RT-PCR Evaluated for Diagnosis of Imported Schistosomiasis
|
By LabMedica International staff writers Posted on 03 Oct 2019 |
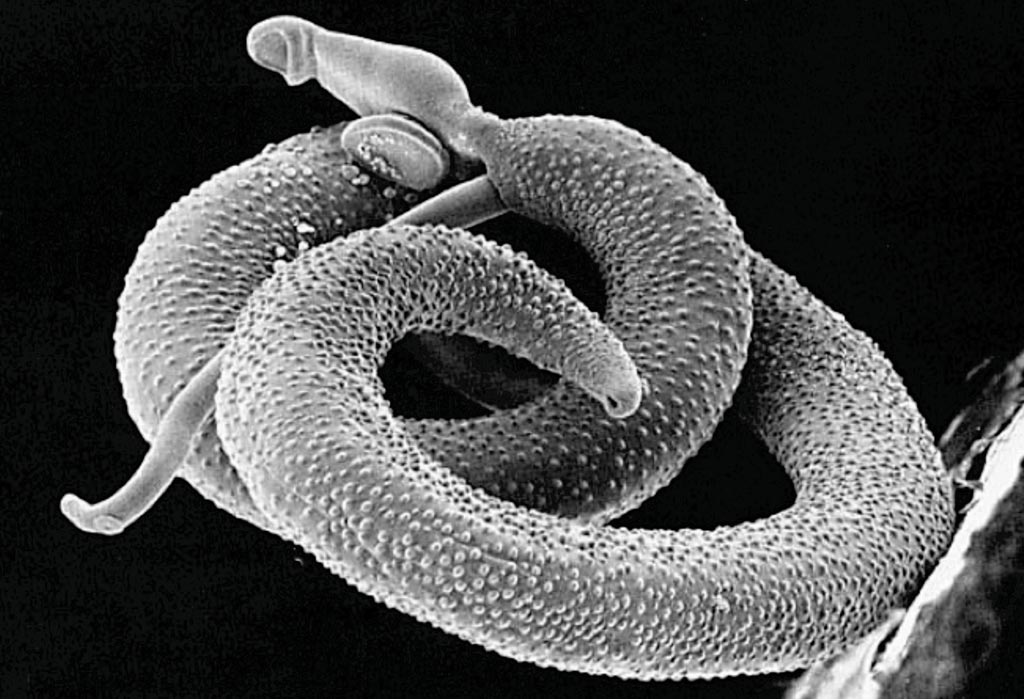
Image: Scanning electron micrograph of a pair of Schistosoma mansoni (Photo courtesy of the Davies Laboratory Uniformed Services University).
Schistosomiasis is one of the most important human parasitic neglected tropical diseases. It is a major source of morbidity and mortality in Africa but also in South America, the Caribbean, the Middle East, and Asia.
The diagnosis of schistosomiasis currently relies on microscopic detection of schistosome eggs in stool or urine samples and serological assays. The poor sensitivity of standard microscopic procedures performed in routine laboratories, makes molecular detection methods of increasing interest.
Scientists at the University of Rennes (Rennes, France) and their colleagues evaluated two in-house real-time Schistosoma polymerase chain reactions (RT-PCRs), targeting respectively S. mansoni [Sm] and S. haematobium [Sh] in excreta, biopsies and sera as potential tools to diagnose active infections and to monitor treatment efficacy. Schistosoma RT-PCRs were performed on 412 samples (124 urine, 86 stools, eight biopsies, 194 sera) from patients with suspected schistosomiasis.
Microscopic examination for the presence of Schistosoma eggs in urine samples was performed by examination of the pellet from micturition obtained after centrifugation. Fresh stool specimens were examined after two concentration techniques (flotation and diphasic methods). Sera were screened for anti-Schistosoma antibodies using the Schistosoma IgG ELISA or Schistosoma Antibody Detection Test Kit, and the Schistosomiasis Fumouze indirect hemagglutination (HA) test. Sera with a positive or a doubtful result by at least one technique were then confirmed using a Western Blot (WB) assay using the Schisto II Western Blot IgG kit.
The scientists reported that compared to microscopy, PCRs significantly increased the sensitivity of diagnosis, from 4% to 10.5% and from 33.7% to 48.8%, for Sh in urine and Sm in stools, respectively. The overall sensitivity of PCR on serum samples was 72.7% and reached 94.1% in patients with positive excreta (microscopy). The specificity of serum PCR was 98.9%. After treatment, serum PCR positivity rates slowly declined from 93.8% at day 30 to 8% at day 360, whereas antibody detection remained positive after one year.
The authors concluded that Schistosoma PCRs clearly outperform standard microscopy on stools and urine and could be part of reference methods combined with WB-based serology, which remains a gold standard for initial diagnosis. The study was published on September 11, 2019, in the journal PLOS Neglected Tropical Diseases.
Related Links:
University of Rennes
The diagnosis of schistosomiasis currently relies on microscopic detection of schistosome eggs in stool or urine samples and serological assays. The poor sensitivity of standard microscopic procedures performed in routine laboratories, makes molecular detection methods of increasing interest.
