Minimal Residual Disease Assessed in AL Amyloidosis Patients
|
By LabMedica International staff writers Posted on 17 Mar 2020 |

Image: The Gallios flow cytometer provides efficient acquisition of superior quality data from up to 10 colors with advanced optical design for enhanced sensitivity for multicolor assays (Photo courtesy of Beckman Coulter).
Amyloid light-chain (AL) amyloidosis, also known as primary amyloidosis, is the most common form of systemic amyloidosis in the USA. Despite achieving a hematologic complete response after treatment, many patients with AL amyloidosis do not attain recovery of organ function and/or experience hematologic relapse.
A persistent plasma cell clone producing amyloidogenic light chains at levels below the detection threshold of traditional serologic methods is hypothesized to impede organ response in some patients. Assessment of minimal residual disease (MRD) may therefore have clinical importance as a more stringent treatment response tool for patients in a hematologic complete response.
Hematologists at the Boston University School of Medicine (Boston, MA, USA) evaluated a total of 86 patients with AL amyloidosis with hematologic complete response (hemCR) achievement on previous evaluation were tested for MRD. Fresh bone marrow aspirate samples from consenting patients with AL amyloidosis were analyzed between February and November 2019. The scientists used 2-tube, 10-color antibody combination multiparametric flow cytometry to assess for MRD at a minimum sensitivity of 1 in 105 nucleated cells. Specimen processing was performed at PhenoPath Laboratories (Seattle, WA, USA) with a target of two million live cellular events acquired on a Gallios flow cytometer (Beckman Coulter, Brea, CA, USA).
The scientists reported that of 65 patients in hematologic complete response, 36 (55%) were found to have a residual clonal plasma cell population in the bone marrow. Comparing the MRD-negative and MRD-positive groups, renal response was observed in 88% versus 64%, cardiac response in 75% versus 59%, and any organ response in 90% versus 75% of patients. Depth of organ response as measured by the percent decrease in 24-hour proteinuria and brain natriuretic peptide was 96% versus 91% and 55% versus 46%, respectively. Notably, the median difference between involved and uninvolved serum free immunoglobulin light chain (dFLC) at diagnosis was significantly higher in the MRD-negative cohort (129 mg/L versus 70 mg/L).
The authors concluded that MRD negativity as a deeper treatment response classification was correlated with higher organ response in patients with AL amyloidosis in hemCR, although differences were not statistically significant. The study was published on March 4, 2020 in the journal Blood Advances.
Related Links:
Boston University School of Medicine
PhenoPath Laboratories
Beckman Coulter
A persistent plasma cell clone producing amyloidogenic light chains at levels below the detection threshold of traditional serologic methods is hypothesized to impede organ response in some patients. Assessment of minimal residual disease (MRD) may therefore have clinical importance as a more stringent treatment response tool for patients in a hematologic complete response.
Hematologists at the Boston University School of Medicine (Boston, MA, USA) evaluated a total of 86 patients with AL amyloidosis with hematologic complete response (hemCR) achievement on previous evaluation were tested for MRD. Fresh bone marrow aspirate samples from consenting patients with AL amyloidosis were analyzed between February and November 2019. The scientists used 2-tube, 10-color antibody combination multiparametric flow cytometry to assess for MRD at a minimum sensitivity of 1 in 105 nucleated cells. Specimen processing was performed at PhenoPath Laboratories (Seattle, WA, USA) with a target of two million live cellular events acquired on a Gallios flow cytometer (Beckman Coulter, Brea, CA, USA).
The scientists reported that of 65 patients in hematologic complete response, 36 (55%) were found to have a residual clonal plasma cell population in the bone marrow. Comparing the MRD-negative and MRD-positive groups, renal response was observed in 88% versus 64%, cardiac response in 75% versus 59%, and any organ response in 90% versus 75% of patients. Depth of organ response as measured by the percent decrease in 24-hour proteinuria and brain natriuretic peptide was 96% versus 91% and 55% versus 46%, respectively. Notably, the median difference between involved and uninvolved serum free immunoglobulin light chain (dFLC) at diagnosis was significantly higher in the MRD-negative cohort (129 mg/L versus 70 mg/L).
The authors concluded that MRD negativity as a deeper treatment response classification was correlated with higher organ response in patients with AL amyloidosis in hemCR, although differences were not statistically significant. The study was published on March 4, 2020 in the journal Blood Advances.
