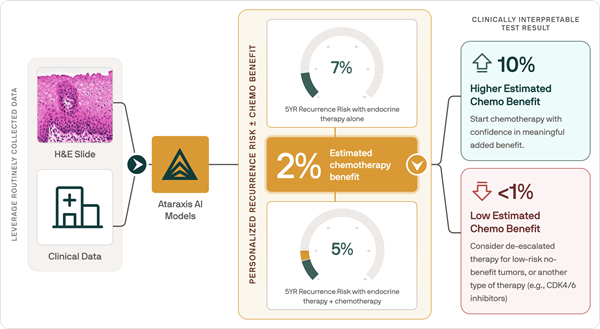
Virus-Like Probes Could Help Make Rapid COVID-19 Testing More Accurate and Reliable
|
By LabMedica International staff writers Posted on 02 Dec 2020 |
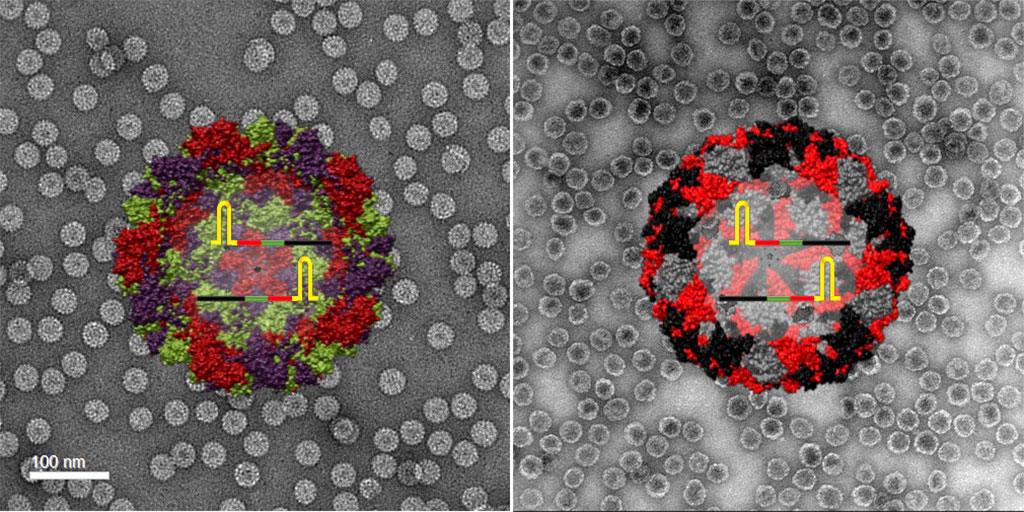
Image: Illustration and TEM image of SARS-CoV-2 positive control made from plant virus-based nanoparticles (left) and bacteriophage nanoparticles (right) (Photo courtesy of Soo Khim Chan/ACS Nano)
A team of nanoengineers has developed new and improved probes, known as positive controls, that could make it easier to validate rapid, point-of-care diagnostic tests for COVID-19 across the globe.
The positive controls, made from virus-like particles by nanoengineers at the University of California San Diego (La Jolla, CA, USA), are stable and easy to manufacture. Researchers say the controls have the potential to improve the accuracy of new COVID-19 tests that are simpler, faster and cheaper, making it possible to expand testing outside the lab.
Positive controls are a staple in the lab - they are used to verify that a test or experiment indeed works. The positive controls that are primarily used to validate today’s COVID-19 tests are naked synthetic RNAs, plasmids or RNA samples from infected patients. But the issue is RNA and plasmids are not stable like viral particles. They can degrade easily and require refrigeration, making them inconvenient and costly to ship around the world or store for long periods of time.
By packaging segments of RNA from the SARS-CoV-2 virus into virus-like particles, the researchers have shown that they can create positive controls for COVID-19 tests that are stable - they can be stored for a week at temperatures up to 40 C (104 F), and retain 70% of their activity even after one month of storage - and can pass detection as the novel coronavirus without being infectious. The team developed two different controls: one made from plant virus nanoparticles, the other from bacteriophage nanoparticles. The controls are run and analyzed right alongside a patient sample, providing a reliable benchmark for what a positive test result should look like.
To make the plant virus-based controls, the researchers use the cowpea chlorotic mottle virus, which infects black-eyed pea plants. They essentially open the virus, remove its RNA contents, replace them with a synthesized RNA template containing specific sequences from the SARS-CoV-2 virus, then close everything back up. The process to make the bacteriophage-based controls starts with plasmids, which are rings of DNA. Inserted into these plasmids are the gene sequences of interest from the SARS-CoV-2 virus, as well as genes coding for surface proteins of the bacteriophage Qbeta. These plasmids are then taken up by bacteria. This process reprograms the bacteria to produce virus-like particles with SARS-CoV-2 RNA sequences on the inside and Qbeta bacteriophage proteins on the outside.
Both controls were validated with clinical samples. A big advantage, the researchers point out, is that unlike the positive controls used today, these can be used in all steps of a COVID-19 test. So far, the researchers have adapted their controls for use in the CDC-authorized RT-PCR test. While this is currently the gold standard for COVID-19 testing, it is expensive, complex, and can take days to return results due to the logistics of sending samples off to a lab with PCR capability. The researchers are now working on adapting the controls for use in less complex diagnostic tests like the RT-LAMP test that can be done on the spot, out of the lab and provide results right away.
“Our goal is to make an impact not necessarily in the hospital, where you have state-of-the-art facilities, but in low-resource, underserved areas that may not have the sophisticated infrastructure or trained personnel,” said Nicole Steinmetz, a professor of nanoengineering at the UC San Diego Jacobs School of Engineering. “It’s a relatively simple nanotechnology approach to make low-tech assays more accurate. This could help break down some of the barriers to mass testing of underserved populations in the U.S. and across the world.”
