POC COVID-19 Test Detects and Differentiates SARS-CoV-2 Alpha Variant from Earlier Strains
|
By LabMedica International staff writers Posted on 20 Apr 2022 |

A newly-developed point-of-care COVID-19 test can detect and differentiate the alpha variant of the SARS-CoV-2 virus from earlier strains in saliva samples.
The new test developed by researchers at the University of Illinois at Urbana-Champaign (Champaign, IL, USA) builds on their previous developments, which allowed samples to bypass the laboratory – first using nasopharyngeal swabs, then with saliva samples. The point-of-care amplification and testing process, called LAMP, is more efficient than PCR because it does not require expensive thermal cycling machines. According to the researchers, the assay does not need RNA extraction and purification steps, similar to the Illinois saliva test.
The updated process takes advantage of a genetic phenomenon called S-gene target failure – which is present in the alpha variant but not in the SARS-CoV-2 virus early strains – to differentiate between the two alpha variants, the researchers said. During the new testing process, specially selected genetic primers are placed onto additively manufactured cartridges and dried before adding the patient samples. The chosen primers are set against the S-gene and are specific for detecting the S-gene target failure against 69–70 deletion in the alpha variant.
Their study confirmed the device’s effectiveness by testing 38 clinical saliva samples, including 20 samples positive for alpha variant. The researchers would like to refine their method to test up to five different viruses, viral strains and variants in a single test, compatible with nasal swab and saliva mediums.
“Our study shows that it is possible to test for variants of the same coronavirus strain in a single point-of-care test that takes 30 minutes using a portable handheld device,” said Rashid Bashir, a professor of bioengineering and the dean of the Grainger College of Engineering at Illinois, who co-led the study. “The new test is scalable to suit future pandemics, COVID-19 or otherwise, and could be used at home or other settings.”
“The new omicron variant also exhibits S-gene target failure and could be tested for by adapting the approach developed in this study,” added bioengineering professor Enrique Valera.
