Researchers Deform Cells to Deliver RNA, Proteins, and Nanoparticles for Many Applications
|
By LabMedica International staff writers Posted on 06 Feb 2013 |
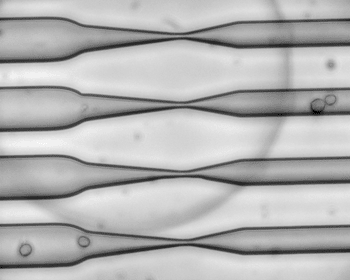
Image: As cells squeeze through a narrow channel, tiny holes open in their membranes, allowing large molecules such as RNA to pass through (Photo courtesy of Armon Sharei and Emily Jackson).
Researchers have found a safe and effective way to push large molecules through the cell membrane by jamming the cells through a narrow constriction that opens up very small, temporary holes in the membrane. Any large molecules drifting outside the cell—such as proteins, RNA, or nanoparticles—can slide through the membrane during this disruption.
Living cells are enclosed by a membrane that closely controls what gets in and out of the cell. This barrier is necessary for cells to control their internal environment, but it makes it more difficult for scientists to deliver large molecules such as nanoparticles for imaging, or proteins that can reprogram them into pluripotent stem cells.
Using this technique, the Massachusetts Institute of Technology (MIT; Cambridge, MA, USA: www.mit.edu) researchers were able to deliver reprogramming proteins and create induced pluripotent stem cells with a success rate 10 to 100 times superior than any existing application. They also used it to deliver nanoparticles, including quantum dots and carbon nanotubes, which can be used to image cells and track what is occurring inside them.
“It’s very useful to be able to get large molecules into cells. We thought it might be interesting if you could have a relatively simple system that could deliver many different compounds,” said Dr. Klavs Jensen, a professor of chemical engineering, professor of materials science and engineering, and a senior author of a paper describing the new device in this week’s issue of the Proceedings of the National Academy of Sciences of the United States of America (PNAS).
Scientists had earlier developed several approaches to get large molecules into cells, but all of them have downsides. DNA or RNA can be parceled into viruses, which are proficient at entering cells, but that approach carries the risk that some of the viral DNA will be incorporated into the host cell. This application is commonly used in lab experiments but has not been approved by the US Food and Drug and Administration (FDA) for use in human patients.
Another way to transport large molecules into a cell is to tag them with a short protein that can penetrate the cell membrane and tug the larger payload along with it. Alternatively, DNA or proteins can be packaged into synthetic nanoparticles that can enter cells. However, these systems frequently need to be remodified depending on the type of cell and substance being delivered. Moreover, with some nanoparticles, a lot of the material ends up stuck in protective sacs called endosomes inside the cell, and there can be potential toxic side effects.
Electroporation, which involves jolting cells with electricity that opens up the cell membrane, is a more general approach but can be damaging to both cells and the material being delivered.
The new MIT system appears to work for many cell types—up to now, the researchers have successfully tested it with more than a dozen types, including both human and mouse cells. It also works in cells taken directly from human patients, which are typically much more difficult to engineer than human cell lines grown specifically for lab research.
The new device builds on earlier research by Jensen and Langer’s labs, in which they used microinjection to push large molecules into cells as they flowed through a microfluidic device. This was not as fast as the researchers hoped, but during these studies, they discovered that when a cell is squeezed through a narrow tube, small holes open in the cell membrane, allowing neighboring molecules to diffuse into the cell.
To take advantage of that, the researchers built rectangular microfluidic chips, about the size of a quarter, with 40 to 70 parallel channels. Cells are suspended in a solution with the material to be delivered and flowed through the channel at high speed—approximately one meter per second. Halfway through the channel, the cells pass through a constriction about 30%–80% smaller than the cells’ diameter. The cells do not sustain any permanent damage, and they maintain their normal functions after the treatment.
The scientists are now studying stem cell manipulation, which has potential for treating a wide range of diseases. They have already shown that they can convert human fibroblast cells into pluripotent stem cells, and now plan to start working on delivering the proteins needed to differentiate stem cells into specialized tissues.
Another promising application is delivering quantum dots—nanoparticles made of semiconducting metals that fluoresce. These dots hold promise for labeling individual proteins or other molecules inside cells, but scientists have had trouble getting them through the cell membrane without being trapped in endosomes.