Related Links:
University of California San Diego
The positive controls, made from virus-like particles by nanoengineers at the University of California San Diego (La Jolla, CA, USA), are stable and easy to manufacture. Researchers say the controls have the potential to improve the accuracy of new COVID-19 tests that are simpler, faster and cheaper, making it possible to expand testing outside the lab.
Positive controls are a staple in the lab - they are used to verify that a test or experiment indeed works. The positive controls that are primarily used to validate today’s COVID-19 tests are naked synthetic RNAs, plasmids or RNA samples from infected patients. But the issue is RNA and plasmids are not stable like viral particles. They can degrade easily and require refrigeration, making them inconvenient and costly to ship around the world or store for long periods of time.
By packaging segments of RNA from the SARS-CoV-2 virus into virus-like particles, the researchers have shown that they can create positive controls for COVID-19 tests that are stable - they can be stored for a week at temperatures up to 40 C (104 F), and retain 70% of their activity even after one month of storage - and can pass detection as the novel coronavirus without being infectious. The team developed two different controls: one made from plant virus nanoparticles, the other from bacteriophage nanoparticles. The controls are run and analyzed right alongside a patient sample, providing a reliable benchmark for what a positive test result should look like.
To make the plant virus-based controls, the researchers use the cowpea chlorotic mottle virus, which infects black-eyed pea plants. They essentially open the virus, remove its RNA contents, replace them with a synthesized RNA template containing specific sequences from the SARS-CoV-2 virus, then close everything back up. The process to make the bacteriophage-based controls starts with plasmids, which are rings of DNA. Inserted into these plasmids are the gene sequences of interest from the SARS-CoV-2 virus, as well as genes coding for surface proteins of the bacteriophage Qbeta. These plasmids are then taken up by bacteria. This process reprograms the bacteria to produce virus-like particles with SARS-CoV-2 RNA sequences on the inside and Qbeta bacteriophage proteins on the outside.
Both controls were validated with clinical samples. A big advantage, the researchers point out, is that unlike the positive controls used today, these can be used in all steps of a COVID-19 test. So far, the researchers have adapted their controls for use in the CDC-authorized RT-PCR test. While this is currently the gold standard for COVID-19 testing, it is expensive, complex, and can take days to return results due to the logistics of sending samples off to a lab with PCR capability. The researchers are now working on adapting the controls for use in less complex diagnostic tests like the RT-LAMP test that can be done on the spot, out of the lab and provide results right away.
“Our goal is to make an impact not necessarily in the hospital, where you have state-of-the-art facilities, but in low-resource, underserved areas that may not have the sophisticated infrastructure or trained personnel,” said Nicole Steinmetz, a professor of nanoengineering at the UC San Diego Jacobs School of Engineering. “It’s a relatively simple nanotechnology approach to make low-tech assays more accurate. This could help break down some of the barriers to mass testing of underserved populations in the U.S. and across the world.”
Related Links:
University of California San Diego
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channelNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read more
Blood Metabolite Test Detects Early Cognitive Decline
Timely identification of individuals at risk of dementia remains difficult because symptoms commonly appear only after significant neurodegeneration. Accessible screening tools that flag subtle cognitive... Read moreMolecular Diagnostics
view channel
cfDNA Methylation Assay Enables Multi-Disease Detection from Single Blood Sample
Early, accurate detection of cancer and organ disease remains limited by cost, reliance on targeted mutation assays, and uncertainty about the signal’s tissue of origin. Many liquid biopsy approaches require... Read more
Rapid Point-of-Care RT-PCR Test Differentiates Influenza A/B and SARS-CoV-2 in Minutes
Respiratory viruses such as influenza A/B and SARS‑CoV‑2 continue to burden urgent care and emergency settings, where rapid, reliable differentiation guides therapy and infection control.... Read moreHematology
view channel
AI-Powered Digital Workflow Standardizes Bone Marrow Aspirate Morphology
Bone marrow aspirate examination is central to diagnosing and monitoring blood cancers and other serious hematologic diseases, yet the process in many laboratories remains manual and highly dependent on... Read more
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read moreImmunology
view channel
Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
Active tuberculosis (TB) remains a leading cause of death and illness worldwide, yet distinguishing contagious disease from latent infection continues to challenge clinicians. Standard screening tools... Read more
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
New Bacterial Target Identified for Early Detection of Noma
Noma is a rapidly progressing orofacial infection that begins as gingivitis and can destroy oral and facial tissues, primarily affecting young children living in extreme poverty. Without treatment, it... Read more
Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
Streptococcus dysgalactiae subspecies equisimilis (SDSE) infections are increasing worldwide and include variants that may lead to severe disease. Researchers now report that whole-genome sequencing of... Read morePathology
view channel
AI Pathology Tool Predicts Chemotherapy Response in Small Cell Lung Cancer
Small cell lung cancer often presents at an extensive stage and progresses rapidly, leaving little time to tailor first-line therapy. Clinicians currently lack biomarkers to guide which patients will benefit... Read more_image.png)
Tumor-Specific Biomarker Predicts Neoadjuvant Immunotherapy Response in Gastric Cancer
Gastric cancer is the fifth most common malignancy and the fourth leading cause of cancer mortality worldwide, with China bearing nearly half of the global burden. Only a subset of patients benefit from... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channelGlobal Partnership Aims to Streamline NGS Tumor Profiling in Oncology Trials
CellCarta and Pillar Biosciences announced a global, multi-year strategic partnership on April 2, 2026 to broaden access to operationally streamlined next-generation sequencing (NGS) tumor profiling for... Read more