Meridian Acquires Euprotein to Further Expand Recombinant Protein Capabilities
|
By LabMedica International staff writers Posted on 03 May 2022 |
Meridian Bioscience, Inc. (Cincinnati, OH, USA) has acquired, through its subsidiary Meridian Life Science, Inc., substantially all of the assets of EUPROTEIN Inc. (North Brunswick Township, NJ, USA).
EUPROTEIN offers custom development and production of high-quality bioresearch reagents, with a particular focus on human and other mammalian proteins and recombinant monoclonal antibodies. The acquisition will help Meridian accelerate its pipeline of new immunological reagents while expanding recombinant capabilities.
"We are excited to welcome EUPROTEIN into the Meridian family," said Jack Kenny, Meridian Bioscience Chief Executive Officer. "We already extensively work with EUPROTEIN on projects with our Life Science R&D team, and they are well-positioned to help us accelerate our pipeline of new immunological reagents."
"The EUPROTEIN team is delighted to join Meridian," added Guangli Wang, Ph.D., Founder, and CEO of EUPROTEIN. "Combining our products and capabilities with Meridian's global reach and stellar reputation for providing the highest quality reagents furthers our vision of making a lasting impact on global healthcare."
Related Links:
Meridian Bioscience, Inc.
EUPROTEIN Inc.
Latest Industry News
- Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
- Integrated DNA Technologies Expands into Clinical Diagnostics
- Co-Diagnostics Agreement Expands Commercial and Distribution Reach in South Asia
- Automated MSI Test Gains IVDR Certification to Guide CRC Therapy
- New Partnership Brings Alzheimer’s Blood Biomarker Test to Community Screening Network
- MGI Tech Strengthens Sequencing Portfolio with Dual Acquisition
- Agilent Technologies Acquires Pathology Diagnostics Company Biocare Medical
- Cepheid Joins CDC Initiative to Strengthen U.S. Pandemic Testing Preparednesss
- QuidelOrtho Collaborates with Lifotronic to Expand Global Immunoassay Portfolio
- WHX Labs in Dubai spotlights leadership skills shaping next-generation laboratories
- AI-Powered Cervical Cancer Test Set for Major Rollout in Latin America
- New Collaboration Brings Automated Mass Spectrometry to Routine Laboratory Testing
- Diasorin and Fisher Scientific Enter into US Distribution Agreement for Molecular POC Platform
- WHX Labs Dubai to Gather Global Experts in Antimicrobial Resistance at Inaugural AMR Leaders’ Summit
- BD and Penn Institute Collaborate to Advance Immunotherapy through Flow Cytometry
- Abbott Acquires Cancer-Screening Company Exact Sciences
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreMolecular Diagnostics
view channel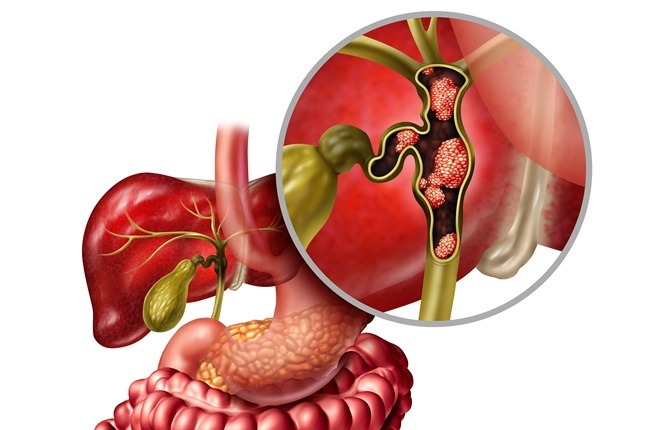
New Molecular Test Boosts Accuracy of Bile Duct Cancer Diagnosis
Bile duct strictures can arise from cancer or benign disease, but their location within ducts connecting the liver, gallbladder, and intestines complicates evaluation. Standard biopsy and cytology may... Read moreAdaptive PCR Platform Improves Consistency in Small-Batch NGS Workflows
PCR amplification during next-generation sequencing (NGS) library preparation can introduce variability, often requiring manual quantification and risking over-cycling artifacts. The issue is especially... Read more
First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
Accurate identification of clonal immunoglobulin heavy chain (IGH) gene rearrangements is central to evaluating suspected B-cell lymphoproliferative disorders, where a single B-cell clone yields a defining... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel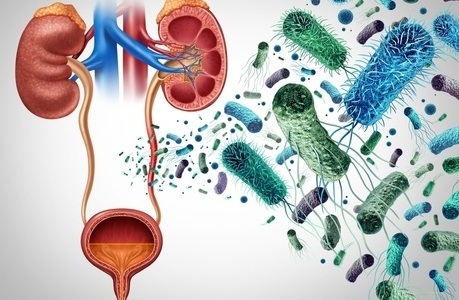
Rapid Urine Test Speeds Antibiotic Selection for UTIs
Urinary tract infections are a common reason for antibiotic prescribing and have led to more than 800,000 hospital admissions across England in the past five years, according to National Health Service data.... Read more
WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Tuberculosis (TB) remains a leading infectious killer, with more than 3,300 deaths and 29,000 new illnesses every day. Diagnostic delays and dependence on centralized laboratory networks continue to impede... Read morePathology
view channel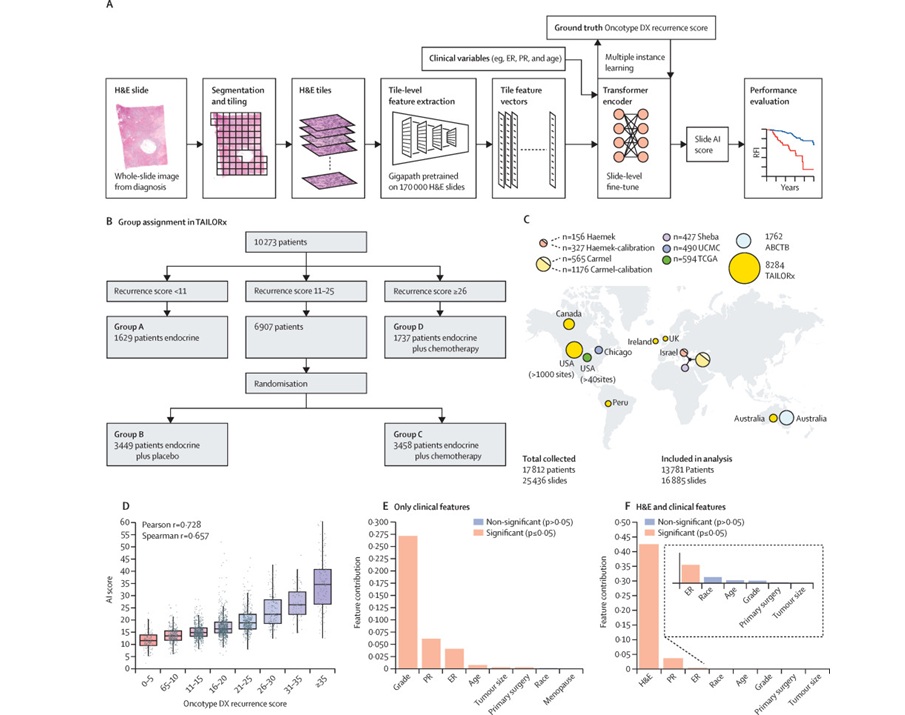
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read more
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more