In earlier work in November 2012, working with MIT graduate student Jungmin Lee and chemistry professor Dr. Moungi Bawendi, the researchers demonstrated that they could get quantum dots inside human cells grown in the laboratory, without the particles becoming confined in endosomes or clumping together. They are now working on getting the dots to tag specific proteins inside the cells.
The researchers are also exploring the possibility of using the new system for vaccination. In theory, scientists could take immune cells from a patient, run them through the microfluidic device and expose them to a viral protein, and then put them back in the patient. Once inside, the cells could provoke an immune response that would confer immunity against the target viral protein.
Related Links:
Massachusetts Institute of Technology
Living cells are enclosed by a membrane that closely controls what gets in and out of the cell. This barrier is necessary for cells to control their internal environment, but it makes it more difficult for scientists to deliver large molecules such as nanoparticles for imaging, or proteins that can reprogram them into pluripotent stem cells.
Using this technique, the Massachusetts Institute of Technology (MIT; Cambridge, MA, USA: www.mit.edu) researchers were able to deliver reprogramming proteins and create induced pluripotent stem cells with a success rate 10 to 100 times superior than any existing application. They also used it to deliver nanoparticles, including quantum dots and carbon nanotubes, which can be used to image cells and track what is occurring inside them.
“It’s very useful to be able to get large molecules into cells. We thought it might be interesting if you could have a relatively simple system that could deliver many different compounds,” said Dr. Klavs Jensen, a professor of chemical engineering, professor of materials science and engineering, and a senior author of a paper describing the new device in this week’s issue of the Proceedings of the National Academy of Sciences of the United States of America (PNAS).
Scientists had earlier developed several approaches to get large molecules into cells, but all of them have downsides. DNA or RNA can be parceled into viruses, which are proficient at entering cells, but that approach carries the risk that some of the viral DNA will be incorporated into the host cell. This application is commonly used in lab experiments but has not been approved by the US Food and Drug and Administration (FDA) for use in human patients.
Another way to transport large molecules into a cell is to tag them with a short protein that can penetrate the cell membrane and tug the larger payload along with it. Alternatively, DNA or proteins can be packaged into synthetic nanoparticles that can enter cells. However, these systems frequently need to be remodified depending on the type of cell and substance being delivered. Moreover, with some nanoparticles, a lot of the material ends up stuck in protective sacs called endosomes inside the cell, and there can be potential toxic side effects.
Electroporation, which involves jolting cells with electricity that opens up the cell membrane, is a more general approach but can be damaging to both cells and the material being delivered.
The new MIT system appears to work for many cell types—up to now, the researchers have successfully tested it with more than a dozen types, including both human and mouse cells. It also works in cells taken directly from human patients, which are typically much more difficult to engineer than human cell lines grown specifically for lab research.
The new device builds on earlier research by Jensen and Langer’s labs, in which they used microinjection to push large molecules into cells as they flowed through a microfluidic device. This was not as fast as the researchers hoped, but during these studies, they discovered that when a cell is squeezed through a narrow tube, small holes open in the cell membrane, allowing neighboring molecules to diffuse into the cell.
To take advantage of that, the researchers built rectangular microfluidic chips, about the size of a quarter, with 40 to 70 parallel channels. Cells are suspended in a solution with the material to be delivered and flowed through the channel at high speed—approximately one meter per second. Halfway through the channel, the cells pass through a constriction about 30%–80% smaller than the cells’ diameter. The cells do not sustain any permanent damage, and they maintain their normal functions after the treatment.
The scientists are now studying stem cell manipulation, which has potential for treating a wide range of diseases. They have already shown that they can convert human fibroblast cells into pluripotent stem cells, and now plan to start working on delivering the proteins needed to differentiate stem cells into specialized tissues.
Another promising application is delivering quantum dots—nanoparticles made of semiconducting metals that fluoresce. These dots hold promise for labeling individual proteins or other molecules inside cells, but scientists have had trouble getting them through the cell membrane without being trapped in endosomes.
In earlier work in November 2012, working with MIT graduate student Jungmin Lee and chemistry professor Dr. Moungi Bawendi, the researchers demonstrated that they could get quantum dots inside human cells grown in the laboratory, without the particles becoming confined in endosomes or clumping together. They are now working on getting the dots to tag specific proteins inside the cells.