Related Links:
Boston University School of Medicine
PhenoPath Laboratories
Beckman Coulter
Latest Hematology News
- Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- Fast and Easy Test Could Revolutionize Blood Transfusions
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
- MRD Tests Could Predict Survival in Leukemia Patients
- Platelet Activity Blood Test in Middle Age Could Identify Early Alzheimer’s Risk
- Microvesicles Measurement Could Detect Vascular Injury in Sickle Cell Disease Patients
- ADLM’s New Coagulation Testing Guidance to Improve Care for Patients on Blood Thinners
- Viscoelastic Testing Could Improve Treatment of Maternal Hemorrhage
- Pioneering Model Measures Radiation Exposure in Blood for Precise Cancer Treatments
- Platelets Could Improve Early and Minimally Invasive Detection of Cancer
- Portable and Disposable Device Obtains Platelet-Rich Plasma Without Complex Equipment
- Disposable Cartridge-Based Test Delivers Rapid and Accurate CBC Results
- First Point-of-Care Heparin Monitoring Test Provides Results in Under 15 Minutes

Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreMolecular Diagnostics
view channel
New Molecular Test Boosts Accuracy of Bile Duct Cancer Diagnosis
Bile duct strictures can arise from cancer or benign disease, but their location within ducts connecting the liver, gallbladder, and intestines complicates evaluation. Standard biopsy and cytology may... Read moreAdaptive PCR Platform Improves Consistency in Small-Batch NGS Workflows
PCR amplification during next-generation sequencing (NGS) library preparation can introduce variability, often requiring manual quantification and risking over-cycling artifacts. The issue is especially... Read more
First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
Accurate identification of clonal immunoglobulin heavy chain (IGH) gene rearrangements is central to evaluating suspected B-cell lymphoproliferative disorders, where a single B-cell clone yields a defining... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
Rapid Urine Test Speeds Antibiotic Selection for UTIs
Urinary tract infections are a common reason for antibiotic prescribing and have led to more than 800,000 hospital admissions across England in the past five years, according to National Health Service data.... Read more
WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Tuberculosis (TB) remains a leading infectious killer, with more than 3,300 deaths and 29,000 new illnesses every day. Diagnostic delays and dependence on centralized laboratory networks continue to impede... Read morePathology
view channel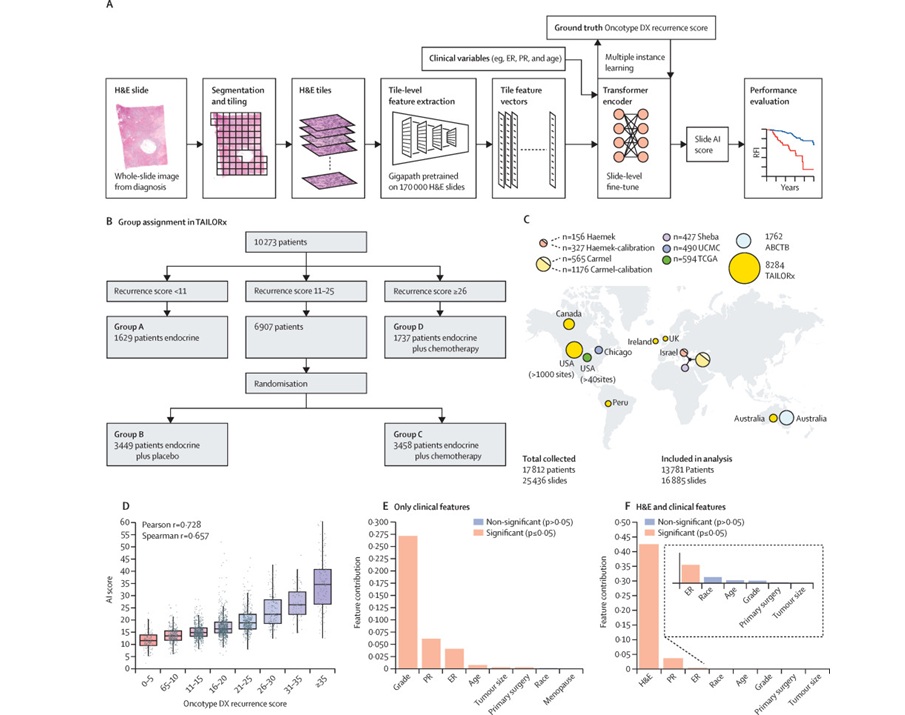
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read more
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
Lunit (Seoul, South Korea), a leading provider of AI for cancer diagnostics and precision oncology, and CellCarta (Montreal, QC, Canada), a global contract research organization (CRO) laboratory serving... Read more