Glucose-Based Test Detects Range of COVID Variants
|
By LabMedica International staff writers Posted on 05 Aug 2022 |
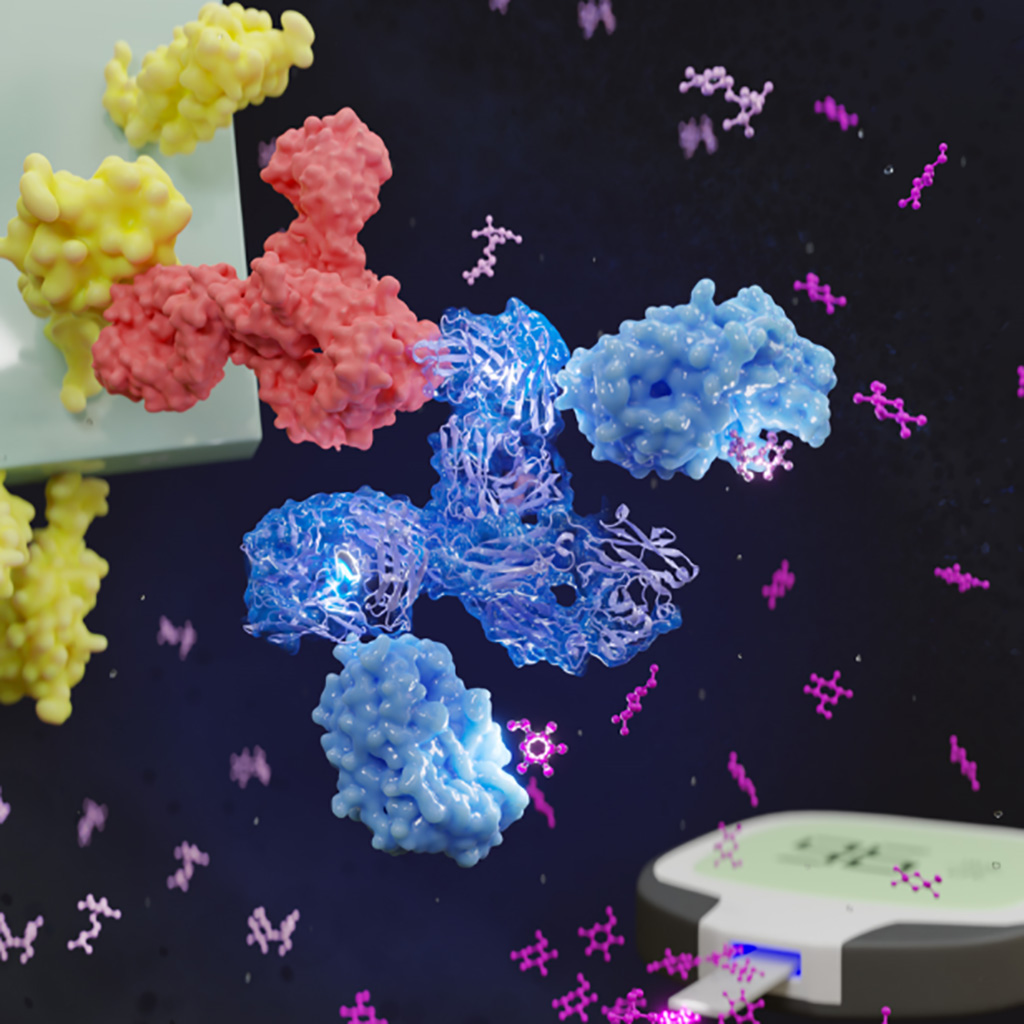
With the focus shifting from COVID-19 infections to immunity, people need to know how protected they are against the illness. Now, that information could be right at their fingertips, literally. A team of scientists has found a way to use common glucose meters, like the ones that many people with diabetes use, to measure the level of COVID-19 antibodies a person has in their blood. Antibodies are proteins created in response to a disease, and remain in the body to fight the next encounter.
Currently, to get a COVID-19 antibody test, people have to get their blood drawn at a health care facility. But scientists at Johns Hopkins University (Baltimore, MD, USA) wanted to create a test that was more affordable, accessible and easy to use that would also help people and policymakers make more informed decisions about mask-wearing, booster vaccinations and public safety measures. They chose a glucose meter as the detection device because many pharmacies across the country sell them fairly inexpensively, unlike the expensive equipment many health care facilities must use to measure antibody levels. It also gives a digital readout, making the results easy to interpret.
The scientists are trying to simplify the test for commercial use. But the process aligns closely with how people with diabetes use the glucose meter. The researchers coated the “spike” protein from SARS-CoV-2, the virus that causes COVID-19, onto a glucose monitoring test strip that they designed. The first step is to add a drop of blood to the strip. Spike protein-targeted antibodies from the blood then bind to the strip. Next, the strip is dipped into an enzyme bath, where the enzymes and antibodies bind. Then, the strip is dipped into a new solution full of the sugar sucrose, and the enzyme breaks the sucrose down into glucose. Finally, the glucose meter tests for glucose, which is proportional to the level of COVID-19 antibodies.