The researchers are also exploring the possibility of using the new system for vaccination. In theory, scientists could take immune cells from a patient, run them through the microfluidic device and expose them to a viral protein, and then put them back in the patient. Once inside, the cells could provoke an immune response that would confer immunity against the target viral protein.
Related Links:
Massachusetts Institute of Technology
Latest BioResearch News
- Telomere Length Abnormalities Linked to Lymphoma Development
- Biomarker Signals Chemotherapy Resistance in Relapsed Small Cell Lung Cancer
- Inflammatory Gene Signature Links Metabolic Disease to Pancreatic Cancer Recurrence
- Study Links Abnormal Gene Splicing to Treatment Response in Metastatic Kidney Cancer
- Research Reveals How Some Aplastic Anemia Patients Recover Bone Marrow Function
- New Molecular Insights Support Diagnosis of Hodgkin Lymphoma
- Epigenetic Signals and Blood Markers Aid Chronic Fatigue Syndrome Diagnosis
- Microenvironment Biomarkers Could Enable Early Lung Cancer Detection
- Study Identifies Protein Changes Driving Immunotherapy Resistance in Multiple Myeloma
- Genetic Analysis Identifies BRCA-Linked Risks Across Multiple Cancers
- Study Identifies Hidden B-Cell Mutations in Autoimmune Disease
- Single-Cell Method Measures RNA and Proteins to Reveal Immune Responses
- Study Links Midlife Vitamin D to Lower Tau in Alzheimer's
- International Consensus Standardizes Tumor Microbiota Detection and Reporting
- Common Metablolic Enzyme Could Predict Response to Cancer Immunotherapy
- Newly Identfied Genetic Variants in MND Support Prognosis and Family Testing
Channels
Clinical Chemistry
view channel
Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
Delays in diagnosing serious psychiatric conditions can leave patients without timely support and complicate treatment planning. For bipolar disorder, average time to diagnosis can exceed nine years, and... Read moreAt-Home Blood and Cognitive Tests Support Dementia Risk Stratification
Dementia places a substantial burden on patients and health systems, and identifying individuals at elevated risk remains difficult at scale. In the UK, almost a million people are estimated to be living... Read moreMolecular Diagnostics
view channel
Digital Aging Twin Quantifies Biological Aging Across Multiple Organ Systems
Chronological age often fails to capture the wide variability in physiological decline among adults, limiting risk stratification and long-term monitoring. Clinical laboratories also lack standardized... Read more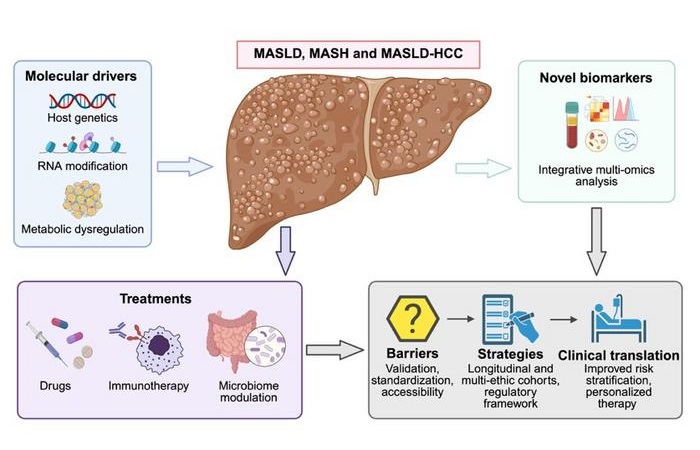
Emerging Biomarkers Advance Early Detection of MASLD and Liver Cancer Risk
Metabolic dysfunction-associated steatotic liver disease (MASLD) affects about 30% of people worldwide and can advance to metabolic dysfunction-associated steatohepatitis (MASH), fibrosis, cirrhosis, and... Read more
Patented Isothermal Amplification Chemistry Advances Decentralized Testing
Molecular diagnostics offer high sensitivity for pathogen detection but typically rely on thermal cycling and specialized instruments, limiting their use outside centralized laboratories.... Read more
Finger-Prick Blood Test Aids Early Tuberculosis Detection and Risk Stratification
Household contacts of people with tuberculosis face an estimated 2% risk of developing disease, yet most are asymptomatic at the time of screening. Early-stage cases are often missed because symptom checks... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read moreMicrobiology
view channel
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read more
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more