WorldLab-EuroMedLab 2023 Technical Exhibition: Review of Innovations
|
By LabMedica International staff writers Posted on 19 Jun 2023 |

The 25th International Congress of Clinical Chemistry and Laboratory Medicine (WorldLab) and 25th European Congress of Clinical Chemistry and Laboratory Medicine (EuroMedLab) were hosted by the Italian Society of Clinical Biochemistry and Clinical Molecular Biology (SIBioC, Rome, Italy; www.sibioc.it), on the occasion of their 55th Annual Congress, at the new Rome Convention Center “The Cloud” from May 21st to 25th, 2023. Organized by MZ Events Srl (Milano, Italy; www.mzevents.it), EuroMedLab-WorldLab 2023 featured innovative and diverse educational workshops, including lectures, symposia, recent advancements in clinical practice and science, poster presentations, and industry exhibits. Distinguished international speakers and key opinion leaders delivered presentations in health care, on recent diagnostic technologies, scientific advances, and challenges.
The biennial EuroMedLab Congresses continue to act as leading forums in the field of laboratory medicine and clinical chemistry, bringing together scientists, laboratory specialists, clinicians, and industry to enable scientific exchange and advancement. This year, the Congress adopted a combined approach of both physical and virtual interactions. There was a particular focus on scientific and technological advancements, as the main objective of the meeting was not only to contribute to the advancement of laboratory medicine and to the dissemination of advanced knowledge but also to foster the creation of an opportunity to establish professional and scientific links/bridges among the participants.
Roche (Basel, Switzerland; www.roche.com) showcased its innovative laboratory solutions and digital diagnostics through - "Innovation Past, Present and Future". Roche featured new innovations, such as several molecular systems from its growing PCR portfolio – the cobas 5800 and the new LightCycler PRO System, which will be launched later this year. A prototype for the new cobas Mass Spec system was unveiled, bringing mass spectrometry into the routine testing environment. Roche also introduced navify Sample Tracking, its cloud-based digital solution for labs to track patient test samples before even reaching lab premises.
Siemens Healthineers (Erlangen, Germany; www.siemens-healthineers.com) presented its newest innovations and trends in clinical diagnostics and POC testing at the industry event. On display at the Siemens booth were the company’s two new solutions for high-volume hematology testing, the Atellica HEMA 570 Analyzer and the Atellica HEMA 580 Analyzer. Both hematology analyzers offer integrated automation and intelligence to eliminate barriers that hamper workflow efficiency and deliver faster patient results.
Snibe (Shenzhen, China; www.snibe.com) showcased its latest products with high-performance features, including the newest member of its MAGLUMI X-series of new-generation chemiluminescence immunoassay (CLIA) analyzers. MAGLUMI X Tech is the core technology powering the X series of fully automatic CLIA analyzers which possess powerful capabilities such as patented pipetting technology, accurate incubation technology, stable and precise measuring technology, multiple expansion solutions, and user-friendly and high-efficiency loading design. These latest high-end features help the MAGLUMI X series CLIA analyzers to provide better immunology diagnostic capabilities for laboratories and hospitals.
Mindray (Shenzhen, China; www.mindray.com) demonstrated its latest technologies and solutions in in-vitro diagnostics, including the company’s ALL IN ONE hematology solution that brings greater efficiency to hematology testing with simplified workflows and ease of operation. Mindray's ALL IN ONE Hematology Solution enables end-to-end automation throughout the process, from tube sorting through to analysis of CBC, ESR, CRP, SAA, and HbA1c with a single EDTA tube, smear preparation, and slide reading. The solution is ideal for core or centralized laboratories with mid-to-high sample volumes and high demand for turnaround times.
QuidelOrtho (San Diego, CA, USA; www.quidelortho.com) demonstrated Savanna – a true sample-to-result point-of-care multiplex molecular solution – alongside its Vitros XT 7600 Integrated System which offers a wide-ranging menu of over 160 assays. Also featured at the industry event was the Triage True High Sensitivity Troponin I Test which empowers medical professionals to make informed decisions that lead to better patient outcomes. QuidelOrtho also participated in the EuroMedLab 2023 Satellite Meeting - Point-of-Care Testing: Home, Hospital and Beyond event. The company’s Vendor Talk by Dr. Holger Gundelach, Head of EMEA POC-IA Business Unit and Clinical Value at QuidelOrtho, on "The time is now – Accelerate patient management at the point-of-care" provided valuable insights and explored the future of POC testing.
