Point-of-Care Testing Evaluated for Early Coagulopathy Diagnosis
|
By LabMedica International staff writers Posted on 09 Jun 2021 |
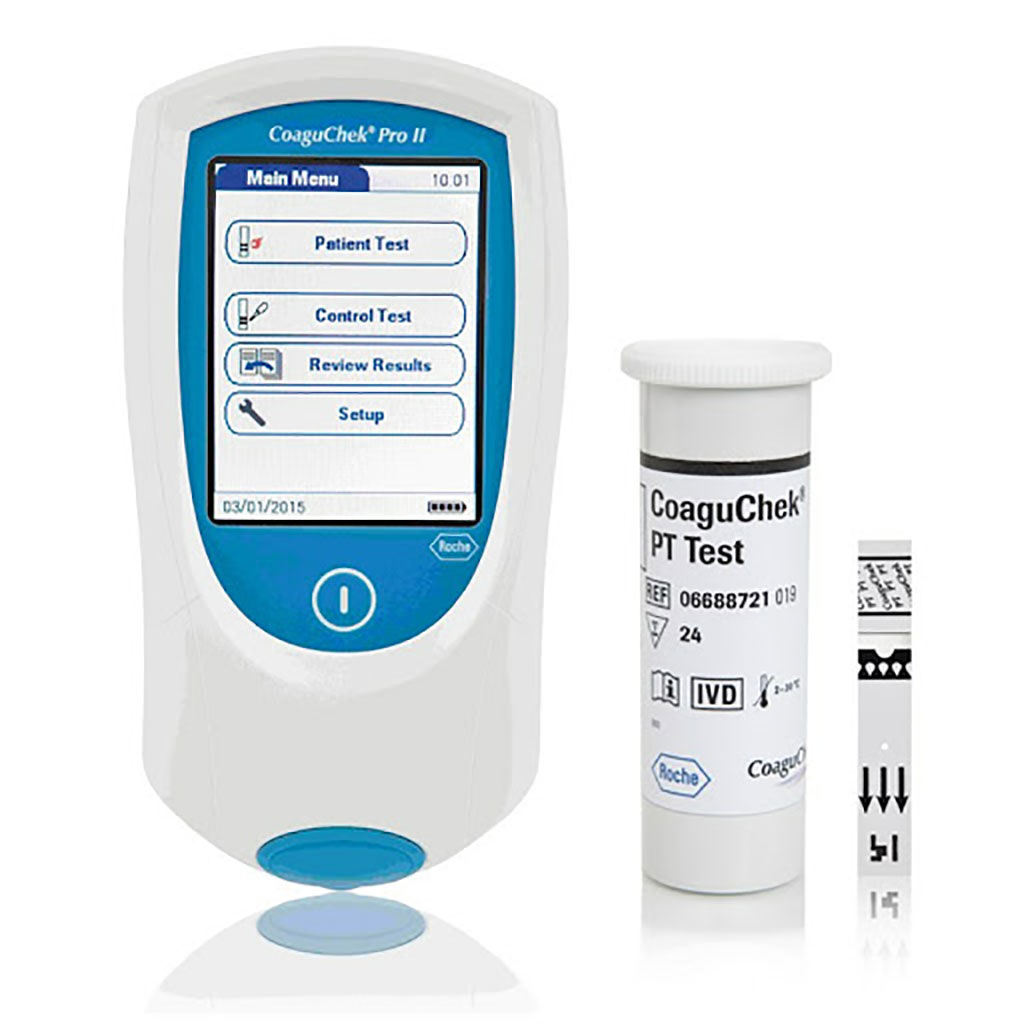
Image: The CoaguChek Pro II Coagulation Meter and CoaguChek PT Test Strips (Photo courtesy of Roche Diagnostics)
Hemorrhage and coagulopathy are particularly relevant complications and are often connected to injuries, surgical interventions, serious diseases, anticoagulant medication or pregnancy/delivery. Acute bleeding requires fast and targeted therapy and therefore, knowledge of the patient's potential to form a clot is crucial.
