Phosphorylation of Tau Protein Inhibits Amyloid-beta Toxicity in Alzheimer’s Model
|
By Gerald M. Slutzky, PhD Posted on 29 Nov 2016 |
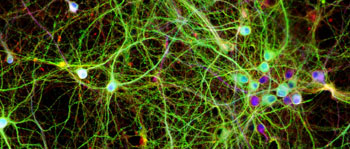
Image: A photomicrograph of neurons growing in culture. The colors highlight the human tau protein in green, a structural component in red, and the DNA inside the cell nucleus in blue (Photo courtesy of Dr. Lars Ittner, University of New South Wales).
A team of Australian Alzheimer's disease (AD) researchers have presented evidence suggesting that phosphorylation of tau protein in the early stages of the disease acts to protect against the toxicity of amyloid-beta (Abeta) plaques, and that this protective effect disappears as the disease progresses.
The prevailing idea among AD researchers has been that Abeta induced phosphorylation of tau, which in turn triggered the neuronal dysfunction that characterized the disease.
Investigators at the University of New South Wales (Sydney, Australia) worked with Alzheimer's disease mouse models and samples of brain tissue obtained from AD patients. They reported in the November 18, 2016, issue of the journal Science that at least in early stages of the disease, site-specific phosphorylation of tau inhibited Abeta toxicity. This specific tau phosphorylation was mediated by the neuronal enzyme p38gamma (p38 mitogen-activated protein kinase) and interfered with postsynaptic toxic signaling complexes engaged by Abeta.
Depletion of p38gamma increased the severity of neuronal circuit aberrations, cognitive deficits, and premature lethality in a mouse model of AD, whereas increasing the activity of p38gamma abolished these deficits. Furthermore, mimicking site-specific tau phosphorylation alleviated Abeta-induced neuronal death and offered protection from its toxicity.
"This study has completely changed our understanding of what happens in the brain during the development of Alzheimer's disease," said senior author Dr. Lars Ittner, professor of medicine at the University of New South Wales. "Amyloid-beta induces toxicity in the neurons but the first step in tau phosphorylation is actually to decrease this toxicity. This is a completely new mindset; that the reason tau becomes modified is actually to protect from damage. We found that p38gamma, which initially offers protection, fades away early in the brains of people with AD, suggesting a loss of protection."
"We set out to find mediators of this progression, which led us quickly to our surprising finding. It was the opposite of what we expected. It was only when we changed our view of the process involved in the development of AD that these results started to make sense," said Dr. Ittner. "We used mice to screen for a very specific toxicity that we knew from previous work is involved in the progression of the disease. Part of our study involved reintroducing p38gamma and increasing its activity. We saw that, in mice, it could prevent memory deficits from happening, so it has true therapeutic potential. If we can stimulate that activity, we may be able to delay or even halt the progression of Alzheimer's disease."
Related Links:
University of New South Wales
The prevailing idea among AD researchers has been that Abeta induced phosphorylation of tau, which in turn triggered the neuronal dysfunction that characterized the disease.
Investigators at the University of New South Wales (Sydney, Australia) worked with Alzheimer's disease mouse models and samples of brain tissue obtained from AD patients. They reported in the November 18, 2016, issue of the journal Science that at least in early stages of the disease, site-specific phosphorylation of tau inhibited Abeta toxicity. This specific tau phosphorylation was mediated by the neuronal enzyme p38gamma (p38 mitogen-activated protein kinase) and interfered with postsynaptic toxic signaling complexes engaged by Abeta.
Depletion of p38gamma increased the severity of neuronal circuit aberrations, cognitive deficits, and premature lethality in a mouse model of AD, whereas increasing the activity of p38gamma abolished these deficits. Furthermore, mimicking site-specific tau phosphorylation alleviated Abeta-induced neuronal death and offered protection from its toxicity.
