed, high sensitivity and high quality of the Lumipulse G SARS-CoV-2 Ag testing solution combined with the flexibility of our platform to fit in the existing Centogene workflow will ensure a trustworthy and efficient testing offer.”
“Over the past months, we have seen an increasing need for easily accessible, reliable, and rapid coronavirus tests,” said Dr. Volkmar Weckesser, Chief Information Officer at Centogene. “We have been able to establish a blueprint for this - having set up test centers at four of Germany’s largest airports in the last six months. We are very excited to now expand on these services with the support of Fujirebio’s advanced technology. With this highly validated, customer-friendly solution, we hope to play an integral role in preventing further outbreaks.”
Lumipulse G SARS-CoV-2 Ag was the first high-sensitive antigen assay launched on a fully automated chemiluminescent platform. It is used on a LUMIPULSE G system for quantitative measurement of SARS-CoV-2 antigen in human nasopharyngeal swab fluid or saliva. It is used as an aid in the diagnosis of a SARS-CoV-2 infection and utilizes proven CLEIA (chemiluminescent enzyme immunoassay) technology. The assay generates fast results (available in up to 40 minutes from sample receipt in the laboratory) using different sample types, including less invasive saliva samples.
Lumipulse G SARS-CoV-2 Ag has been used by Japanese authorities since August this year for quarantine screening of arriving travelers in major international Japanese airports. Centogene has an exclusive cooperation with Lufthansa. As part of the agreement, Fujirebio will supply Lumipulse G SARS-CoV-2 Ag assays running on fully automated CLEIA-based LUMIPULSE G1200 instruments. Sample collection is to be performed by Centogene via the usual oropharyngeal swabs, with the analysis of the samples performed at Centogene test laboratories, and the LUMIPULSE G1200 then supporting by delivering high quality results in up to only 40 minutes from sample receipt in the laboratory. These instruments will be an integrated part of the existing sophisticated automated Centogene workflow that ensures fast and direct delivery of results to customers on their smartphones or digital devices.
“We are very pleased that Centogene has chosen Fujirebio and the Lumipulse G platform to step up their current RT-PCR based COVID-19 testing,” said Christiaan De Wilde, CEO of Fujirebio Europe. “We are certain that the speed, high sensitivity and high quality of the Lumipulse G SARS-CoV-2 Ag testing solution combined with the flexibility of our platform to fit in the existing Centogene workflow will ensure a trustworthy and efficient testing offer.”
“Over the past months, we have seen an increasing need for easily accessible, reliable, and rapid coronavirus tests,” said Dr. Volkmar Weckesser, Chief Information Officer at Centogene. “We have been able to establish a blueprint for this - having set up test centers at four of Germany’s largest airports in the last six months. We are very excited to now expand on these services with the support of Fujirebio’s advanced technology. With this highly validated, customer-friendly solution, we hope to play an integral role in preventing further outbreaks.”
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel
Long-Term Data Show PSA Screening Modestly Reduces Prostate Cancer Deaths
Prostate cancer is among the most common cancers in men, and the role of population screening has remained controversial because of overdiagnosis and overtreatment. Health systems have sought clearer,... Read more
Urine-Based Nanosensor Tracks Lung Cancer and Fibrosis Noninvasively
Lung cancer remains difficult to monitor for early progression and treatment resistance, while pulmonary fibrosis continues to pose major challenges for early diagnosis. Clinicians need repeatable, noninvasive... Read moreMolecular Diagnostics
view channel
New Computational Tool Reveals Genetic Driver of Idiopathic Neuropathy
Peripheral neuropathy is a common neurological disorder that causes pain, sensory loss, imbalance, and weakness, affecting an estimated 12%–20% of people in the U.S. and nearly 30% of adults over age 65.... Read more
Breast Cancer-Specific Signatures Link Genome Instability to Outcomes
Genomic instability is a hallmark of cancer, but most genomic analyses have relied on broad signatures shared across multiple malignancies, limiting their precision for individual tumor types.... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read morePathology
view channel
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read more
AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
Non–muscle invasive bladder cancer has highly variable outcomes, complicating surveillance and treatment planning. Risk assessment typically relies on stage, grade, and tumor size, leaving uncertainty... Read moreTechnology
view channel
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read more
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read moreIndustry
view channel
AI-Powered Multi-Functional Analyzer Wins German Innovation Award
Hematology services are increasingly delivered across distributed care settings, where limited staffing and complex workflows can extend turnaround times. Advanced morphology review still often depends... Read more