New Tool Developed for Diagnosis of Chronic HBV Infection
|
By LabMedica International staff writers Posted on 23 Oct 2019 |
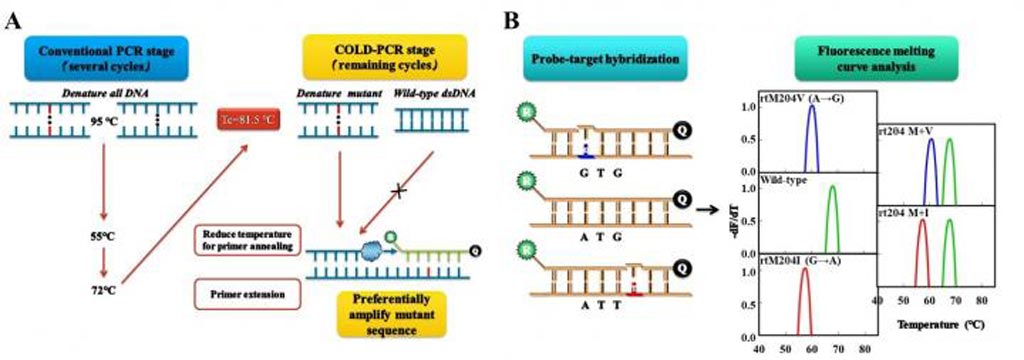
Image: Diagram workflow for co-amplification at lower denaturation temperature PCR (COLD-PCR)/fluorescence melting curve analysis (FMCA) (Photo courtesy of The First Affiliated Hospital of Fujian Medical University).
A team of Chinese researchers has described the development of a new and powerful tool for the diagnosis and treatment of chronic hepatitis B virus (HBV) infection.
Methods currently used to diagnose and monitor chronic hepatitis B (CHB) infection are for the most part based on dynamic and real-time HBV DNA, genotype, and reverse transcriptase (RT) mutation analysis. However, the methods used to perform these analyses are limited by poor sensitivity or inability to detect more than one mutation at a time. Other methods are too cumbersome or expensive for clinical use.
Investigators at Fujian Medical University (Peoples Republic of China) have improved this situation by establishing a highly sensitive co-amplification at lower denaturation temperature PCR (COLD-PCR) coupled with probe-based fluorescence melting curve analysis (FMCA) for precision diagnosis of CHB patients.
COLD-PCR (co-amplification at lower denaturation temperature-PCR) is a modified Polymerase Chain Reaction (PCR) protocol that enriches variant alleles from a mixture of wildtype and mutation-containing DNA. The ability to preferentially amplify and identify minority alleles and low-level somatic DNA mutations in the presence of excess wildtype alleles is useful for the detection of mutations.
Melting curve analysis is an assessment of the dissociation characteristics of double-stranded DNA during heating. As the temperature is raised, the double strand begins to dissociate leading to a rise in the absorbance intensity, hyperchromicity. The temperature at which 50% of DNA is denatured is known as the melting temperature. The information gathered can be used to infer the presence and identity of single-nucleotide polymorphism (SNP) mutations.
The COLD-PCR/FMCA method was shown to be able to detect HBV DNA, genotypes, and RT mutations, simultaneously. The analytical performance of this method, including imprecision, accuracy, sensitivity, detection limits, linear range, and its ability to detect minor variants was systematically evaluated.
Results revealed that the COLD-PCR/FMCA method could detect HBV mutations at much lower concentrations than other techniques such as PCR/FMCA or PCR Sanger sequencing (1% vs. 10% vs. 20%, respectively). The new technique could also distinguish different phases of HBV infection according to the proportion and type of mutations as well as by detecting HBV DNA.
"Guidelines have confirmed that dynamic monitoring of HBV DNA, genotypes, and reverse transcriptase (RT) mutant DNA is of great importance to assess infection status, predict disease progression, and judge treatment efficacy in HBV-infected patients," senior author Dr. Qishui Ou, a researcher in laboratory medicine at The First Afilliated Hospital of Fujian Medical University. "We believe COLD-PCR/FMCA provides a powerful laboratory tool for precise diagnosis and treatment of HBV-infected patients."
