Immunochromatographic Strip Test Developed for Rift Valley Fever
|
By LabMedica International staff writers Posted on 03 Oct 2019 |
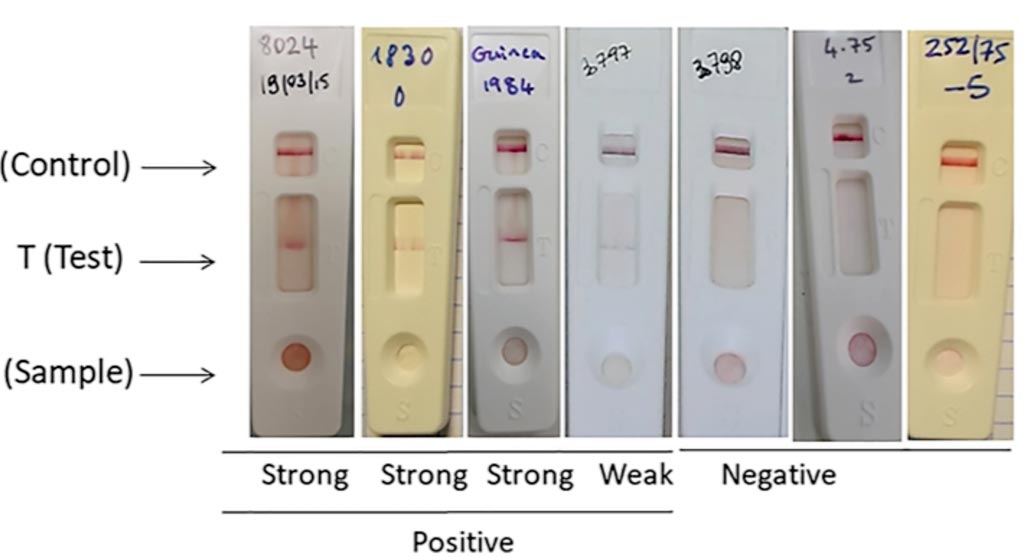
Image: Rift Valley Fever (RVF) lateral flow immunochromatographic (LFT) strip test for the detection of RVF infection using the two selected monoclonal antibodies (Photo courtesy of University of Montpellier).
Rift Valley fever (RVF) is an emerging mosquito-borne disease that affects a wide range of animals and human beings in Africa and the Arabian Peninsula. A rapid and specific test for RVF diagnosis at the site of a suspected outbreak is crucial for the implementation of control measures.
Patients who become ill usually experience fever, generalized weakness, back pain, and dizziness at the onset of the illness. Typically, patients recover within two days to one week after onset of illness. However, a small percentage (8-10%) of people infected with RVFV develops much more severe symptoms. There is a growing concern that RVFV will extend its current range due to the wide variety of mosquito species able to transmit to several mammal hosts.
Scientists at the University of Montpellier (Montpellier, France) and their colleagues developed a first-line lateral flow immunochromatographic strip test (LFT) for the detection of the nucleoprotein (N) of the RVF virus (RVFV). Its diagnostic performance characteristics were evaluated using reference stocks isolates recovered from different hosts and in geographic regions mimicking clinical specimens and from known RVF negative serum samples.
The test strip was constructed on the principles of immunochromatography using colloidal-gold-labeled monoclonal antibodies (Mabs). The team used the two Mabs generated against the N protein of RVFV: the Mab 8E10-4A4 gold conjugate and the Mab 10H3-4E4-3D5. Mab 10H3-4E4-3D5 was immobilized onto a nitrocellulose membrane for the test line zone and rabbit anti-mouse antibodies were immobilized for the control line zone to capture unbound Mab. The test (T) and control (C) lines were checked for the development of color after 10 minutes and again after 30 minutes as it might take longer time for weak positives to form a visual band scored subjectively from negative to strong.
A total of 25 isolated strains mimicking clinical specimens of different geographical origins and 10 clinical samples originating from an ongoing outbreak in 2019 detected RVF positive by a TaqMan RT-PCR technique which is considered as the current reference detection system were also detected positive by the LFT giving a diagnostic sensitivity of 100%. Diagnostic specificity (DSp) was assessed on 160 serum samples known to be negative for RVFV by sero-neutralization test and cELISA but collected in tropical countries where RVF has been circulating to detect possible cross reactions or in other areas where RVF introduction is considered at risk. The test gave a DSp of 98.81% and there was an absence of cross-reactivity with viruses belonging to different genera.
The authors concluded that the specificity and sensitivity of the evaluated test are lower than the ones of molecular-based techniques (LAMP, PCR) but are adequate for specific rapid initial detection of RVF outbreaks or disease surveillance in control programs. The fact no specialized reagents and laboratory equipment are needed, make this assay a valuable, first-line diagnostic tool in resource-poor diagnostic territories for on-site RVFV detection, however the staff require training. The study was published on September 11, 2019, in the journal PLOS NTD.
Related Links:
University of Montpellier
Patients who become ill usually experience fever, generalized weakness, back pain, and dizziness at the onset of the illness. Typically, patients recover within two days to one week after onset of illness. However, a small percentage (8-10%) of people infected with RVFV develops much more severe symptoms. There is a growing concern that RVFV will extend its current range due to the wide variety of mosquito species able to transmit to several mammal hosts.
Scientists at the University of Montpellier (Montpellier, France) and their colleagues developed a first-line lateral flow immunochromatographic strip test (LFT) for the detection of the nucleoprotein (N) of the RVF virus (RVFV). Its diagnostic performance characteristics were evaluated using reference stocks isolates recovered from different hosts and in geographic regions mimicking clinical specimens and from known RVF negative serum samples.