Randox (Crumlin, UK; www.randox.com) introduced the new Acusera third-party controls designed for comprehensive test menu consolidation in laboratory internal quality control. These third-party controls are "true," meaning they are not biased towards or optimized for any specific reagent, method, or instrument, and hence offer an objective assessment of performance. Also on display at the industry event was Randox's Vivalytic, an all-in-one molecular diagnostic solution. This compact benchtop platform consolidates the complete molecular workflow, including extraction, PCR amplification, and detection.
Abbott (Lake Forest, IL, USA: www.abbott.com) introduced GLP Systems, an innovative total laboratory automation (TLA) solution, offering proven technology with more flexibility and options to meet high volume needs. The GLP TLA system can markedly boost the efficacy of laboratory testing. It allows labs to concentrate on crucial tasks by maximizing uptime with infinite redundancy, streamlining workflow, and reducing turnaround time.
DiaSorin (Saluggia, Italy; www.diasorin.com) presented its industry-leading offering of fully-automated chemiluminescent immunoassay (CLIA) panels, designed specifically for diagnosing infectious diseases. Among its products showcased at the event, the highlight was the LIAISON MeMed BV test - DiaSorin's groundbreaking solution for rapidly and accurately distinguishing between bacterial and viral infections, thereby facilitating quicker, more informed decisions regarding treatment and patient management. The LIAISON MeMed BV test is the first of its kind, fully automated solution that uses data based on the host's response.
Sebia (Lisses, France; www.sebia.com) highlighted its new Alegria 2 instrument, a fully automated, sample-to-result solution. The instrument utilizes ORGENTEC’s unique and comprehensive Alegria Monotest portfolio, boasting over 100 parameters. A key distinguishing feature of the Alegria system is its flexibility to run any test from the wide ORGENTEC portfolio at any given time. Physicians can request tailor-made biomarker profiles to be analyzed for individual patients, ensuring the best possible patient care.
Binding Site (Birmingham, UK; www.bindingsite.com) showcased its Optilite system, the leading solution for multiple myeloma and immune status testing. Optilite represents a significant breakthrough in special protein testing. The innovative system has been fully optimized to simplify complex analytical processes, delivering unparalleled efficiency, workflow optimization, and confidence in test results. With an array of intelligent features, Optilite sets a new standard in the protein laboratory field by seamlessly integrating cutting-edge technology and advanced software.
As a silver sponsor of the Congress, Werfen (Barcelona, Spain; www.werfen.com) highlighted its comprehensive and integrated line of acute care diagnostics. This included the company’s blood gas family, featuring the GEM Premier and Avoximeter product lines, as well as the whole blood hemostasis family, featuring the ROTEM, Hemochron, and VerifyNow product lines. Werfen also promoted two educational sessions that took place during the event and sponsored the Satellite Meeting “Point-of-Care Testing: Home, Hospital and Beyond” - a forum for companies and industry experts to discuss topics around innovations and the future of POCT.
Menarini Diagnostics (Florence, Italy; www.menarinidiagnostics.com) introduced its brand new PRIME MDx all-in-one solution. This fully automated sample-to-result system is specifically designed to streamline laboratory workflows for real-time PCR diagnostic assays, offering exceptional efficiency, flexibility, and ease of use. Dedicated software enables automatic result generation and interpretation, leading to high-quality results, workflow standardization, and improved laboratory efficiency. The continuous workflow feature allows users to initiate a new loading process (extraction/setup) while a previous PCR is still running.
As a bronze sponsor of the Congress, Bioperfectus (Shanghai, China; www.bioperfectus.com) showcased the SAW-48 automated nucleic acid extraction workstation, an in-vitro diagnostic medical device that combines sample loading, nucleic acid purification, and PCR setup functions (PCR master mix preparation and nucleic acid pipetting) into one instrument. The SAW-48 offers the same basic functions as the SAW-96, but in a smaller and lighter form factor. Also on display at the Bioperfectus booth was the SSNP-2000B nucleic acid extraction system, a laboratory medical device that integrates state-of-the-art technologies to enable automatic nucleic acid extraction of up to 32 samples at a time.
Greiner Bio-One (Monroe, NC, USA; www.gbo.com) collaborated with Tracie Healthcare Solutions (Munich, Germany; www.tracie.health) to jointly present a complete concept of a digitalized sample collection process. Tracie, an innovative digital healthcare company, provides full support for the sample collection process to eliminate errors and ensure the best possible diagnosis for patients. Through the implementation of Tracie's software, the entire sample collection process is digitized, eliminating the need for manual steps and enabling clear patient identification. This not only enhances patient safety and simplifies day-to-day operations but also improves the overall quality of medical care.