In the past, glucose meters have been studied as a means for measuring other chemicals besides sugar. But previous studies ran into a common problem. The challenge was to make a protein that could simultaneously measure the number of antibodies and convert the signal into glucose, so it could then be measured by the glucose meter. To do this, they merged the antibody and enzyme chemically, but the efficiency of the process was too low to be scalable to population-level screening. Instead of merging the two proteins chemically, the team realized they needed to merge them genetically into a new protein.
In a recent study, the researchers tested serum samples from at least six people who had COVID-19 and were undergoing treatment and at least six people who tested negative for the virus. The team found that the glucose-based test was on par with the gold-standard detection method used at health care facilities, pharmacies and testing sites, specifically the enzyme-linked immunosorbent assay (ELISA). The investigators tested the same samples with the glucose monitor test and the industry standard test. They observed a 95% positive and a 96% negative agreement. This means the two tests showed very similar results when testing for samples that were positive and negative for COVID-19.
The researchers have obtained a provisional patent and are reaching out to biotechnology companies to commercialize the technology. However, the researchers want to see what else the glucose-based test can do first. The test not only works for a range of COVID variants but also, potentially, for any disease that produces antibodies in the blood. All they need to do is switch the disease’s correlating protein on the test strip. The team is planning on doing additional studies to simplify the test’s process and analyze its versatility.
“We created something new, something that is not biologically existent in the world right now,” said Netz Arroyo, Ph.D., assistant professor of pharmacology and molecular sciences, one of the inventors of the new approach. “We still think we can improve the reagent and do more with it, and so a part of the process we’re undergoing right now is to see if we can make it even better. And the better we make the reagent, the more commercial interest we’ll get.”
Related Links:
Johns Hopkins University
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel
New CA19-9 Cutoff Value Helps Identify High-Risk Pancreatic Cancer Patients
Pancreatic ductal adenocarcinoma (PDAC) is frequently diagnosed at an advanced stage and remains one of the most lethal solid tumors. Clinicians commonly use serum carbohydrate antigen 19-9 (CA19-9) to... Read more
Blood-Based Biomarkers Show Promise for Psychosis Risk Prediction
Psychosis commonly emerges in adolescence or early adulthood and can severely disrupt social and occupational functioning. Hallucinations, delusions, and disorganized thinking often evolve gradually, hindering... Read moreMolecular Diagnostics
view channel
FDA Approves Expanded Liquid Biopsy Panel for Advanced Cancer Profiling
Timely, comprehensive tumor profiling helps clinicians make treatment selection decisions for patients with advanced cancer. Blood-based approaches can provide actionable insights from a simple draw and... Read more
Microbial Saliva Test Could Help Triage Esophageal Cancer Risk
Esophageal squamous cell carcinoma (ESCC) is highly lethal, partly because many patients are diagnosed only after swallowing becomes difficult and treatment options are largely palliative.... Read more
Expanded DPYD Genotyping Test Supports Safer Chemotherapy Dosing
Fluoropyrimidines such as 5-fluorouracil (5-FU) are chemotherapy drugs prescribed to more than two million cancer patients each year, but 10–20% of patients can experience severe, and sometimes fatal,... Read more
Multi-Omics Profiling Helps Predict BCG Response and Recurrence in Bladder Cancer
High-risk non–muscle-invasive bladder cancer frequently recurs after therapy, with about 30% of patients relapsing and roughly 10% dying within two years despite tumor resection, surveillance, and Bacillus... Read moreHematology
view channel
Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
Iron deficiency in school-age children can affect brain development, learning, growth, and physical performance, yet early deficiency may be missed when screening focuses mainly on anemia.... Read more
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read moreImmunology
view channel
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read more
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read moreMicrobiology
view channel
New AMR Assay Supports Rapid Infection Control Screening in Hospitals
As antimicrobial resistance spreads worldwide, healthcare-associated infections are placing a growing burden on hospitals, increasing the need for faster and broader diagnostic solutions.... Read more
Diagnostic Gaps Complicate Bundibugyo Ebola Outbreak Response in Congo
In eastern Democratic Republic of the Congo, communities are confronting a resurgence of Bundibugyo ebolavirus, a rarer species for which no vaccines or treatments have been approved. Ebola is a highly... Read more
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read morePathology
view channel
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read more
AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
Non–muscle invasive bladder cancer has highly variable outcomes, complicating surveillance and treatment planning. Risk assessment typically relies on stage, grade, and tumor size, leaving uncertainty... Read moreTechnology
view channel
AI-Enabled Assistant Unifies Molecular Workflow Planning and Support
Clinical laboratories and research groups face increasingly complex molecular workflows and expanding technical documentation spread across multiple systems. Fragmented digital tools can slow experiment... Read more
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read moreIndustry
view channel
Strategic Collaboration Advances RNA Foundation Models for Precision Oncology
Bulk RNA sequencing is increasingly used to study tumor biology, but standard analyses often reduce results to gene-level summaries that miss important transcript variants and mutation patterns.... Read more
























