Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
|
By LabMedica International staff writers Posted on 01 Nov 2022 |
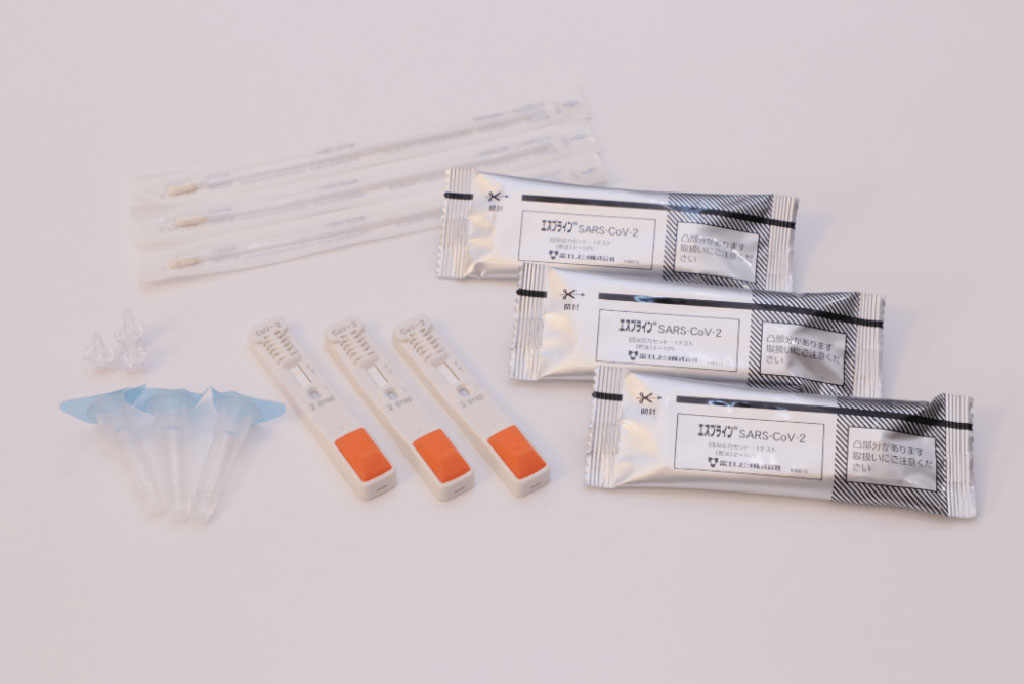
Many health care providers are hopeful regarding COVID-19 testing programs that use accurate rapid antigen tests to reduce the burden on the health care system through early diagnosis of infection. Currently, various antigen-detecting rapid diagnostic tests (Ag-RDTs) for SARS-CoV-2 are commercially available.
The widespread use of testing outside medical institutions suggests that anterior nasal specimens, which can be safely self-collected by the patient, are now being used instead of nasopharyngeal specimens collected by medical staff. Therefore, to evaluate the usefulness of specimens collected from the anterior nasal cavity, understanding the viral load and diagnostic accuracy of tests performed using anterior nasal specimens is vital.
Pediatricians at the Jichi Medical University (Tochigi, Japan) and their colleagues conducted a prospective cross-sectional study from February 12, 2021 to September 30, 2021. They enrolled 16 patients (age range, <1 month-76 years) and samples were collected simultaneously from anterior nasal and nasopharyngeal sites continuously during hospitalization. The primary end points were the diagnostic accuracy of the Ag-RDT and utility of anterior nasal specimens.
The scientists determined the diagnostic performance of the RapidTesta SARS-CoV-2 test (Sekisui Medical Co., Ltd., Tokyo, Japan) and compare it with that of RT-PCR, using specimens collected from the anterior nasal and nasopharyngeal sites. Furthermore, ESPLINE SARS-CoV-2 (Fujirebio Co., Ltd., Tokyo, Japan), an Ag-RDT that has been marketed and widely used since May 2020, was used as a comparator representing the efficacy of common Ag-RDTs.
Four specimen swabs were taken simultaneously from each patient on admission: an anterior nasal and nasopharyngeal specimen from each nostril and also repeat samples. To judge the RapidTesta SARS-CoV-2 results objectively, the team evaluated the results using the RapidTesta Reader after visual assessment. RNA extraction was performed using the QIAamp Viral RNA Mini Kit (Qiagen, Hilden, Germany) with a 140-µL aliquot of each sample. A one-step RT-PCR (Thermo Fisher Scientific, Waltham, MA, USA) was performed which targets nucleotide 2 gene-specific primers for SARS-CoV-2.
The investigators reported that in total 226 sets of paired samples were obtained. In 88.2% of specimens, the viral load was high at the nasopharyngeal site. The mean cycle threshold values for the anterior nasal and nasopharyngeal sites were 32.4 and 29.9, respectively. Using the real-time polymerase chain reaction results as a reference, the Ag-RDT showed a 100% sensitivity up to day 6 of the illness, using specimens with moderate or high viral load (cycle threshold <30) from either site. From day 7, the sensitivity was 70.4% - 90.6% and 83.9% - 84.6% for the anterior nasal and nasopharyngeal sites, respectively. The specificity remained at 100%.