Scientists at the University of Rennes (Rennes, France) and their colleagues evaluated two in-house real-time Schistosoma polymerase chain reactions (RT-PCRs), targeting respectively S. mansoni [Sm] and S. haematobium [Sh] in excreta, biopsies and sera as potential tools to diagnose active infections and to monitor treatment efficacy. Schistosoma RT-PCRs were performed on 412 samples (124 urine, 86 stools, eight biopsies, 194 sera) from patients with suspected schistosomiasis.
Microscopic examination for the presence of Schistosoma eggs in urine samples was performed by examination of the pellet from micturition obtained after centrifugation. Fresh stool specimens were examined after two concentration techniques (flotation and diphasic methods). Sera were screened for anti-Schistosoma antibodies using the Schistosoma IgG ELISA or Schistosoma Antibody Detection Test Kit, and the Schistosomiasis Fumouze indirect hemagglutination (HA) test. Sera with a positive or a doubtful result by at least one technique were then confirmed using a Western Blot (WB) assay using the Schisto II Western Blot IgG kit.
The scientists reported that compared to microscopy, PCRs significantly increased the sensitivity of diagnosis, from 4% to 10.5% and from 33.7% to 48.8%, for Sh in urine and Sm in stools, respectively. The overall sensitivity of PCR on serum samples was 72.7% and reached 94.1% in patients with positive excreta (microscopy). The specificity of serum PCR was 98.9%. After treatment, serum PCR positivity rates slowly declined from 93.8% at day 30 to 8% at day 360, whereas antibody detection remained positive after one year.
The authors concluded that Schistosoma PCRs clearly outperform standard microscopy on stools and urine and could be part of reference methods combined with WB-based serology, which remains a gold standard for initial diagnosis. The study was published on September 11, 2019, in the journal PLOS Neglected Tropical Diseases.
Related Links:
University of Rennes
Latest Microbiology News
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Blood-Based Viral Signature Identified in Crohn’s Disease
- Hidden Gut Viruses Linked to Colorectal Cancer Risk
- Three-Test Panel Launched for Detection of Liver Fluke Infections
- Rapid Test Promises Faster Answers for Drug-Resistant Infections
- CRISPR-Based Technology Neutralizes Antibiotic-Resistant Bacteria
- Comprehensive Review Identifies Gut Microbiome Signatures Associated With Alzheimer’s Disease
- AI-Powered Platform Enables Rapid Detection of Drug-Resistant C. Auris Pathogens
- New Test Measures How Effectively Antibiotics Kill Bacteria
- New Antimicrobial Stewardship Standards for TB Care to Optimize Diagnostics
- New UTI Diagnosis Method Delivers Antibiotic Resistance Results 24 Hours Earlier
- Breakthroughs in Microbial Analysis to Enhance Disease Prediction
Channels
Clinical Chemistry
view channel
Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
Colorectal cancer (CRC) is a leading cause of cancer-related death worldwide, with more than 60% of cases still diagnosed at a late stage. Uptake of existing screening tools remains suboptimal,... Read more
Automated NfL Assay Supports Monitoring of Neurological Disorders
Neuroaxonal injury occurs across a wide range of neurological disorders and remains difficult to monitor noninvasively over time. Blood-based measurement of neurofilament light chain (NfL) provides a biologically... Read moreMolecular Diagnostics
view channel
New Respiratory Panel Expands Pathogen Detection to 25 Targets
Respiratory infections often present with overlapping symptoms, complicating differential diagnosis in acute and community settings. The stakes are higher for older adults, young children, and people with... Read more
Simple Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
Alzheimer’s disease affects millions worldwide but remains difficult to detect at its earliest, pre-symptomatic stage. Clinicians need tools that can identify biological changes before cognitive symptoms... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read more
Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
Early identification of treatment response and relapse remains a major challenge in solid tumors, where minimal residual disease is difficult to detect with routine imaging and blood tests.... Read morePathology
view channel
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read more
AI-Powered Tool to Transform Dermatopathology Workflow
Skin cancer accounts for the largest number of cancer diagnoses in the United States, placing sustained pressure on pathology services. Diagnostic interpretation can be variable for challenging melanocytic... Read moreTechnology
view channel
Online Tool Supports Family Screening for Inherited Cancer Risk
Genetic test results in oncology often have implications for relatives who may share inherited cancer risk. Many health systems lack structured processes to help patients alert family members, limiting... Read more
Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
Pneumonia is commonly confirmed with chest X-rays or laboratory assays that can take hours, delaying clinical decisions in acute and outpatient settings. Breath-based diagnostics promise faster answers... Read moreIndustry
view channel
Integrated DNA Technologies Expands into Clinical Diagnostics
Integrated DNA Technologies (IDT; Coralville, Iowa, USA) has announced the launch of Archer FUSIONPlex-HT Dx and VARIANTPlex-HT Dx. This launch marks the company’s first in vitro diagnostic (IVD) offerings... Read more