The test strip was constructed on the principles of immunochromatography using colloidal-gold-labeled monoclonal antibodies (Mabs). The team used the two Mabs generated against the N protein of RVFV: the Mab 8E10-4A4 gold conjugate and the Mab 10H3-4E4-3D5. Mab 10H3-4E4-3D5 was immobilized onto a nitrocellulose membrane for the test line zone and rabbit anti-mouse antibodies were immobilized for the control line zone to capture unbound Mab. The test (T) and control (C) lines were checked for the development of color after 10 minutes and again after 30 minutes as it might take longer time for weak positives to form a visual band scored subjectively from negative to strong.
A total of 25 isolated strains mimicking clinical specimens of different geographical origins and 10 clinical samples originating from an ongoing outbreak in 2019 detected RVF positive by a TaqMan RT-PCR technique which is considered as the current reference detection system were also detected positive by the LFT giving a diagnostic sensitivity of 100%. Diagnostic specificity (DSp) was assessed on 160 serum samples known to be negative for RVFV by sero-neutralization test and cELISA but collected in tropical countries where RVF has been circulating to detect possible cross reactions or in other areas where RVF introduction is considered at risk. The test gave a DSp of 98.81% and there was an absence of cross-reactivity with viruses belonging to different genera.
The authors concluded that the specificity and sensitivity of the evaluated test are lower than the ones of molecular-based techniques (LAMP, PCR) but are adequate for specific rapid initial detection of RVF outbreaks or disease surveillance in control programs. The fact no specialized reagents and laboratory equipment are needed, make this assay a valuable, first-line diagnostic tool in resource-poor diagnostic territories for on-site RVFV detection, however the staff require training. The study was published on September 11, 2019, in the journal PLOS NTD.
Related Links:
University of Montpellier
Latest Microbiology News
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Blood-Based Viral Signature Identified in Crohn’s Disease
- Hidden Gut Viruses Linked to Colorectal Cancer Risk
- Three-Test Panel Launched for Detection of Liver Fluke Infections
- Rapid Test Promises Faster Answers for Drug-Resistant Infections
Channels
Clinical Chemistry
view channel
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read more
Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
Routine blood tests that measure low-density lipoprotein (LDL), commonly known as “bad” cholesterol, are widely used to guide lipid-lowering therapy, but they do not always provide a complete picture of... Read more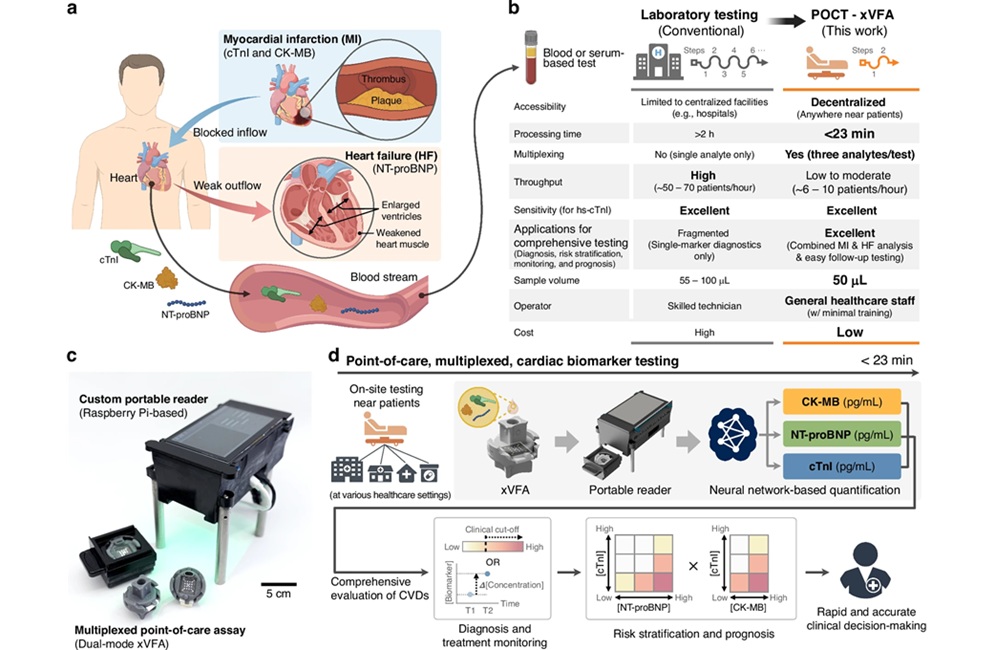
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel
Blood-Based Epigenetic Signals Enable Osteosarcoma Disease Monitoring
Osteosarcoma is a rare but aggressive pediatric bone cancer where recurrence and metastasis remain difficult to detect early. Imaging-based surveillance can miss small lesions and exposes children to repeated... Read more
Host–Virus Genetic Interactions Drive Nasopharyngeal Cancer Risk
Epstein–Barr virus (EBV) infects more than 95% of adults worldwide, yet only a small fraction develops EBV‑associated cancers such as nasopharyngeal carcinoma. Explaining this divergence requires understanding... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read morePathology
view channel
Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
Colorectal cancer is common and often lethal, and therapeutic decision-making is complicated by heterogeneous tumor microenvironments. Immunotherapy benefits only a small subset of patients, around 5%,... Read more
Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
Antibody-drug conjugates (ADC) are reshaping oncology, yet scalable biomarkers that reliably predict which patients will benefit remain limited as treatment regimens and combinations grow more complex.... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
Partnership Expands Ultrasensitive WGS Assay for for Hematologic Malignancies and MRD Monitoring
Tempus AI and Predicta Biosciences announced the commercial expansion of a co-branded whole‑genome sequencing assay GenoPredicta, which is intended for comprehensive genomic characterization of hematologic... Read more