As a bronze sponsor of the Congress, Sansure Biotech (Hunan, China; www.sansureglobal.com) demonstrated its comprehensive range of IVD solutions, including the iPonatic III portable molecular workstation which stands out for its ability to deliver extremely accurate test results within 8 to 45 minutes. Equipped with innovative one-tube fast release and rapid PCR amplification technology, it enhances point of care testing (POCT) by optimizing steps such as sample handling, nucleic acid extraction, purification, PCR amplification, and result processing. Sansure also presented its newly-launched Natch 16S nucleic acid extraction system which employs magnetic bead technology and boasts ten different mixing modes.
Diagnostica Stago (Asnières-sur-Seine, France; www.stago.com) unveiled its new coagulation line- the sthemO and sthemE product ranges. The sthemO 301, the first analyzer in the sthemO hemostasis series, is a high-throughput, fully automated coagulation analyzer designed for medium to large laboratories and can be integrated into automated tracks. Also included in the new range of sthemO analyzers by Stago is the sthemO 201. This bench-top analyzer, suitable for smaller laboratories, offers the same level of efficiency and analytical performance as the sthemO 301. In order to optimize these two novel systems, Stago has also launched new eSolutions under the sthemE umbrella. The sthemE Manager is designed to facilitate data and information exchange between one or several in vitro diagnostic analyzers and laboratory information systems.
Beckman Coulter (Brea, CA, USA; www.beckmancoulter.com) unveiled DxI 9000 Access, its brand-new high throughput immunoassay analyzer that is designed for superior laboratory performance and demands no daily maintenance. The DxI 9000 is the most efficient immunoassay analyzer in terms of footprint, boasting the ability to process up to 215 tests per hour per square meter (tests/hr/m2). Its unique ZeroDaily Maintenance feature further enhances the device's uptime performance, as confirmed by beta users who report that it redefines their workday by eliminating daily maintenance tasks and reducing yearly maintenance routines by up to a staggering 96%.
Thermo Fisher Scientific (Waltham, MA, USA; www.thermofisher.com) demonstrated the Thermo Scientific B·R·A·H·M·S KRYPTOR compact PLUS fully automated, random-access benchtop immunoanalyzer that delivers remarkable precision, thereby improving patient outcomes. Thermo Fisher also displayed the Thermo Scientific Indiko, a fully automated benchtop analyzer for clinical chemistry and specialty testing. This device offers genuine walk-away time for the operator once it's loaded. Also on display was the Thermo Scientific TSQ Altis triple quadrupole mass spectrometer, which delivers unparalleled accuracy and precision for low-level compound detection and quantitation in complex matrices.
LumiraDx (London, UK; www.lumiradx.com) presented its LumiraDx Platform which is designed to perform multiple tests on one single instrument wherever needed. The LumiraDx Platform is similar to a portable laboratory, compact yet capable of delivering lab-grade results in a variety of care settings, thereby bringing clinical decision-making closer to the patient. This rapid, high-performing, POC diagnostic solution employs active microfluidic technology, providing results for a range of analytes, including NT-proBNP, HbA1c, and CRP, within minutes.
LGC Clinical Diagnostics (Middlesex, UK; www.lgcgroup.com) demonstrated how automated processes with its Multichem independent QC and IAMQC QC data management software can enhance QC efficiencies in the laboratory. LGC’s brands include three IVD manufacturers of QMTs (Maine Standards Company, SeraCare and Technopath Clinical Diagnostics) and one manufacturer of viral and bacterial antigens and antibodies (The Native Antigen Company). LGC showcased various product offerings, including Multichem Independent Quality Control, VALIDATE calibration and verification materials, as well as its leading serology portfolio – ACCURUN, alongside its Clinical Genomics portfolio including Seraseq for NGS.
Diesse Diagnostica Senese (Rigoni, Italy; www.diesse.it) unveiled its new innovative immunochemistry analyzer, the CHORUS EVO which has been designed in line with the company’s aim of developing instruments featuring cutting-edge technology that meet Italian design and its vision of "Diagnostics Evolution". CHORUS EVO is a new-generation analytical instrument that integrates innovative digital technology, paving the way for the integration of artificial intelligence functions. In order to achieve this, DIESSE is collaborating with the SAIHUB consortium, a network of companies that specialize in applying artificial intelligence in life sciences.
