Simple Blood Test Can Detect Breast and Ovarian Cancer Risk without Genetic Sequencing
|
By LabMedica International staff writers Posted on 12 Jun 2023 |

The BRCA1 and BRCA2 genes, critical for DNA repair, can sometimes mutate and cause an increased risk for certain types of cancer, including breast, ovarian, pancreatic, and prostate cancer. In the US, those with a family history of these cancers may be advised to undergo genetic testing for mutations in these genes. If found positive, they could opt for additional screening or surgery to mitigate their risk. However, in the US, only 10% out of the estimated one million presumed carriers of BRCA1/2 mutations are aware of their status. Furthermore, in India, BRCA mutation tests are not widely accessible. A more efficient detection method could potentially allow many more individuals to become aware of their inherited high cancer risk.
Researchers from Dana-Farber Cancer Institute (Boston, MA, USA) and their collaborators have discovered an alternative method to identify increased cancer risk associated with BRCA1 and BRCA2 mutations, which doesn't involve genetic sequencing. Instead of searching for BRCA1/2 gene mutations, the assessment focuses on functional changes that take place when the pathway regulated by these genes is impaired. This innovative risk-detection technique could lay the groundwork for a more accessible, affordable, and potentially more comprehensive method of identifying an inherited risk for breast or ovarian cancer.
The researchers were able to identify increased risk via the presence of a specific group of microRNAs in the blood. MicroRNAs, short, non-coding, hairpin-shaped RNA molecules that circulate in our blood, are emerging as powerful disease biomarkers. The presence of a certain group of microRNAs indicates an active breakdown in the DNA repair process, similar to the distinctive knocking sound from a malfunctioning car engine. Conversely, the absence of this group suggests a working DNA repair process, as smooth as a purring engine.
In the proof-of-concept study, the researchers analyzed blood samples from 653 individuals sourced from six biorepositories located in the US, Poland, and India. Approximately half of the samples tested positive for BRCA mutations. Utilizing next-generation sequencing, the team identified the microRNAs in each sample and created a data set containing the microRNAs and BRCA mutation status for each sample. By applying machine learning, they were able to identify a unique microRNA pattern exclusively present in the samples positive for BRCA mutations. Their model correctly identified the presence of BRCA mutations 94% of the time, confirming this approach as a novel method to identify increased cancer risk.
While this proof-of-concept study does not form the foundation for a clinical test that could be included in an annual exam, it does hint at the feasibility of an affordable clinical test. PCR tests, such as those used for detecting COVID-19, could replace next-generation sequencing to detect the microRNA signature, thereby making it affordable and accessible. The researchers believe such as test could provide a more comprehensive risk assessment compared to genetic sequencing of the BRCA1 and BRCA2 genes, as it captures active signs of pathology, irrespective of the gene mutations causing it. Currently, genetic testing only detects known defects in a few specific genes that might disrupt the DNA repair process.
“What if we had an accessible and affordable test for inherited cancer risk that you can get done as an annual physical, the same way we test for diabetes or heart disease risk?” said senior author and Dana-Farber researcher Dipanjan Chowdhury, PhD. “These study results suggest that such a test is in the realm of possibility.”
