Robust System Offers High Multiplexing Capabilities
|
By LabMedica International staff writers Posted on 25 Nov 2008 |
A robust multiplexing system for medium and high volume laboratories measures up to 500 analytes in a single well.
Called the FlexMap 3D system, the new technology provides both protein and nucleic acid applications on one platform, and has applications in market segments such as molecular diagnostics, HLA testing, life science research, and markets as diverse as biodefense, animal health, and agro-biotech.
Designed by Luminex Corporation (Austin, TX, USA) the FlexMap 3D (three dimensional) is suitable for clinical reference labs, pharmaceutic labs, and academic core labs that are interested in enhanced workflow and higher multiplexing capabilities using the company's xMap bead-based technology. The new instrument displays improved analytical performance, front-end automation, and enhanced throughput. It is built on flow cytometry, microspheres, lasers, digital signal processing, and traditional chemistry. Featuring a flexible, open-architecture design, xMap technology can be configured to perform a wide variety of bioassays quickly, cost-effectively, and accurately.
"We are very excited to announce our first commercial shipment of FlexMap 3D," said Douglas Bryant, executive vice president and COO of Luminex. "The increased speed, increased multiplexing capabilities, as well as the improved workflow of the instrument will offer researchers and clinicians rapid and accurate results to improve the health and well-being of people around the world." Full market release of the instrument is expected in the first quarter of 2009.
The Luminex systems are flexible analyzers based on the principles of three-dimensional bead arrays. Due to the enhanced liquid kinetics of the microspheres in solution, these arrays offer superior uniformity and faster reaction times than standard two-dimensional planar arrays. The proprietary microsphere dyeing process also provides higher levels of multiplexing than traditional methods like enzyme-linked immunosorbant assays (ELISAs) or the real-time polymerase chain reaction (RT-PCR). Luminex licenses its instruments and array-based assays to companies in clinical diagnostics and biologic research
Related Links:
Luminex Corporation
Called the FlexMap 3D system, the new technology provides both protein and nucleic acid applications on one platform, and has applications in market segments such as molecular diagnostics, HLA testing, life science research, and markets as diverse as biodefense, animal health, and agro-biotech.
Designed by Luminex Corporation (Austin, TX, USA) the FlexMap 3D (three dimensional) is suitable for clinical reference labs, pharmaceutic labs, and academic core labs that are interested in enhanced workflow and higher multiplexing capabilities using the company's xMap bead-based technology. The new instrument displays improved analytical performance, front-end automation, and enhanced throughput. It is built on flow cytometry, microspheres, lasers, digital signal processing, and traditional chemistry. Featuring a flexible, open-architecture design, xMap technology can be configured to perform a wide variety of bioassays quickly, cost-effectively, and accurately.
"We are very excited to announce our first commercial shipment of FlexMap 3D," said Douglas Bryant, executive vice president and COO of Luminex. "The increased speed, increased multiplexing capabilities, as well as the improved workflow of the instrument will offer researchers and clinicians rapid and accurate results to improve the health and well-being of people around the world." Full market release of the instrument is expected in the first quarter of 2009.