The record number of scientific abstracts (over 2100) submitted by scientists and laboratory professionals from across the world for presentation, and a record number of corporate members and non-members participating in the industry exhibition made Roma 2023 the best attended and most successful EuroMedLab event ever held. The 26th WorldLab will be convening in Dubai, UAE, on May 26-30, 2024. The 26th EuroMedLab is scheduled for May 18-22, 2025, in Brussels, Belgium.
Related Links:
Latest Industry News
- AI-Powered Cervical Cancer Test Set for Major Rollout in Latin America
- New Collaboration Brings Automated Mass Spectrometry to Routine Laboratory Testing
- Diasorin and Fisher Scientific Enter into US Distribution Agreement for Molecular POC Platform
- WHX Labs Dubai to Gather Global Experts in Antimicrobial Resistance at Inaugural AMR Leaders’ Summit
- BD and Penn Institute Collaborate to Advance Immunotherapy through Flow Cytometry
- Abbott Acquires Cancer-Screening Company Exact Sciences
- Roche and Freenome Collaborate to Develop Cancer Screening Tests
- Co-Diagnostics Forms New Business Unit to Develop AI-Powered Diagnostics
- Qiagen Acquires Single-Cell Omics Firm Parse Biosciences
- Puritan Medical Products Showcasing Innovation at AMP2025 in Boston
- Advanced Instruments Merged Under Nova Biomedical Name
- Bio-Rad and Biodesix Partner to Develop Droplet Digital PCR High Complexity Assays
- Hologic to be Acquired by Blackstone and TPG
- Bio-Techne and Oxford Nanopore to Accelerate Development of Genetics Portfolio
- Terumo BCT and Hemex Health Collaborate to Improve Access to Testing for Hemoglobin Disorders
- Revvity and Sanofi Collaborate on Program to Revolutionize Early Detection of Type 1 Diabetes
Channels
Clinical Chemistry
view channel
New PSA-Based Prognostic Model Improves Prostate Cancer Risk Assessment
Prostate cancer is the second-leading cause of cancer death among American men, and about one in eight will be diagnosed in their lifetime. Screening relies on blood levels of prostate-specific antigen... Read more
Extracellular Vesicles Linked to Heart Failure Risk in CKD Patients
Chronic kidney disease (CKD) affects more than 1 in 7 Americans and is strongly associated with cardiovascular complications, which account for more than half of deaths among people with CKD.... Read moreMolecular Diagnostics
view channel
Diagnostic Device Predicts Treatment Response for Brain Tumors Via Blood Test
Glioblastoma is one of the deadliest forms of brain cancer, largely because doctors have no reliable way to determine whether treatments are working in real time. Assessing therapeutic response currently... Read more
Blood Test Detects Early-Stage Cancers by Measuring Epigenetic Instability
Early-stage cancers are notoriously difficult to detect because molecular changes are subtle and often missed by existing screening tools. Many liquid biopsies rely on measuring absolute DNA methylation... Read more
“Lab-On-A-Disc” Device Paves Way for More Automated Liquid Biopsies
Extracellular vesicles (EVs) are tiny particles released by cells into the bloodstream that carry molecular information about a cell’s condition, including whether it is cancerous. However, EVs are highly... Read more
Blood Test Identifies Inflammatory Breast Cancer Patients at Increased Risk of Brain Metastasis
Brain metastasis is a frequent and devastating complication in patients with inflammatory breast cancer, an aggressive subtype with limited treatment options. Despite its high incidence, the biological... Read moreHematology
view channel
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read more
Fast and Easy Test Could Revolutionize Blood Transfusions