"Our goal was to establish a more practical and inexpensive method with high sensitivity to detect genotype and RT mutations while detecting HBV DNA," said Dr. Ou. "Until now there have not been high-throughput approaches to detect HBV DNA, genotype, and RT mutations simultaneously. Therefore, it is necessary to establish a more practical and inexpensive method with high sensitivity to detect genotype and RT mutations while detecting HBV DNA. COLD-PCR/FMCA has that potential."
The study was published in the October 10, 2019, online edition of the Journal of Molecular Diagnostics.
Related Links:
Fujian Medical University
Methods currently used to diagnose and monitor chronic hepatitis B (CHB) infection are for the most part based on dynamic and real-time HBV DNA, genotype, and reverse transcriptase (RT) mutation analysis. However, the methods used to perform these analyses are limited by poor sensitivity or inability to detect more than one mutation at a time. Other methods are too cumbersome or expensive for clinical use.
Investigators at Fujian Medical University (Peoples Republic of China) have improved this situation by establishing a highly sensitive co-amplification at lower denaturation temperature PCR (COLD-PCR) coupled with probe-based fluorescence melting curve analysis (FMCA) for precision diagnosis of CHB patients.
COLD-PCR (co-amplification at lower denaturation temperature-PCR) is a modified Polymerase Chain Reaction (PCR) protocol that enriches variant alleles from a mixture of wildtype and mutation-containing DNA. The ability to preferentially amplify and identify minority alleles and low-level somatic DNA mutations in the presence of excess wildtype alleles is useful for the detection of mutations.
Melting curve analysis is an assessment of the dissociation characteristics of double-stranded DNA during heating. As the temperature is raised, the double strand begins to dissociate leading to a rise in the absorbance intensity, hyperchromicity. The temperature at which 50% of DNA is denatured is known as the melting temperature. The information gathered can be used to infer the presence and identity of single-nucleotide polymorphism (SNP) mutations.
The COLD-PCR/FMCA method was shown to be able to detect HBV DNA, genotypes, and RT mutations, simultaneously. The analytical performance of this method, including imprecision, accuracy, sensitivity, detection limits, linear range, and its ability to detect minor variants was systematically evaluated.
Results revealed that the COLD-PCR/FMCA method could detect HBV mutations at much lower concentrations than other techniques such as PCR/FMCA or PCR Sanger sequencing (1% vs. 10% vs. 20%, respectively). The new technique could also distinguish different phases of HBV infection according to the proportion and type of mutations as well as by detecting HBV DNA.
"Guidelines have confirmed that dynamic monitoring of HBV DNA, genotypes, and reverse transcriptase (RT) mutant DNA is of great importance to assess infection status, predict disease progression, and judge treatment efficacy in HBV-infected patients," senior author Dr. Qishui Ou, a researcher in laboratory medicine at The First Afilliated Hospital of Fujian Medical University. "We believe COLD-PCR/FMCA provides a powerful laboratory tool for precise diagnosis and treatment of HBV-infected patients."
"Our goal was to establish a more practical and inexpensive method with high sensitivity to detect genotype and RT mutations while detecting HBV DNA," said Dr. Ou. "Until now there have not been high-throughput approaches to detect HBV DNA, genotype, and RT mutations simultaneously. Therefore, it is necessary to establish a more practical and inexpensive method with high sensitivity to detect genotype and RT mutations while detecting HBV DNA. COLD-PCR/FMCA has that potential."
The study was published in the October 10, 2019, online edition of the Journal of Molecular Diagnostics.