Related Links:
Dana-Farber Cancer Institute
Latest Molecular Diagnostics News
- Diagnostic Device Predicts Treatment Response for Brain Tumors Via Blood Test
- Blood Test Detects Early-Stage Cancers by Measuring Epigenetic Instability
- Two-in-One DNA Analysis Improves Diagnostic Accuracy While Saving Time and Costs
- “Lab-On-A-Disc” Device Paves Way for More Automated Liquid Biopsies
- New Tool Maps Chromosome Shifts in Cancer Cells to Predict Tumor Evolution
- Blood Test Identifies Inflammatory Breast Cancer Patients at Increased Risk of Brain Metastasis
- Newly-Identified Parkinson’s Biomarkers to Enable Early Diagnosis Via Blood Tests
- New Blood Test Could Detect Pancreatic Cancer at More Treatable Stage
- Liquid Biopsy Could Replace Surgical Biopsy for Diagnosing Primary Central Nervous Lymphoma
- New Tool Reveals Hidden Metabolic Weakness in Blood Cancers
- World's First Blood Test Distinguishes Between Benign and Cancerous Lung Nodules
- Rapid Test Uses Mobile Phone to Identify Severe Imported Malaria Within Minutes
- Gut Microbiome Signatures Predict Long-Term Outcomes in Acute Pancreatitis
- Blood Test Promises Faster Answers for Deadly Fungal Infections
- Blood Test Could Detect Infection Exposure History
- Urine-Based MRD Test Tracks Response to Bladder Cancer Surgery
Channels
Clinical Chemistry
view channel
New PSA-Based Prognostic Model Improves Prostate Cancer Risk Assessment
Prostate cancer is the second-leading cause of cancer death among American men, and about one in eight will be diagnosed in their lifetime. Screening relies on blood levels of prostate-specific antigen... Read more
Extracellular Vesicles Linked to Heart Failure Risk in CKD Patients
Chronic kidney disease (CKD) affects more than 1 in 7 Americans and is strongly associated with cardiovascular complications, which account for more than half of deaths among people with CKD.... Read moreHematology
view channel
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read more
Fast and Easy Test Could Revolutionize Blood Transfusions
Blood transfusions are a cornerstone of modern medicine, yet red blood cells can deteriorate quietly while sitting in cold storage for weeks. Although blood units have a fixed expiration date, cells from... Read more
Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
High-volume hemostasis sections must sustain rapid turnaround while managing reruns and reflex testing. Manual tube handling and preanalytical checks can strain staff time and increase opportunities for error.... Read more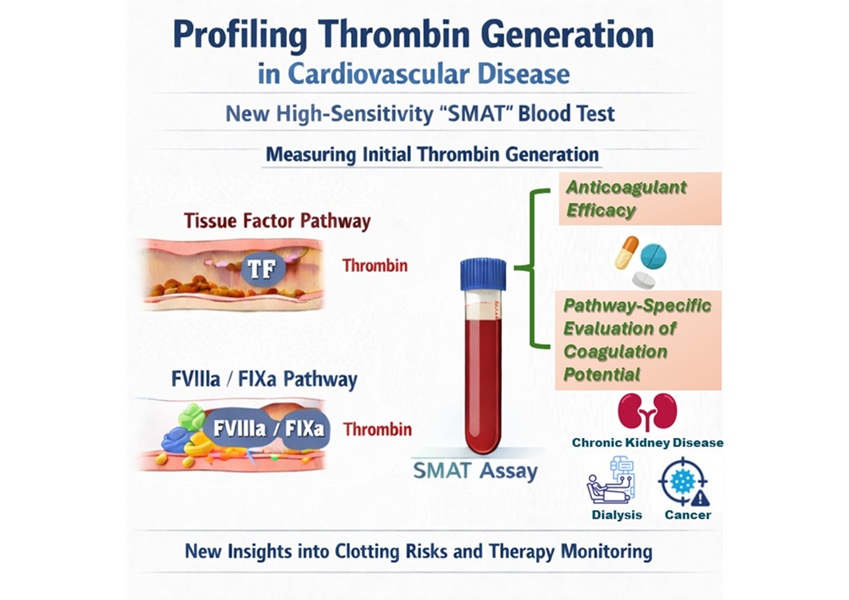
High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
Blood clotting is essential for preventing bleeding, but even small imbalances can lead to serious conditions such as thrombosis or dangerous hemorrhage. In cardiovascular disease, clinicians often struggle... Read moreImmunology
view channelBlood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
Small cell lung cancer (SCLC) is an aggressive disease with limited treatment options, and even newly approved immunotherapies do not benefit all patients. While immunotherapy can extend survival for some,... Read more
Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
Targeted cancer therapies such as PARP inhibitors can be highly effective, but only for patients whose tumors carry specific DNA repair defects. Identifying these patients accurately remains challenging,... Read more
Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