Blood transfusions are a cornerstone of modern medicine, yet red blood cells can deteriorate quietly while sitting in cold storage for weeks. Although blood units have a fixed expiration date, cells from... Read more
Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
High-volume hemostasis sections must sustain rapid turnaround while managing reruns and reflex testing. Manual tube handling and preanalytical checks can strain staff time and increase opportunities for error.... Read more
High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
Blood clotting is essential for preventing bleeding, but even small imbalances can lead to serious conditions such as thrombosis or dangerous hemorrhage. In cardiovascular disease, clinicians often struggle... Read moreImmunology
view channelBlood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
Small cell lung cancer (SCLC) is an aggressive disease with limited treatment options, and even newly approved immunotherapies do not benefit all patients. While immunotherapy can extend survival for some,... Read more
Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
Targeted cancer therapies such as PARP inhibitors can be highly effective, but only for patients whose tumors carry specific DNA repair defects. Identifying these patients accurately remains challenging,... Read more
Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
Immunotherapy has transformed cancer treatment, but only a small proportion of patients experience lasting benefit, with response rates often remaining between 10% and 20%. Clinicians currently lack reliable... Read moreMicrobiology
view channel
Comprehensive Review Identifies Gut Microbiome Signatures Associated With Alzheimer’s Disease
Alzheimer’s disease affects approximately 6.7 million people in the United States and nearly 50 million worldwide, yet early cognitive decline remains difficult to characterize. Increasing evidence suggests... Read moreAI-Powered Platform Enables Rapid Detection of Drug-Resistant C. Auris Pathogens
Infections caused by the pathogenic yeast Candida auris pose a significant threat to hospitalized patients, particularly those with weakened immune systems or those who have invasive medical devices.... Read morePathology
view channel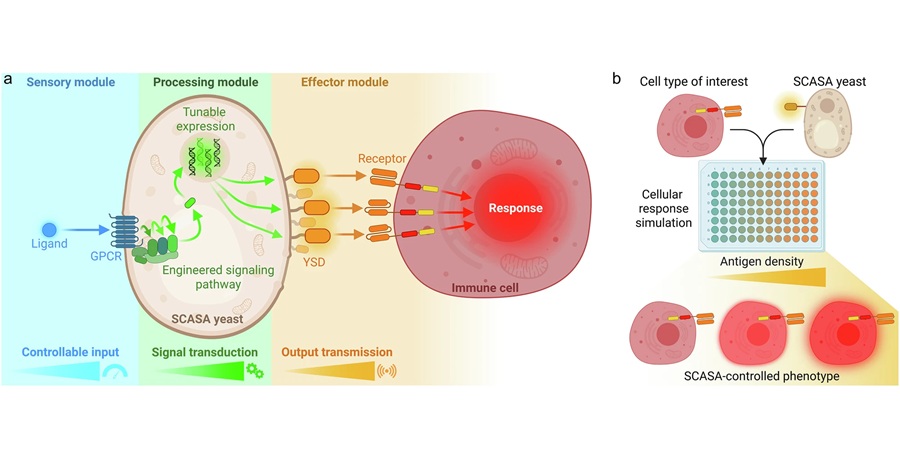
Engineered Yeast Cells Enable Rapid Testing of Cancer Immunotherapy
Developing new cancer immunotherapies is a slow, costly, and high-risk process, particularly for CAR T cell treatments that must precisely recognize cancer-specific antigens. Small differences in tumor... Read more
First-Of-Its-Kind Test Identifies Autism Risk at Birth
Autism spectrum disorder is treatable, and extensive research shows that early intervention can significantly improve cognitive, social, and behavioral outcomes. Yet in the United States, the average age... Read moreTechnology
view channel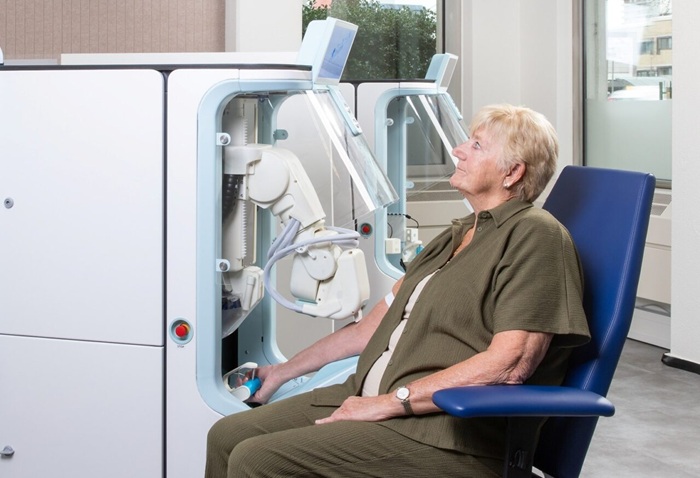
Robotic Technology Unveiled for Automated Diagnostic Blood Draws
Routine diagnostic blood collection is a high‑volume task that can strain staffing and introduce human‑dependent variability, with downstream implications for sample quality and patient experience.... Read more
ADLM Launches First-of-Its-Kind Data Science Program for Laboratory Medicine Professionals
Clinical laboratories generate billions of test results each year, creating a treasure trove of data with the potential to support more personalized testing, improve operational efficiency, and enhance patient care.... Read moreAptamer Biosensor Technology to Transform Virus Detection
Rapid and reliable virus detection is essential for controlling outbreaks, from seasonal influenza to global pandemics such as COVID-19. Conventional diagnostic methods, including cell culture, antigen... Read more