Treating bleeding complications, rapid hemostatic optimization (interventional, surgical, mechanical or medication) and targeted therapy are therapeutic goals and are therefore equivalent to those of coagulopathy treatment. The earlier a coagulopathy is diagnosed, the earlier a targeted therapy can be initiated. The international normalized ratio (INR) value is of particular importance for the diagnosis and therapy of coagulopathy.
Intensive Care Physicians at the University Hospital Frankfurt (Frankfurt, Germany) and their colleagues included in a study, one group that included hemorrhagic patients from the department of obstetrics (obstetric group = OG), and another group comprising patients admitted to the emergency department (emergency group = EG). The primary outcome measure was the difference between two INR results of one blood sample determined by point-of-care testing (POCT) and standard laboratory testing (SL).
After blood sampling, a drop of blood was applied immediately to the test stripe of the POCT device, CoaguChek Pro II (Roche Diagnostics GmbH, Mannheim, Germany). The CoaguChek Pro II is a portable device requiring an 8-μl sample volume. It measures the international normalized ratio (INR) based on an electrochemical reaction. Samples for SL analysis were sent to the central laboratory via a pneumatic dispatch system (EG) or personal-based transport (OG) for automated analysis with an ACL TOP 700 Hemostasis testing System, (Werfen GmbH, Munich, Germany) in combination with PT Re Combiplastin 2G reagents.
The team reported that INR results between POCT and SLA showed a high and significant correlation. POCT results were reported significantly more quickly (EG: 1.1 versus 39.6 minutes; OG: 2.0 versus 75 minutes) and required less time for analysis (EG: 0.3 versus 24.0 minutes; OG: 0.5 versus 45.0 minutes) compared to SLA. The time for transportation with the pneumatic tube was significantly shorter (8.0 versus 18.5 minutes) than with the personal-based transport system. The estimated blood loss was between 800 mL and 1300 mL.
The authors concluded that strip-based test systems may be suitable methods for the emergency diagnosis of hemorrhagic patients because their measurement results are available significantly more quickly and seem to support the use of point-of-care INR-devices to rule out pathological INR-values in bleeding patients. The test strip-based methods can be used as diagnostic elements in hemotherapy algorithms to implement fast and targeted hemotherapy that can positively impact the clinical outcomes of patients. The study was published on May 23, 2021 in the journal Practical Laboratory Medicine.
Related Links:
University Hospital Frankfurt
Roche Diagnostics
Werfen
Treating bleeding complications, rapid hemostatic optimization (interventional, surgical, mechanical or medication) and targeted therapy are therapeutic goals and are therefore equivalent to those of coagulopathy treatment. The earlier a coagulopathy is diagnosed, the earlier a targeted therapy can be initiated. The international normalized ratio (INR) value is of particular importance for the diagnosis and therapy of coagulopathy.
Intensive Care Physicians at the University Hospital Frankfurt (Frankfurt, Germany) and their colleagues included in a study, one group that included hemorrhagic patients from the department of obstetrics (obstetric group = OG), and another group comprising patients admitted to the emergency department (emergency group = EG). The primary outcome measure was the difference between two INR results of one blood sample determined by point-of-care testing (POCT) and standard laboratory testing (SL).
After blood sampling, a drop of blood was applied immediately to the test stripe of the POCT device, CoaguChek Pro II (Roche Diagnostics GmbH, Mannheim, Germany). The CoaguChek Pro II is a portable device requiring an 8-μl sample volume. It measures the international normalized ratio (INR) based on an electrochemical reaction. Samples for SL analysis were sent to the central laboratory via a pneumatic dispatch system (EG) or personal-based transport (OG) for automated analysis with an ACL TOP 700 Hemostasis testing System, (Werfen GmbH, Munich, Germany) in combination with PT Re Combiplastin 2G reagents.
The team reported that INR results between POCT and SLA showed a high and significant correlation. POCT results were reported significantly more quickly (EG: 1.1 versus 39.6 minutes; OG: 2.0 versus 75 minutes) and required less time for analysis (EG: 0.3 versus 24.0 minutes; OG: 0.5 versus 45.0 minutes) compared to SLA. The time for transportation with the pneumatic tube was significantly shorter (8.0 versus 18.5 minutes) than with the personal-based transport system. The estimated blood loss was between 800 mL and 1300 mL.
