First-of-Its-Kind Saliva-Based Rapid Test Uses DNA Aptamer to Detect COVID-19
|
By LabMedica International staff writers Posted on 18 May 2022 |
As the world moves beyond the acute phase of the pandemic amidst the slow rate of vaccination in developing countries, the combination of ultra-low cost, highly accurate testing and rising availability of therapeutics may present an option to alleviate the effects of COVID-19 for the vast majority of people so that everyone can continue with their daily lives. Now, a newly-launched affordable, saliva-based rapid test uses a DNA aptamer instead of an antibody to detect COVID-19 and is read by a UV Spectrophotometer. DNA aptamers are believed to offer a structural advantage over other enzyme-based approaches in sensitivity, specificity, quality of production, lower costs, test formats and other areas.
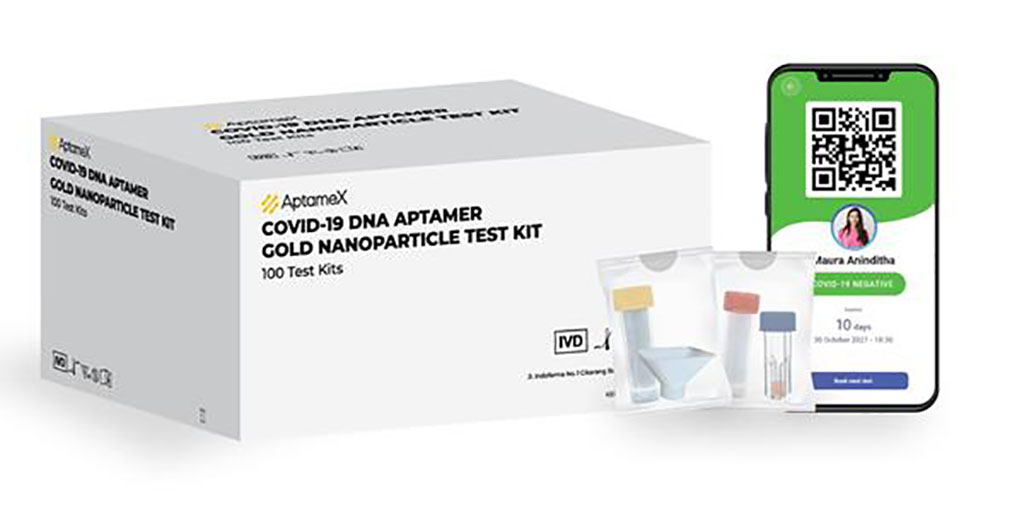
The AptameX COVID-19 rapid test from Achiko AG (Zurich, Switzerland) is an innovative rapid test that uses DNA aptamers instead of antigens to detect the SARS-CoV-2 virus. Aptamers are single-stranded DNA or RNA oligonucleotides that bind to specific targets via shape recognition with high affinity and specificity. As aptamers are synthetic, they can be produced at a low cost and in scale. By using aptamer technology, the company has been able to design a friendlier and better customer experience that achieves higher degrees of accuracy than nearly all other rapid tests and at a lower cost to the end consumer.
The AptameX COVID-19 rapid test comes in the form of a user-friendly, saliva-based rapid test. First, a patient rinses their mouth with a standard non-alcoholic dental mouthwash, followed by purified water. The patient then spits into a sample collection tube which is then processed by a lab. The sample is diluted in water and added to a cuvette containing reagent. After 10 minutes, a salt quench is added and after one minute, the sample is ready to be scanned by a standard UV-spectrophotometer. The rapid test is separated into the sampling and software, plus the spectrophotometer. Future iterations may be packaged with other approved spectrophotometers to provide a more convenient solution.
Through differentiated chemistry, Achiko has been able to develop a user-friendly, saliva-based test that is sensitive at the important higher CT values. In a clinical environment, the company had successfully classified results when compared against a PCR test, at above 97% sensitivity and 97% specificity at an average CT score of 28, substantially higher than what many rapid tests are reporting. Achiko has successfully registered AptameX under the European Union’s Medical Device Research (MDR) and In Vitro Diagnostic Regulation (IVDR), and has obtained a CE Mark that grants it access to 27 countries in the European Union for its COVID-19 AptameX test and system, establishing a foundation for applying for regulatory approval across the world.
“This is the first major mainstream use of DNA aptamers in a consumer testing product and a fantastic milestone for Achiko,” stated Steven Goh, CEO of Achiko AG. “This matters as the world moves to treating COVID-19 as endemic, key sectors such as aged care, healthcare, manufacturing, and many others need to find a way to cope with COVID’s challenges. PCR testing whilst accurate is often impractical in an endemic setting, and recent research indicates that many rapid tests are simply not sensitive enough to detect the more recent variants.”
Related Links:
Achiko AG
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read more
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read moreMolecular Diagnostics
view channel
Blood-Based Proteomic Test May Predict Treatment Response in Non-Small Cell Lung Cancer
Lung cancer remains the leading cause of cancer death, with non-small cell lung cancer (NSCLC) accounting for most cases. Treatment decisions are often made without a clear indication of how a patient... Read more
Ultrasensitive MRD Blood Test Detects Early Breast Cancer Recurrence
SAGA Diagnostics (Morrisville, NC, USA), a company specializing in tumor-informed, blood-based cancer detection and precision medicine, announced the publication of a new study evaluating its Pathlight... Read more
Position Statements Outline Evidence Standards for Multi-Cancer Detection Tests
Cancer screening is intended to reduce mortality, but policy decisions often depend on early indicators that may not fully reflect true survival benefit. The emergence of blood-based tests capable of detecting... Read moreHematology
view channel
Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
Iron deficiency in school-age children can affect brain development, learning, growth, and physical performance, yet early deficiency may be missed when screening focuses mainly on anemia.... Read more
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read moreImmunology
view channel
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read more
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read more
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read moreTechnology
view channel
Mailed Screening Kits Help Reduce Colorectal Cancer Screening Gaps
Colorectal cancer screening is a longstanding preventive priority, yet participation and follow-up remain uneven across patient groups. Safety‑net primary care settings often face barriers that limit screening... Read more
Algorithm Panel Aids Liver Fibrosis Assessment and Liver Cancer Surveillance
Chronic liver disease is common and often progresses silently, increasing the risk of cirrhosis and hepatocellular carcinoma when not detected early. With an estimated 1.5 billion people affected worldwide... Read moreIndustry
view channelWerfen and Oxford Nanopore Collaborate on Transplant Assay Development
Werfen, a company focused on specialized diagnostics, has announced a strategic collaboration with Oxford Nanopore Technologies, which develops nanopore-based sequencing technology, to validate Werfen’s... Read more