The Luminex systems are flexible analyzers based on the principles of three-dimensional bead arrays. Due to the enhanced liquid kinetics of the microspheres in solution, these arrays offer superior uniformity and faster reaction times than standard two-dimensional planar arrays. The proprietary microsphere dyeing process also provides higher levels of multiplexing than traditional methods like enzyme-linked immunosorbant assays (ELISAs) or the real-time polymerase chain reaction (RT-PCR). Luminex licenses its instruments and array-based assays to companies in clinical diagnostics and biologic research
Related Links:
Luminex Corporation
Latest Lab Technologies News
Channels
Clinical Chemistry
view channel
AI-Based Blood Test Diagnose Multiple Brain Disorders from Blood Sample
Diagnosing the cause of age-related cognitive symptoms remains challenging because clinical presentations of neurodegenerative diseases often overlap, and multiple pathologies can co-occur... Read more
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read moreMolecular Diagnostics
view channel
RNA Profiling Uncovers Therapeutic Targets in Solid Tumors
Many patients with advanced solid tumors exhaust broad DNA panel testing yet still lack biomarkers that match guideline-recommended therapies, limiting access to targeted options. Expanding molecular profiling... Read more
Whole Genome Sequencing in Routine Care Expands Rare Disease Detection
Rare diseases often involve prolonged diagnostic journeys that delay clinical decision-making and complicate family planning. As phenotypes become more heterogeneous, sequencing-based methods are increasingly... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
Active tuberculosis (TB) remains a leading cause of death and illness worldwide, yet distinguishing contagious disease from latent infection continues to challenge clinicians. Standard screening tools... Read more
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
New Bacterial Target Identified for Early Detection of Noma
Noma is a rapidly progressing orofacial infection that begins as gingivitis and can destroy oral and facial tissues, primarily affecting young children living in extreme poverty. Without treatment, it... Read more
Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
Streptococcus dysgalactiae subspecies equisimilis (SDSE) infections are increasing worldwide and include variants that may lead to severe disease. Researchers now report that whole-genome sequencing of... Read morePathology
view channel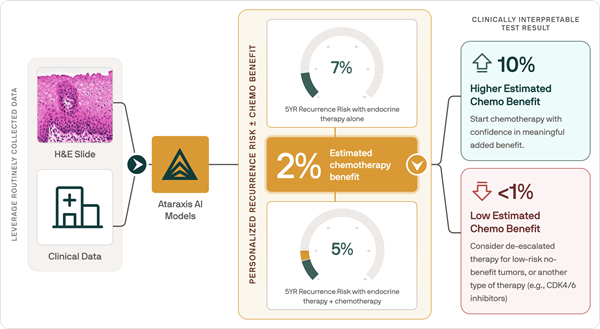
AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer is typically guided by recurrence risk and population-level averages rather than patient-specific benefit. However, existing clinicopathologic... Read more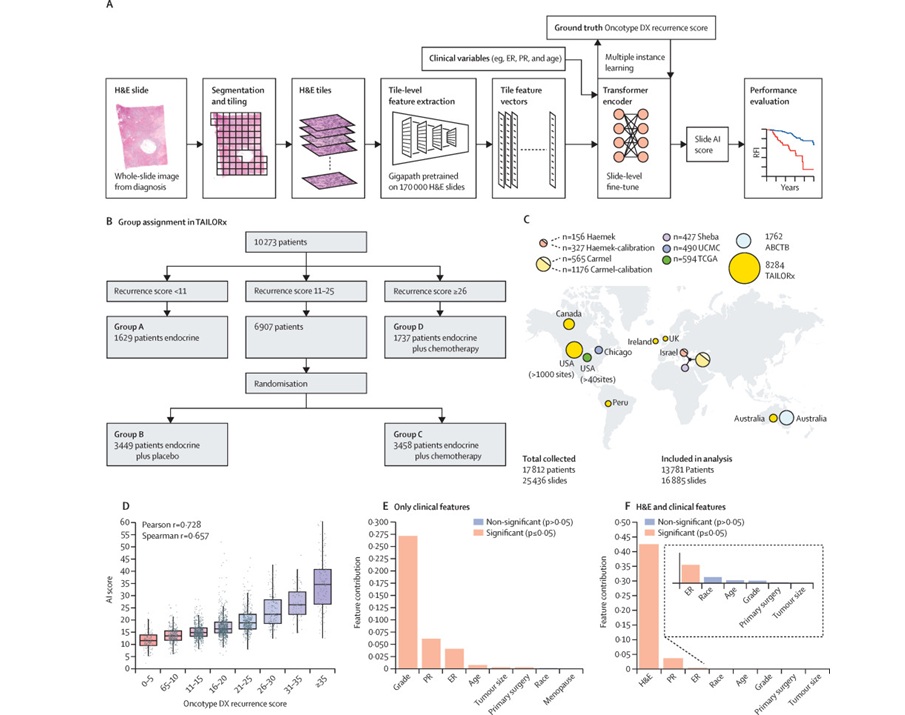
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read moreIndustry
view channel
Takara Bio USA and Hamilton Partner Partner to Automate NGS Library Preparation
Takara Bio USA, Inc. (San Jose, CA, USA), a wholly owned subsidiary of Takara Bio Inc., and Hamilton Company (Reno, NV, USA) announced a development and co-marketing agreement to deliver integrated, automated... Read more