Related Links:
University of Illinois at Urbana-Champaign
Latest COVID-19 News
- New Immunosensor Paves Way to Rapid POC Testing for COVID-19 and Emerging Infectious Diseases
- Long COVID Etiologies Found in Acute Infection Blood Samples
- Novel Device Detects COVID-19 Antibodies in Five Minutes
- CRISPR-Powered COVID-19 Test Detects SARS-CoV-2 in 30 Minutes Using Gene Scissors
- Gut Microbiome Dysbiosis Linked to COVID-19
- Novel SARS CoV-2 Rapid Antigen Test Validated for Diagnostic Accuracy
- New COVID + Flu + R.S.V. Test to Help Prepare for `Tripledemic`
- AI Takes Guesswork Out Of Lateral Flow Testing
- Fastest Ever SARS-CoV-2 Antigen Test Designed for Non-Invasive COVID-19 Testing in Any Setting
- Rapid Antigen Tests Detect Omicron, Delta SARS-CoV-2 Variants
- Health Care Professionals Showed Increased Interest in POC Technologies During Pandemic, Finds Study
- Set Up Reserve Lab Capacity Now for Faster Response to Next Pandemic, Say Researchers
- Blood Test Performed During Initial Infection Predicts Long COVID Risk
- Low-Cost COVID-19 Testing Platform Combines Sensitivity of PCR and Speed of Antigen Tests
- Finger-Prick Blood Test Identifies Immunity to COVID-19
- Quick Test Kit Determines Immunity Against COVID-19 and Its Variants
Channels
Clinical Chemistry
view channel
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read more
Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
Routine blood tests that measure low-density lipoprotein (LDL), commonly known as “bad” cholesterol, are widely used to guide lipid-lowering therapy, but they do not always provide a complete picture of... Read more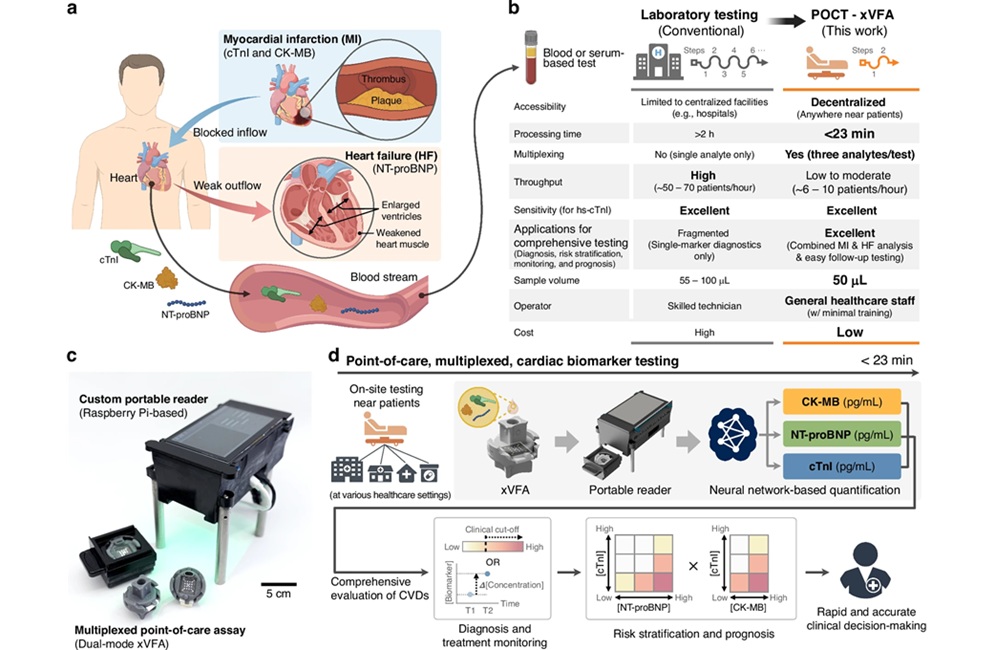
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel
Blood-Based Epigenetic Signals Enable Osteosarcoma Disease Monitoring
Osteosarcoma is a rare but aggressive pediatric bone cancer where recurrence and metastasis remain difficult to detect early. Imaging-based surveillance can miss small lesions and exposes children to repeated... Read more
Host–Virus Genetic Interactions Drive Nasopharyngeal Cancer Risk
Epstein–Barr virus (EBV) infects more than 95% of adults worldwide, yet only a small fraction develops EBV‑associated cancers such as nasopharyngeal carcinoma. Explaining this divergence requires understanding... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read moreMicrobiology
