LumiraDx Launches Rapid Onsite COVID-19 Surveillance Test for Simultaneous Testing of Five Samples on Portable Instrument
|
By LabMedica International staff writers Posted on 03 Sep 2021 |

Image: The LumiraDx SARS-CoV-2 Ag Surveillance Test allows simultaneous testing of up to five samples on a small, portable instrument (Photo courtesy of LumiraDx)
LumiraDx (London, UK) has announced the US launch of its COVID-19 Surveillance Test, for environments such as schools and workplaces.
The LumiraDx SARS-CoV-2 Ag Surveillance Test allows simultaneous testing of up to five samples on a small, portable instrument with cost per sample as low as USD 4. With digital results in less than 12 minutes from sample application and a comprehensive connectivity solution, it is designed to measure population health metrics in real-time to help prevent the spread of infection using an affordable, easy-to-use, onsite testing platform. LumiraDx's announcement comes at a time when schools across the US are assessing how to both bring students back to in-person learning while also monitoring for outbreaks to ensure they can stay safe and open.
The LumiraDx SARS-CoV-2 Ag Surveillance Test, used on the LumiraDx Platform, has demonstrated 100.0% positive agreement and 96.6% negative agreement with its comparator, the LumiraDx SARS-CoV-2 Ag Test. It allows rapid, high-sensitivity surveillance testing in both CLIA and non-CLIA certified settings. The company also offers the LumiraDx SARS-CoV-2 Ag Test and SARS-CoV-2 Ab Test for clinical use in CLIA waived settings on the LumiraDx Platform, both of which were granted Emergency Use Authorization by the FDA.
“Our rapid onsite COVID-19 Surveillance Test offers options that can fit into a variety of different use-cases to support a safe return to school or the workplace and instill confidence in groups about their environment,” said Pooja Pathak, LumiraDx Chief Product Officer. “By pooling up to five samples at one time, the LumiraDx SARS-CoV-2 Ag Surveillance test provides an efficient, accurate and cost-effective way to implement a surveillance testing program.”
Related Links:
LumiraDx
The LumiraDx SARS-CoV-2 Ag Surveillance Test allows simultaneous testing of up to five samples on a small, portable instrument with cost per sample as low as USD 4. With digital results in less than 12 minutes from sample application and a comprehensive connectivity solution, it is designed to measure population health metrics in real-time to help prevent the spread of infection using an affordable, easy-to-use, onsite testing platform. LumiraDx's announcement comes at a time when schools across the US are assessing how to both bring students back to in-person learning while also monitoring for outbreaks to ensure they can stay safe and open.
The LumiraDx SARS-CoV-2 Ag Surveillance Test, used on the LumiraDx Platform, has demonstrated 100.0% positive agreement and 96.6% negative agreement with its comparator, the LumiraDx SARS-CoV-2 Ag Test. It allows rapid, high-sensitivity surveillance testing in both CLIA and non-CLIA certified settings. The company also offers the LumiraDx SARS-CoV-2 Ag Test and SARS-CoV-2 Ab Test for clinical use in CLIA waived settings on the LumiraDx Platform, both of which were granted Emergency Use Authorization by the FDA.
“Our rapid onsite COVID-19 Surveillance Test offers options that can fit into a variety of different use-cases to support a safe return to school or the workplace and instill confidence in groups about their environment,” said Pooja Pathak, LumiraDx Chief Product Officer. “By pooling up to five samples at one time, the LumiraDx SARS-CoV-2 Ag Surveillance test provides an efficient, accurate and cost-effective way to implement a surveillance testing program.”
