First-Ever CRISPR-Based POC System Delivers COVID-19 Test Results in 18 Minutes
|
By LabMedica International staff writers Posted on 11 Apr 2022 |

A CRISPR-based molecular point-of-care (POC) COVID-19 test may be used to deliver accurate, actionable, lab-quality results for SARS-CoV-2 in as little as 18 minutes.
Proof Diagnostics, Inc. (Cambridge, MA, USA) has announced the submission of its Emergency Use Authorization (EUA) request for review to the U.S. Food and Drug Administration (FDA) for its POC molecular diagnostic test for COVID-19 (SARS-CoV-2). If authorized, the Proof Lab Test System would be the first POC system that employs CRISPR-based nucleic acid detection to offer a cost-effective, user-friendly, and easily scalable test. The company's hope is to allow clinicians and patients to achieve laboratory-level sensitivity and specificity at the POC in as little as 18 minutes.
The Proof Lab Test System comprises small, modular and portable equipment that is easy to set up and operate. The low-cost reader and cartridges make testing scalable. The system uses highly sensitive detection methods and gold-standard specificity to rapidly complete the process from sample collection to results in as little as 22 minutes.
Additionally, the company is designing the Proof Diagnostics mobile app to seamlessly connect with up to six Proof Lab readers via Bluetooth. This will allow users to monitor the progress of multiple COVID-19 tests, keep and view up to 50 results filtered by time and date on each synced reader, and easily export detailed test results. The company is also designing its app to enable third-party integrations - to EHRs, for example - and the storage of large volumes of data in its HIPAA-compliant cloud solution.
"The delivery of novel chemistry, combined with Proof's innovative technology platform, to FDA EUA submission in less than two years is no small feat. CRISPR has long promised to make a difference in peoples' lives and we are incredibly proud of the scientific innovation that our team developed. We hope that the FDA authorizes our CRISPR-based test so that we can empower communities to navigate their next step in person with confidence and peace of mind," said Omar Abudayyeh, co-founder of Proof Diagnostics.
"Submitting our EUA to the Food and Drug Administration is a significant achievement for Proof Diagnostics as we embark on the future of rapid POC and at-home testing, ensuring optimal accuracy to help detect disease prior to the demonstration of symptoms. The Proof Lab Test System has the potential to become a valuable tool for clinicians and patients to make more informed health decisions that can help deter the spread of disease," said Sid Shenai, co-founder and CEO of Proof Diagnostics."
Related Links:
Proof Diagnostics, Inc.
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel
Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
Alzheimer’s disease is the most common cause of dementia, yet confirmatory testing remains invasive and hard to access. Diagnosis currently takes an average of 3.5 years, and about 75% of people with dementia... Read more
FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
Accurate evaluation of androgen status often requires concordant measurement of total testosterone, free testosterone, and sex hormone‑binding globulin. Reference methods such as equilibrium dialysis with... Read moreMolecular Diagnostics
view channel
Realistic Mock Samples Aim to Speed Cervical Cancer Test Development
Cervical cancer remains highly preventable, yet screening access is limited in many low- and middle-income settings. Gold-standard tests for high-risk human papillomavirus (HPV) detect viral DNA or messenger... Read more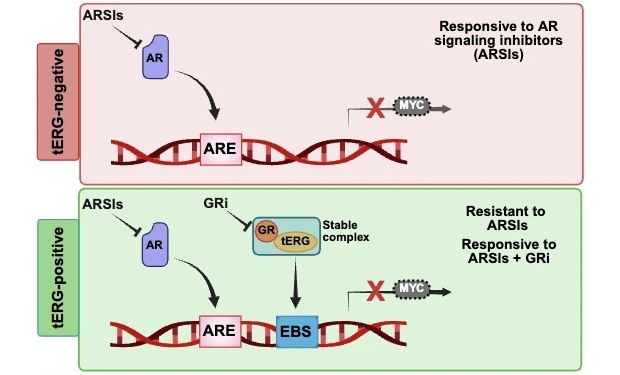
Molecular Marker Identifies Hormone Therapy Resistance Pathway in Prostate Cancer
Most prostate cancers depend on androgen signaling, making hormone suppression or blockade a central treatment strategy. Although many patients respond initially, tumors often adapt and eventually progress,... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read moreMicrobiology
view channel
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read more
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more