Related Links:
Fujian Medical University
Latest BioResearch News
- Genetic Testing Identifies Greater Inherited Sudden Cardiac Arrest Risk in Younger Individuals
- Hidden 'Jumping Gene' Variant Linked to Higher Pancreatic Cancer Risk
- Common White Blood Cells Produce Schizophrenia-Linked Protein
- Nanopore Method Captures RNA Folding at Single-Molecule Resolution
- Tumor Microenvironment Marker Linked to Worse Survival in Solid Tumors
- Hidden Immune Gene Defect May Explain Kaposi Sarcoma Susceptibility
- Genetic Markers May Help Predict Amputation Risk in Peripheral Artery Disease
- Gene Signature Shows Promise for Depression Biomarker Testing
- AI-Driven Tumor Profiling Initiative Targets Precision Therapy Development
- Researchers Map Protein and Glycosylation Across 15 Human Body Fluids
- Telomere Length Abnormalities Linked to Lymphoma Development
- Biomarker Signals Chemotherapy Resistance in Relapsed Small Cell Lung Cancer
- Inflammatory Gene Signature Links Metabolic Disease to Pancreatic Cancer Recurrence
- Study Links Abnormal Gene Splicing to Treatment Response in Metastatic Kidney Cancer
- Research Reveals How Some Aplastic Anemia Patients Recover Bone Marrow Function
- New Molecular Insights Support Diagnosis of Hodgkin Lymphoma
Channels
Clinical Chemistry
view channel
New CA19-9 Cutoff Value Helps Identify High-Risk Pancreatic Cancer Patients
Pancreatic ductal adenocarcinoma (PDAC) is frequently diagnosed at an advanced stage and remains one of the most lethal solid tumors. Clinicians commonly use serum carbohydrate antigen 19-9 (CA19-9) to... Read more
Blood-Based Biomarkers Show Promise for Psychosis Risk Prediction
Psychosis commonly emerges in adolescence or early adulthood and can severely disrupt social and occupational functioning. Hallucinations, delusions, and disorganized thinking often evolve gradually, hindering... Read moreMolecular Diagnostics
view channel
New RNA Origami Method Supports Faster Targeted Testing for Repeat Expansion Disorders
Repeat expansion disorders drive conditions such as myotonic dystrophy, Huntington’s disease, and amyotrophic lateral sclerosis (ALS), yet accurately sizing the mutated sequences remains difficult.... Read more
FDA Approves Expanded Liquid Biopsy Panel for Advanced Cancer Profiling
Timely, comprehensive tumor profiling helps clinicians make treatment selection decisions for patients with advanced cancer. Blood-based approaches can provide actionable insights from a simple draw and... Read moreHematology
view channel
Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
Iron deficiency in school-age children can affect brain development, learning, growth, and physical performance, yet early deficiency may be missed when screening focuses mainly on anemia.... Read more
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read moreImmunology
view channel
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read more
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read moreMicrobiology
view channel
New AMR Assay Supports Rapid Infection Control Screening in Hospitals
As antimicrobial resistance spreads worldwide, healthcare-associated infections are placing a growing burden on hospitals, increasing the need for faster and broader diagnostic solutions.... Read more
Diagnostic Gaps Complicate Bundibugyo Ebola Outbreak Response in Congo
In eastern Democratic Republic of the Congo, communities are confronting a resurgence of Bundibugyo ebolavirus, a rarer species for which no vaccines or treatments have been approved. Ebola is a highly... Read more
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read morePathology
view channel
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read more
AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
Non–muscle invasive bladder cancer has highly variable outcomes, complicating surveillance and treatment planning. Risk assessment typically relies on stage, grade, and tumor size, leaving uncertainty... Read moreTechnology
view channel
AI-Enabled Assistant Unifies Molecular Workflow Planning and Support
Clinical laboratories and research groups face increasingly complex molecular workflows and expanding technical documentation spread across multiple systems. Fragmented digital tools can slow experiment... Read more
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read moreIndustry
view channel
Strategic Collaboration Advances RNA Foundation Models for Precision Oncology
Bulk RNA sequencing is increasingly used to study tumor biology, but standard analyses often reduce results to gene-level summaries that miss important transcript variants and mutation patterns.... Read more

