Related Links:
LumiraDx
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel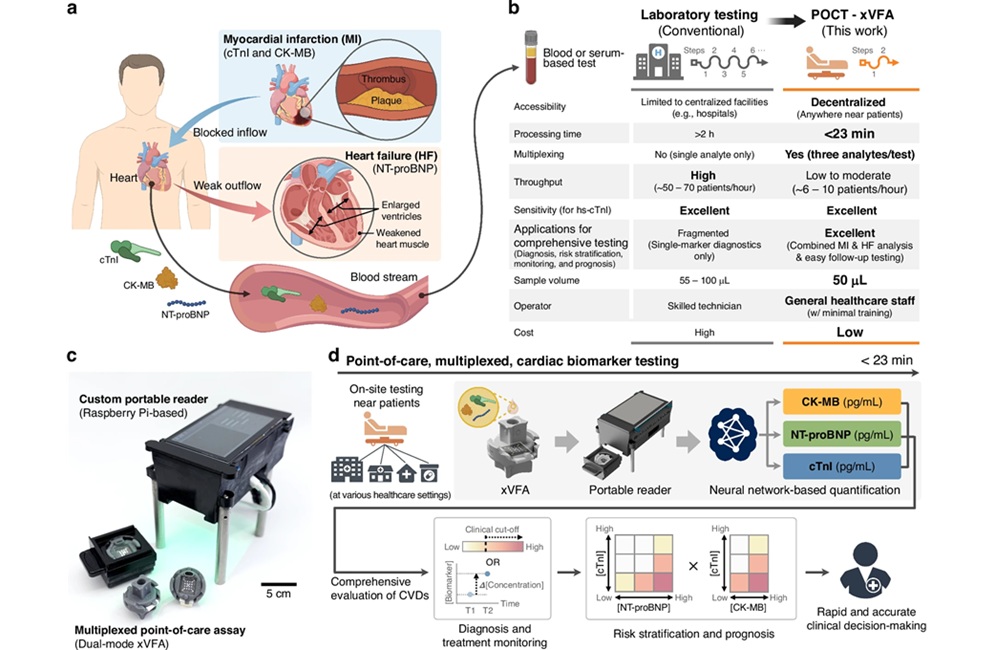
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel
Genome Sequencing Identifies Noncoding Variants Causing Neonatal Diabetes
Neonatal diabetes is a rare form of diabetes that presents in the first six months of life and is driven by genetic changes, yet many affected families still lack definitive diagnoses. Genetic studies... Read more
Genetic Markers Predict GLP-1 Weight-Loss Response and Side Effects
Glucagon-like peptide-1 (GLP-1) receptor agonists such as semaglutide and tirzepatide are now widely used for weight management, yet individual responses vary considerably, with some patients experiencing... Read moreHematology
view channel
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read more
New Platelet Function Assay Enables Monitoring of Antiplatelet Therapy
Monitoring response to antiplatelet therapy remains challenging for many clinical laboratories. Aggregation-based assays and cartridge systems often require specialized personnel, dedicated instruments,... Read moreImmunology
view channelCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read more
Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
Active tuberculosis (TB) remains a leading cause of death and illness worldwide, yet distinguishing contagious disease from latent infection continues to challenge clinicians. Standard screening tools... Read more
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel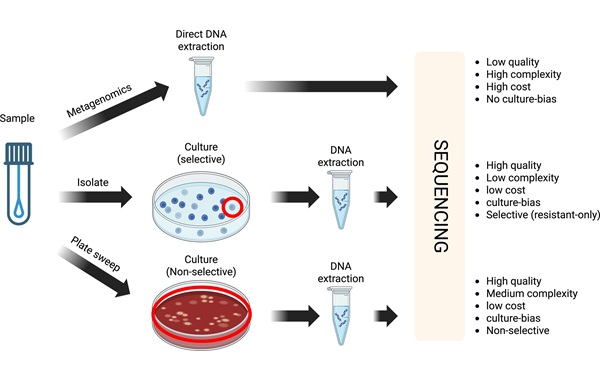
Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
Intensive care units face persistent threats from hospital-acquired infections, increasingly driven by drug-resistant bacteria. Rapidly pinpointing environmental reservoirs and transmission hotspots remains... Read more
New Bacterial Target Identified for Early Detection of Noma
Noma is a rapidly progressing orofacial infection that begins as gingivitis and can destroy oral and facial tissues, primarily affecting young children living in extreme poverty. Without treatment, it... Read morePathology
view channelAI Improves Completeness of Complex Cancer Pathology Reports
Oncology teams increasingly rely on pathology reports that integrate histopathology, immunohistochemistry, and rapidly expanding biomarker testing. As patients live longer and undergo repeated analyses... Read more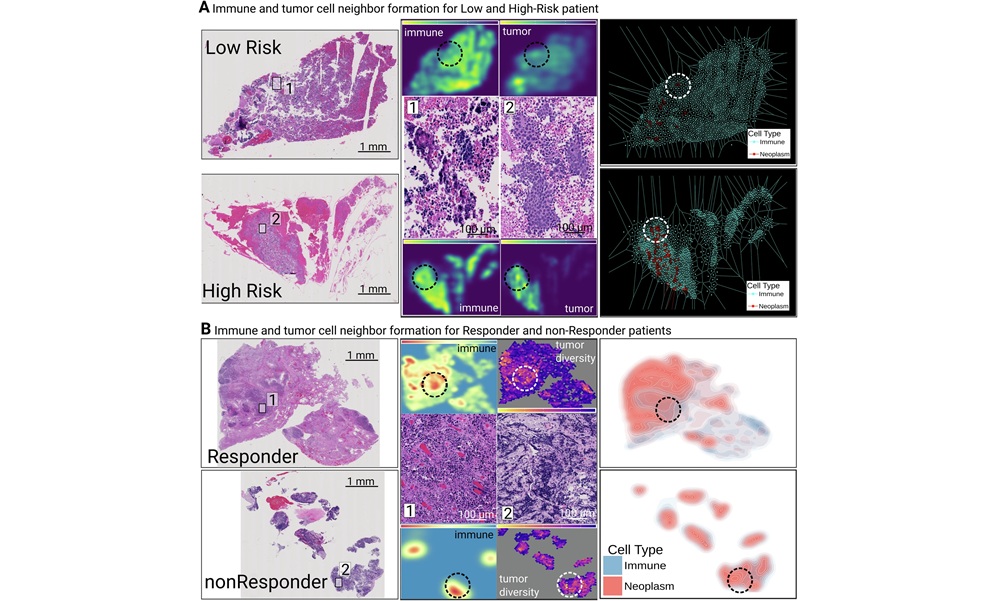
AI Tool Predicts Chemotherapy Response in Small Cell Lung Cancer
Small cell lung cancer often presents at an extensive stage and progresses rapidly, leaving little time to tailor first-line therapy. Clinicians currently lack biomarkers to guide which patients will benefit... Read more_image.png)
Tumor-Specific Biomarker Predicts Neoadjuvant Immunotherapy Response in Gastric Cancer
Gastric cancer is the fifth most common malignancy and the fourth leading cause of cancer mortality worldwide, with China bearing nearly half of the global burden. Only a subset of patients benefit from... Read moreTechnology
view channel
Noninvasive Sputum Test Detects Early Lung Cancer
Early detection remains critical for improving outcomes in lung cancer, yet clinicians increasingly encounter indeterminate pulmonary nodules found incidentally or through screening, complicating decision-making.... Read more
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel