Synchrotron X-Ray Crystallography Generates Insulin-Insulin Receptor Binding Images
|
By LabMedica International staff writers Posted on 24 Jan 2013 |
The light generated by a state-of-the-art particle accelerator was used to capture X-ray crystallographic images of the three-dimensional interaction between insulin and its receptor.
Insulin receptor signaling has a central role in mammalian biology, regulating cellular metabolism, growth, division, differentiation, and survival. Insulin resistance contributes to the development of diseases such as type II diabetes mellitus and Alzheimer’s disease. Abnormal signaling generated by cross talk with the homologous type 1 insulin-like growth factor receptor (IGF1R) occurs in various cancers. Despite more than thirty years of research, it has not been possible to document the three-dimensional structure of the insulin-insulin receptor due to the complexity of producing the receptor protein.
In a paper published in the January 9, 2013, online edition of the journal Nature an international research time described the use of the Australian Synchrotron to capture the three-dimensional structure of insulin bound to the insulin receptor.
The Australian Synchrotron (Clayton, Australia) is a light source facility that uses particle accelerators to produce a beam of high-energy electrons that are placed within a storage ring that circulates the electrons to create synchrotron light. The electron beams travel at just under the speed of light - about 299,792 kilometers per second, and the intense light they produce is filtered and adjusted to travel down separate beamlines to separate end stations where are placed a variety of experimental equipment including one for protein crystallography.
X-ray crystallographic images generated by the Synchrotron revealed the sparse direct interaction of insulin with the first leucine-rich-repeat domain (L1) of the insulin receptor. Instead, the hormone engaged the insulin receptor carboxy-terminal alpha-chain (alphaCT) segment, which was itself remodeled on the face of L1 upon insulin binding. Contact between insulin and L1 was restricted to insulin B-chain residues. The alphaCT segment displaced the B-chain C-terminal beta-strand away from the hormone core, revealing the mechanism of a long-proposed conformational switch in insulin upon receptor engagement. This mode of hormone-receptor recognition is thought to be novel within the broader family of receptor tyrosine kinases.
"We have now found that the insulin hormone engages its receptor in a very unusual way," said senior author Dr. Michael C. Lawrence, associate professor of structural biology at Walter and Eliza Hall Institute of Medical Research (Melbourne, Australia). "Both insulin and its receptor undergo rearrangement as they interact - a piece of insulin folds out and key pieces within the receptor move to engage the insulin hormone."
"Understanding how insulin interacts with the insulin receptor is fundamental to the development of novel insulins for the treatment of diabetes," said Dr. Lawrence. "Until now we have not been able to see how these molecules interact with cells. We can now exploit this knowledge to design new insulin medications with improved properties, which is very exciting. Insulin is a key treatment for diabetics, but there are many ways that its properties could potentially be improved. This discovery could conceivably lead to new types of insulin that could be given in ways other than injection, or an insulin that has improved properties or longer activity so that it does not need to be taken as often. It may also have ramifications for diabetes treatment in developing nations, by creating insulin that is more stable and less likely to degrade when not kept cold, an angle being pursued by our collaborators. Our findings are a new platform for developing these kinds of medications."
Related Links:
Australian Synchrotron
Walter and Eliza Hall Institute of Medical Research
Insulin receptor signaling has a central role in mammalian biology, regulating cellular metabolism, growth, division, differentiation, and survival. Insulin resistance contributes to the development of diseases such as type II diabetes mellitus and Alzheimer’s disease. Abnormal signaling generated by cross talk with the homologous type 1 insulin-like growth factor receptor (IGF1R) occurs in various cancers. Despite more than thirty years of research, it has not been possible to document the three-dimensional structure of the insulin-insulin receptor due to the complexity of producing the receptor protein.
In a paper published in the January 9, 2013, online edition of the journal Nature an international research time described the use of the Australian Synchrotron to capture the three-dimensional structure of insulin bound to the insulin receptor.
The Australian Synchrotron (Clayton, Australia) is a light source facility that uses particle accelerators to produce a beam of high-energy electrons that are placed within a storage ring that circulates the electrons to create synchrotron light. The electron beams travel at just under the speed of light - about 299,792 kilometers per second, and the intense light they produce is filtered and adjusted to travel down separate beamlines to separate end stations where are placed a variety of experimental equipment including one for protein crystallography.
X-ray crystallographic images generated by the Synchrotron revealed the sparse direct interaction of insulin with the first leucine-rich-repeat domain (L1) of the insulin receptor. Instead, the hormone engaged the insulin receptor carboxy-terminal alpha-chain (alphaCT) segment, which was itself remodeled on the face of L1 upon insulin binding. Contact between insulin and L1 was restricted to insulin B-chain residues. The alphaCT segment displaced the B-chain C-terminal beta-strand away from the hormone core, revealing the mechanism of a long-proposed conformational switch in insulin upon receptor engagement. This mode of hormone-receptor recognition is thought to be novel within the broader family of receptor tyrosine kinases.