view channel
Rapid Color Test Stratifies Virulent and Resistant Staph Strains
Staphylococcus aureus (golden staph) remains a leading cause of infection-related mortality worldwide, responsible for more than a million deaths each year. Rapidly distinguishing highly virulent or a... Read more
Syndromic Panel Enables Rapid Identification of Bloodstream Infections
Bloodstream infections require rapid identification of causative pathogens and resistance determinants to guide therapy, yet laboratories often face pressure to deliver clinically relevant results quickly... Read more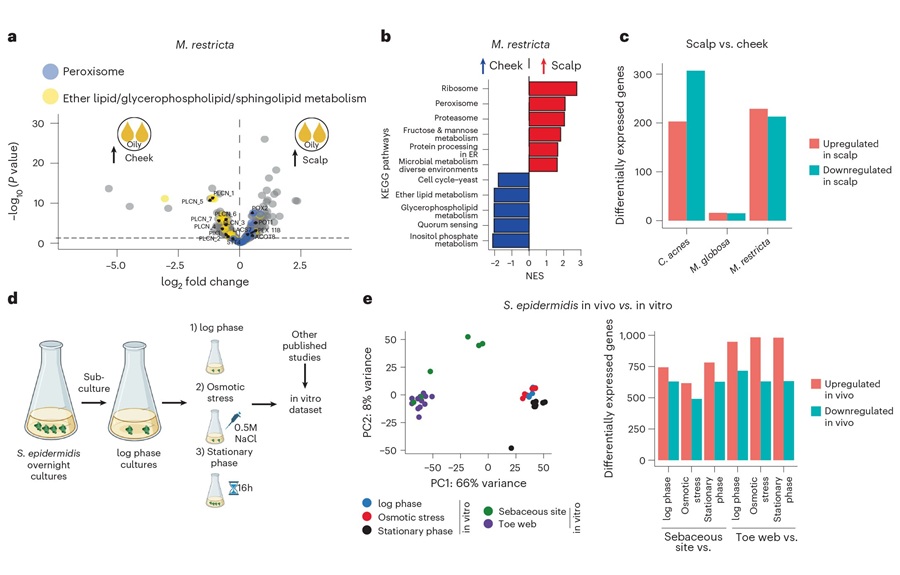
RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
Human skin carries diverse microbial communities that influence barrier function and inflammation, yet identifying which organisms are metabolically active has been challenging. DNA-based surveys catalog... Read more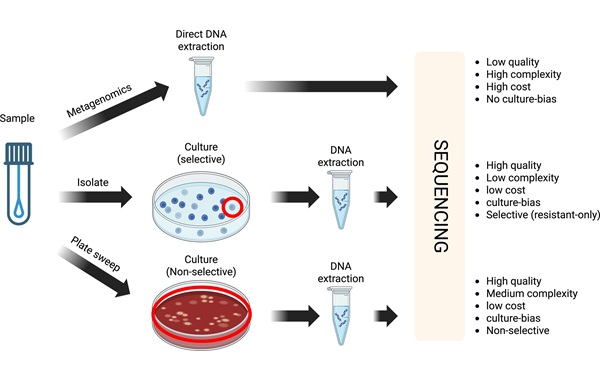
Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
Intensive care units face persistent threats from hospital-acquired infections, increasingly driven by drug-resistant bacteria. Rapidly pinpointing environmental reservoirs and transmission hotspots remains... Read morePathology
view channel
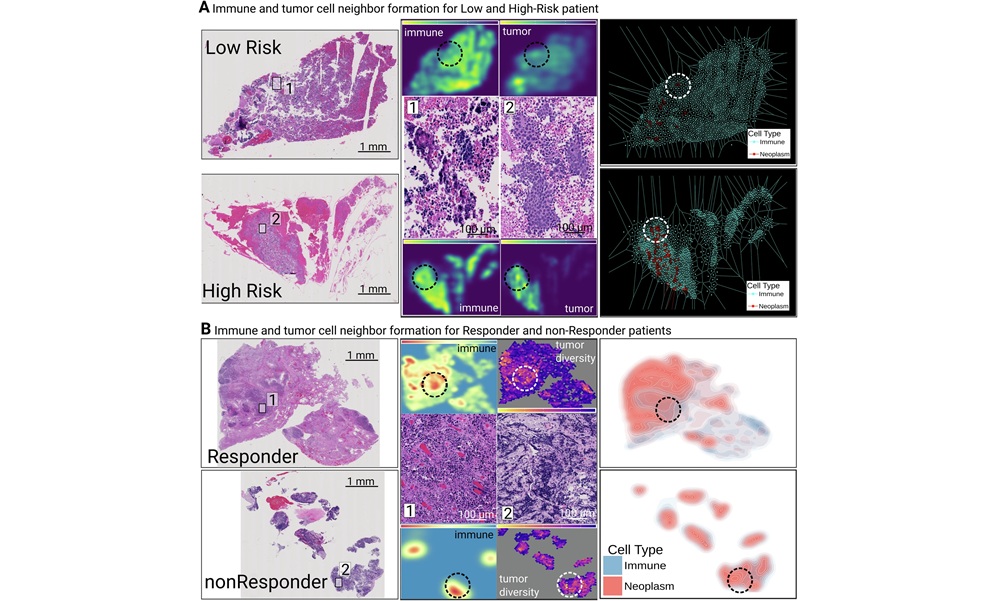
Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
Colorectal cancer is common and often lethal, and therapeutic decision-making is complicated by heterogeneous tumor microenvironments. Immunotherapy benefits only a small subset of patients, around 5%,... Read more
Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
Antibody-drug conjugates (ADC) are reshaping oncology, yet scalable biomarkers that reliably predict which patients will benefit remain limited as treatment regimens and combinations grow more complex.... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
Partnership Expands Ultrasensitive WGS Assay for for Hematologic Malignancies and MRD Monitoring
Tempus AI and Predicta Biosciences announced the commercial expansion of a co-branded whole‑genome sequencing assay GenoPredicta, which is intended for comprehensive genomic characterization of hematologic... Read more