Immunotherapy has transformed cancer treatment, but only a small proportion of patients experience lasting benefit, with response rates often remaining between 10% and 20%. Clinicians currently lack reliable... Read moreMicrobiology
view channel
Comprehensive Review Identifies Gut Microbiome Signatures Associated With Alzheimer’s Disease
Alzheimer’s disease affects approximately 6.7 million people in the United States and nearly 50 million worldwide, yet early cognitive decline remains difficult to characterize. Increasing evidence suggests... Read moreAI-Powered Platform Enables Rapid Detection of Drug-Resistant C. Auris Pathogens
Infections caused by the pathogenic yeast Candida auris pose a significant threat to hospitalized patients, particularly those with weakened immune systems or those who have invasive medical devices.... Read morePathology
view channel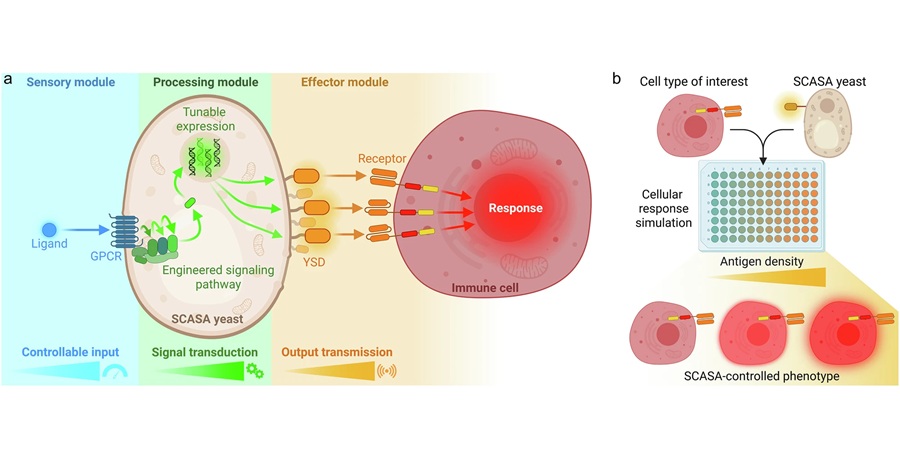
Engineered Yeast Cells Enable Rapid Testing of Cancer Immunotherapy
Developing new cancer immunotherapies is a slow, costly, and high-risk process, particularly for CAR T cell treatments that must precisely recognize cancer-specific antigens. Small differences in tumor... Read more
First-Of-Its-Kind Test Identifies Autism Risk at Birth
Autism spectrum disorder is treatable, and extensive research shows that early intervention can significantly improve cognitive, social, and behavioral outcomes. Yet in the United States, the average age... Read moreTechnology
view channel
Robotic Technology Unveiled for Automated Diagnostic Blood Draws
Routine diagnostic blood collection is a high‑volume task that can strain staffing and introduce human‑dependent variability, with downstream implications for sample quality and patient experience.... Read more
ADLM Launches First-of-Its-Kind Data Science Program for Laboratory Medicine Professionals
Clinical laboratories generate billions of test results each year, creating a treasure trove of data with the potential to support more personalized testing, improve operational efficiency, and enhance patient care.... Read moreAptamer Biosensor Technology to Transform Virus Detection
Rapid and reliable virus detection is essential for controlling outbreaks, from seasonal influenza to global pandemics such as COVID-19. Conventional diagnostic methods, including cell culture, antigen... Read more
AI Models Could Predict Pre-Eclampsia and Anemia Earlier Using Routine Blood Tests
Pre-eclampsia and anemia are major contributors to maternal and child mortality worldwide, together accounting for more than half a million deaths each year and leaving millions with long-term health complications.... Read moreIndustry
view channelNew Collaboration Brings Automated Mass Spectrometry to Routine Laboratory Testing
Mass spectrometry is a powerful analytical technique that identifies and quantifies molecules based on their mass and electrical charge. Its high selectivity, sensitivity, and accuracy make it indispensable... Read more
AI-Powered Cervical Cancer Test Set for Major Rollout in Latin America
Noul Co., a Korean company specializing in AI-based blood and cancer diagnostics, announced it will supply its intelligence (AI)-based miLab CER cervical cancer diagnostic solution to Mexico under a multi‑year... Read more
Diasorin and Fisher Scientific Enter into US Distribution Agreement for Molecular POC Platform
Diasorin (Saluggia, Italy) has entered into an exclusive distribution agreement with Fisher Scientific, part of Thermo Fisher Scientific (Waltham, MA, USA), for the LIAISON NES molecular point-of-care... Read more