The authors concluded that strip-based test systems may be suitable methods for the emergency diagnosis of hemorrhagic patients because their measurement results are available significantly more quickly and seem to support the use of point-of-care INR-devices to rule out pathological INR-values in bleeding patients. The test strip-based methods can be used as diagnostic elements in hemotherapy algorithms to implement fast and targeted hemotherapy that can positively impact the clinical outcomes of patients. The study was published on May 23, 2021 in the journal Practical Laboratory Medicine.
Related Links:
University Hospital Frankfurt
Roche Diagnostics
Werfen
Latest Hematology News
- Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
- Advanced CBC-Derived Indices Integrated into Hematology Platforms
- Blood Test Enables Early Detection of Multiple Myeloma Relapse
- Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
- Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
- Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
- Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
- New Platelet Function Assay Enables Monitoring of Antiplatelet Therapy
- Open Multi-Omics Platform Identifies Prognostic Subtypes in Blood Cancers
- AI-Powered Digital Workflow Standardizes Bone Marrow Aspirate Morphology
- Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- Fast and Easy Test Could Revolutionize Blood Transfusions
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
Channels
Clinical Chemistry
view channel
FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
Accurate evaluation of androgen status often requires concordant measurement of total testosterone, free testosterone, and sex hormone‑binding globulin. Reference methods such as equilibrium dialysis with... Read more
CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
Specialized care settings frequently evaluate patients aged 50 years and older who present with signs and symptoms of cognitive decline to determine whether amyloid pathology linked to Alzheimer’s disease... Read moreMolecular Diagnostics
view channel
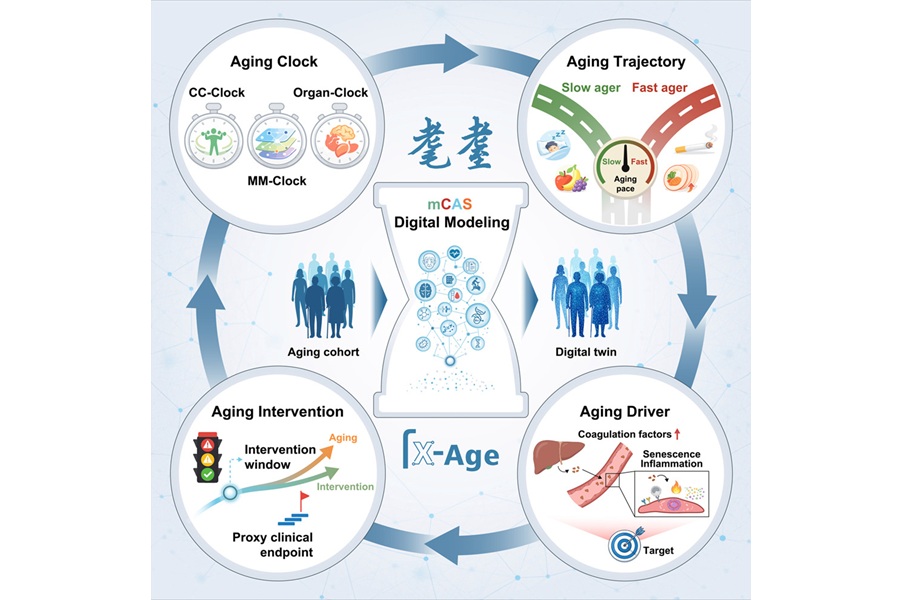
Blood-Based “Ageing Clock” Helps Predict Dementia Risk and Earlier Onset
Dementia imposes a growing health burden, affecting an estimated 982,000 people in the UK, with cases projected to reach 1.4 million by 2040. Earlier identification of those most likely to develop disease... Read more
Blood Test Refines Biopsy Decisions in Prostate Cancer
Prostate biopsy decisions remain challenging, with many men undergoing invasive procedures that ultimately yield negative results. In the U.S., more than a million prostate biopsies are performed each... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read moreMicrobiology
view channel
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read more
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more