The authors concluded that they had demonstrated the diagnostic accuracy of the newly developed RapidTesta SARS-CoV-2 test for the accurate diagnosis of COVID-19 in terms of both sensitivity and specificity. The anterior nasal cavity was shown to be useful as a specimen collection site. The study was published on November 1, 2022 in the International Journal of Infectious Diseases.
Related Links:
Jichi Medical University
Sekisui Medical Co., Ltd
Fujirebio
Thermo Fisher Scientific
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
- Siemens Launches New Combination Tests for SARS-CoV-2 and Seasonal Respiratory Pathogens
Channels
Clinical Chemistry
view channel
Blood Test Detects Testicular Cancer Missed by Standard Markers
Testicular cancer most often affects adolescents and young adults and is highly treatable when found early. Diagnosis can be difficult when tumors do not produce sufficient levels of standard blood-based... Read more
Routine Blood Tests Identify Biomarkers Linked to PTSD
Post-traumatic stress disorder (PTSD) is associated with a range of chronic physical health conditions and affects multiple organ systems. Clinical laboratories routinely measure blood analytes that reflect... Read moreMolecular Diagnostics
view channel
CRISPR-Based Test Identifies Multiple Respiratory Viruses Simultaneously
Respiratory virus co-circulation complicates differential diagnosis, as overlapping symptoms can obscure etiology. Multiplex testing typically depends on multiple enzymes or fluorophores and multistep... Read more
Multi-Omic Assay Predicts Recurrence and Radiation Benefit in Early Breast Cancer
Early-stage invasive breast cancer is frequently managed with breast-conserving surgery followed by adjuvant radiation therapy, but the magnitude of benefit from radiation varies among patients.... Read more
Portable Test Detects Tuberculosis from Tongue Swabs in 30 Minutes
Despite decades of effective drug regimens, tuberculosis remains the leading cause of death from an infectious disease, driven in part by limited access to accurate, rapid testing. Conventional diagnostics... Read more
Blood Test Receives FDA Breakthrough Status to Differentiate Schizophrenia and Bipolar Disorder
Differentiating schizophrenia from bipolar I disorder in symptomatic patients is challenging because early presentations often overlap. Current diagnostic pathways rely heavily on subjective assessments... Read moreHematology
view channel
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read more
Blood Test Enables Early Detection of Multiple Myeloma Relapse
Bone marrow biopsies remain central to diagnosing and monitoring multiple myeloma, yet the procedure is painful, invasive, and often repeated over time. Older patients—who represent most new cases—can... Read moreImmunology
view channel
Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
Sepsis remains a time-critical condition in which rapid risk assessment is often hindered by reliance on centralized laboratory testing. The global burden is substantial, with an estimated 166 million... Read more
Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
Accurate laboratory diagnosis of early Lyme disease remains challenging because serologic responses may be limited soon after infection. Missed detection at this stage can delay evaluation and management... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
Diabetic kidney disease is a leading cause of chronic kidney disease and end-stage kidney disease, affecting 20%–40% of people with diabetes and more than 107 million individuals worldwide as of 2021.... Read more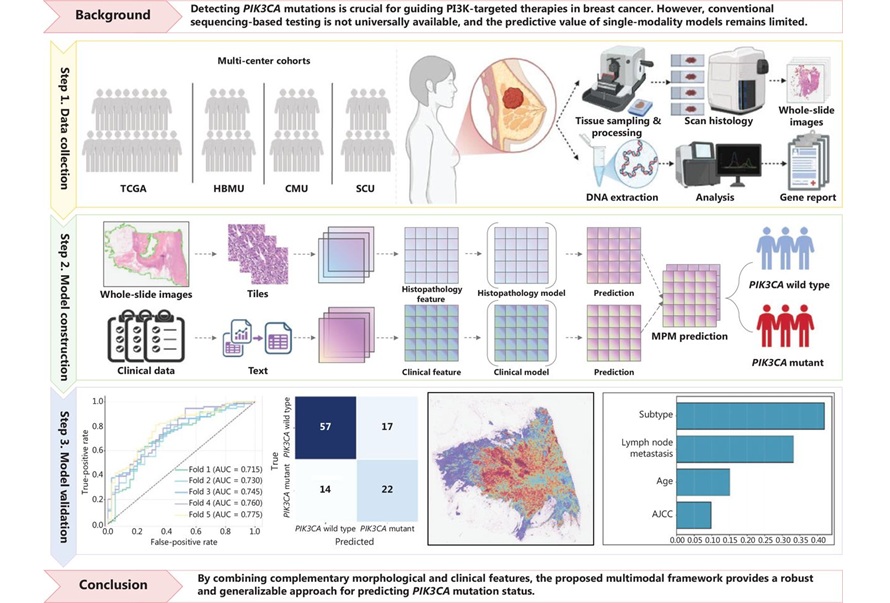
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read moreTechnology
view channel
Microfluidic Single-Cell Assay Predicts Breast Cancer Risk
Risk stratification for breast cancer remains imprecise, as population-based models and breast density can over- or underestimate individual risk, potentially leading to over- or under-screening.... Read more