"We have now found that the insulin hormone engages its receptor in a very unusual way," said senior author Dr. Michael C. Lawrence, associate professor of structural biology at Walter and Eliza Hall Institute of Medical Research (Melbourne, Australia). "Both insulin and its receptor undergo rearrangement as they interact - a piece of insulin folds out and key pieces within the receptor move to engage the insulin hormone."
"Understanding how insulin interacts with the insulin receptor is fundamental to the development of novel insulins for the treatment of diabetes," said Dr. Lawrence. "Until now we have not been able to see how these molecules interact with cells. We can now exploit this knowledge to design new insulin medications with improved properties, which is very exciting. Insulin is a key treatment for diabetics, but there are many ways that its properties could potentially be improved. This discovery could conceivably lead to new types of insulin that could be given in ways other than injection, or an insulin that has improved properties or longer activity so that it does not need to be taken as often. It may also have ramifications for diabetes treatment in developing nations, by creating insulin that is more stable and less likely to degrade when not kept cold, an angle being pursued by our collaborators. Our findings are a new platform for developing these kinds of medications."
Related Links:
Australian Synchrotron
Walter and Eliza Hall Institute of Medical Research
Latest BioResearch News
- Study Identifies Protein Changes Driving Immunotherapy Resistance in Multiple Myeloma
- Genetic Analysis Identifies BRCA-Linked Risks Across Multiple Cancers
- Study Identifies Hidden B-Cell Mutations in Autoimmune Disease
- Single-Cell Method Measures RNA and Proteins to Reveal Immune Responses
- Study Links Midlife Vitamin D to Lower Tau in Alzheimer's
- International Consensus Standardizes Tumor Microbiota Detection and Reporting
- Common Metablolic Enzyme Could Predict Response to Cancer Immunotherapy
- Newly Identfied Genetic Variants in MND Support Prognosis and Family Testing
- Innate Immunity Variants Associated With Earlier Breast Cancer in BRCA1 Carriers
- Genetic Cause Identified for Severe Infant Epilepsy
- Study Reveals Diagnostic and Therapeutic Target in Rare Pancreatic Tumors
- Researchers Identify Survival Pathway Undermining Targeted Cancer Drugs
- Large-Scale Study Maps DNA Damage Signatures Across Multiple Cancers
- Study Identifies Distinct Immune Signatures to Early Depression and Psychosis
- Genetic Mutation Behind Aggressive Adult Leukemia Offers Treatment Clues
- Disease Gene Discovery Advances Diagnosis of Rare Movement Disorders
Channels
Clinical Chemistry
view channel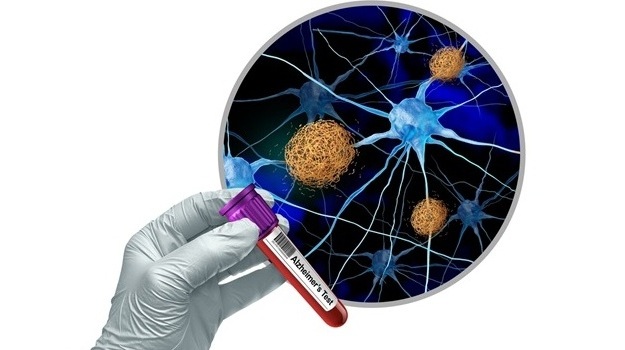
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read more
Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
Routine blood tests that measure low-density lipoprotein (LDL), commonly known as “bad” cholesterol, are widely used to guide lipid-lowering therapy, but they do not always provide a complete picture of... Read more
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel
Blood-Based Epigenetic Signals Enable Osteosarcoma Disease Monitoring
Osteosarcoma is a rare but aggressive pediatric bone cancer where recurrence and metastasis remain difficult to detect early. Imaging-based surveillance can miss small lesions and exposes children to repeated... Read more
Host–Virus Genetic Interactions Drive Nasopharyngeal Cancer Risk
Epstein–Barr virus (EBV) infects more than 95% of adults worldwide, yet only a small fraction develops EBV‑associated cancers such as nasopharyngeal carcinoma. Explaining this divergence requires understanding... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read moreMicrobiology
view channel
mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
Central nervous system (CNS) infections such as encephalitis, ventriculitis, and brain abscess are among the most time-sensitive diagnostic challenges in neurology. Standard testing can return negative... Read more
Rapid Color Test Stratifies Virulent and Resistant Staph Strains
Staphylococcus aureus (golden staph) remains a leading cause of infection-related mortality worldwide, responsible for more than a million deaths each year. Rapidly distinguishing highly virulent or a... Read morePathology
view channel
AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Reliable prognostic profiling and biomarker screening are essential to guide oncology treatment decisions, while laboratories must balance speed and resource constraints. Earlier identification of high‑risk... Read more
Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
Cutaneous squamous cell carcinoma (cSCC) is the world’s second most common skin cancer, and while many cases are treatable, a subset becomes highly aggressive and therapy‑resistant. Identifying molecular... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
Foundation Medicine to Expand MRD Portfolio with Pathlight Platform
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more