"This study has completely changed our understanding of what happens in the brain during the development of Alzheimer's disease," said senior author Dr. Lars Ittner, professor of medicine at the University of New South Wales. "Amyloid-beta induces toxicity in the neurons but the first step in tau phosphorylation is actually to decrease this toxicity. This is a completely new mindset; that the reason tau becomes modified is actually to protect from damage. We found that p38gamma, which initially offers protection, fades away early in the brains of people with AD, suggesting a loss of protection."
"We set out to find mediators of this progression, which led us quickly to our surprising finding. It was the opposite of what we expected. It was only when we changed our view of the process involved in the development of AD that these results started to make sense," said Dr. Ittner. "We used mice to screen for a very specific toxicity that we knew from previous work is involved in the progression of the disease. Part of our study involved reintroducing p38gamma and increasing its activity. We saw that, in mice, it could prevent memory deficits from happening, so it has true therapeutic potential. If we can stimulate that activity, we may be able to delay or even halt the progression of Alzheimer's disease."
Related Links:
University of New South Wales
Latest BioResearch News
- Lung Cancer Study Reveals Cellular Program Behind Therapy Resistance
- Tumor Genome Marker May Predict Treatment Benefit in Pediatric Cancers
- Lysosomal Gene Defect Linked to Severe Childhood Brain Disorders
- Genetic Testing Identifies Greater Inherited Sudden Cardiac Arrest Risk in Younger Individuals
- Hidden 'Jumping Gene' Variant Linked to Higher Pancreatic Cancer Risk
- Common White Blood Cells Produce Schizophrenia-Linked Protein
- Nanopore Method Captures RNA Folding at Single-Molecule Resolution
- Tumor Microenvironment Marker Linked to Worse Survival in Solid Tumors
- Hidden Immune Gene Defect May Explain Kaposi Sarcoma Susceptibility
- Genetic Markers May Help Predict Amputation Risk in Peripheral Artery Disease
- Gene Signature Shows Promise for Depression Biomarker Testing
- AI-Driven Tumor Profiling Initiative Targets Precision Therapy Development
- Researchers Map Protein and Glycosylation Across 15 Human Body Fluids
- Telomere Length Abnormalities Linked to Lymphoma Development
- Biomarker Signals Chemotherapy Resistance in Relapsed Small Cell Lung Cancer
- Inflammatory Gene Signature Links Metabolic Disease to Pancreatic Cancer Recurrence
Channels
Clinical Chemistry
view channel
Simple Dual-Tau Blood Test Detects and Stages Alzheimer’s Disease
Alzheimer’s disease is typically confirmed and staged with positron emission tomography scans and cerebrospinal fluid testing, procedures that are costly and invasive. Broader access to minimally invasive... Read more
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read moreMolecular Diagnostics
view channel
Genomic Test Guides Chemotherapy Decisions in Early-Stage Breast Cancer
Selecting adjuvant therapy for early-stage hormone receptor-positive breast cancer depends on accurately assessing long-term risk of distant recurrence. Clinical features alone can leave uncertainty about... Read more
Simple Cytogenetic Method Could Improve Classification of ALL Subtypes
Many cancers deviate from the normal chromosome number, but the clinical impact of extreme chromosome loss remains unclear. This widespread genomic disruption is associated with aggressive disease and... Read more
Blood-Based Assay Enables Noninvasive Monitoring of Sarcoma Immunotherapy Response
Sarcomas remain difficult to monitor during immunotherapy, as low tumor mutation burden can limit traditional circulating tumor DNA approaches and repeat tissue biopsies are often impractical in advanced disease.... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
Blood-Based Method Tracks Gene Activity in the Living Brain
Real-time measurement of gene activity in the brain has been limited by assays requiring destructive tissue sampling. Tracking active genes could reveal how the body responds to environmental factors,... Read more
FDA Approval Expands Automated PD-L1 Testing Across Solid Tumors
Clinical laboratories play a central role in guiding immunotherapy by reporting programmed death ligand-1 (PD‑L1) status across multiple solid tumors. Many sites are standardizing this work on fully automated... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel