Laser-Heated Plasmonic Nanoparticles Safely Cause Localized Tumor Destruction
|
By LabMedica International staff writers Posted on 16 Aug 2016 |
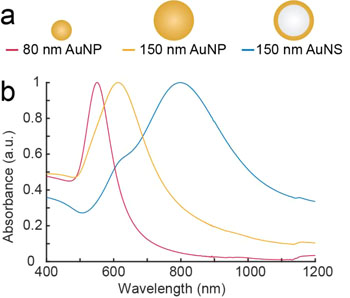
Image: The experiments were carried out with nanoparticles of different sizes and structures. The first two of the series consisted of solid gold and the last consisted of a core of glass with a surface of gold. The beads were illuminated with near infrared light with wavelengths of 807 nanometers and 1064 nanometers. The most effective nanoparticle was the gold-plated glass bead (Photo courtesy of Kamilla Nørregaard, University of Copenhagen).
Cancer therapy based on plasmonic nanoparticles that homed to tumors, warmed on exposure to laser irradiation, and then selectively killed the cancer cells was optimized and shown to be effective in a mouse xenograft model.
Plasmonic nanoparticle-based photothermal cancer therapy is a promising new tool to inflict localized and irreversible damage to tumor tissue by heating, without harming surrounding healthy tissue.
Plasmonic resonance is a phenomenon that occurs when light is reflected off thin metal films, which may be used to measure interaction of biomolecules on the surface. An electron charge density wave arises at the surface of the film when light is reflected at the film under specific conditions. A fraction of the light energy incident at a defined angle can interact with the delocalized electrons in the metal film (plasmon) thus reducing the reflected light intensity. The angle of incidence at which this occurs is influenced by the refractive index close to the backside of the metal film, to which target molecules are immobilized. If ligands in a mobile phase running along a flow cell bind to the surface molecules, the local refractive index changes in proportion to the mass being immobilized. This can be monitored in real time by detecting changes in the intensity of the reflected light.
Investigators at the University of Copenhagen (Denmark) combined plasmonic resonance with nanoparticles made from gold or gold-covered glass as the basis for hyperthermic anticancer therapy. The effectiveness of the approach was assessed by PET (positron emission tomography).
The investigators described in the August 2, 2016, online edition of the journal Scientific Reports the quantification of heat generation and absorption cross-section of single irradiated nanoparticles using a temperature sensitive lipid-based assay that compared results to their theoretically predicted photo-absorption.
In vivo, the heat generation of irradiated nanoparticles was evaluated in human tumor xenografts in mice using 2-deoxy-2-[F-18]fluoro-D-glucose (18F-FDG) PET imaging. To validate the use of this platform, the investigators quantified the photothermal efficiency of near infrared resonant silica-gold nanoshells (AuNSs) and benchmarked this against the heating of colloidal spherical, solid gold nanoparticles (AuNPs). As expected, both in vitro and in vivo the heat generation of the resonant AuNSs performed better than the non-resonant AuNPs. Furthermore, the results showed that PET imaging could be reliably used to monitor early treatment response of photothermal treatment.
In the mouse study, 150 nanometer gold-covered glass nanoparticles were injected into tumors and then illuminated with near-infrared laser light, which penetrated through the tissue. In contrast to conventional radiation therapy, the near-infrared laser light caused no burn damage to the tissue that it passed through. Within an hour following the treatment, the investigators could detect cancer cell death on PET scans and determined that the effect continued for at least two days.
"The treatment involves injecting tiny nanoparticles directly into the cancer. Then you heat up the nanoparticles from outside using lasers. It is a strong interaction between the nanoparticles and the laser light, which causes the particles to heat up. What then happens is that the heated particles damage or kill the cancer cells," said senior author Dr. Lene Oddershede, professor of biophysics at the University of Copenhagen. "As physicists we have great expertise in the interaction between light and nanoparticles and we can very accurately measure the temperature of the heated nanoparticles. The effectiveness depends on the right combination between the structure and material of the particles, their physical size, and the wavelength of the light."
"Now we have proven that the method works. In the longer term, we would like the method to work by injecting the nanoparticles into the bloodstream, where they end up in the tumors that may have metastasized. With the PET scans we can see where the tumors are and irradiate them with lasers, while also effectively assessing how well the treatment has worked shortly after the irradiation. In addition, we will coat the particles with chemotherapy, which is released by the heat and which will also help kill the cancer cells," said Dr. Oddershede.
Related Links:
University of Copenhagen
Plasmonic nanoparticle-based photothermal cancer therapy is a promising new tool to inflict localized and irreversible damage to tumor tissue by heating, without harming surrounding healthy tissue.
Plasmonic resonance is a phenomenon that occurs when light is reflected off thin metal films, which may be used to measure interaction of biomolecules on the surface. An electron charge density wave arises at the surface of the film when light is reflected at the film under specific conditions. A fraction of the light energy incident at a defined angle can interact with the delocalized electrons in the metal film (plasmon) thus reducing the reflected light intensity. The angle of incidence at which this occurs is influenced by the refractive index close to the backside of the metal film, to which target molecules are immobilized. If ligands in a mobile phase running along a flow cell bind to the surface molecules, the local refractive index changes in proportion to the mass being immobilized. This can be monitored in real time by detecting changes in the intensity of the reflected light.
Investigators at the University of Copenhagen (Denmark) combined plasmonic resonance with nanoparticles made from gold or gold-covered glass as the basis for hyperthermic anticancer therapy. The effectiveness of the approach was assessed by PET (positron emission tomography).
The investigators described in the August 2, 2016, online edition of the journal Scientific Reports the quantification of heat generation and absorption cross-section of single irradiated nanoparticles using a temperature sensitive lipid-based assay that compared results to their theoretically predicted photo-absorption.
In vivo, the heat generation of irradiated nanoparticles was evaluated in human tumor xenografts in mice using 2-deoxy-2-[F-18]fluoro-D-glucose (18F-FDG) PET imaging. To validate the use of this platform, the investigators quantified the photothermal efficiency of near infrared resonant silica-gold nanoshells (AuNSs) and benchmarked this against the heating of colloidal spherical, solid gold nanoparticles (AuNPs). As expected, both in vitro and in vivo the heat generation of the resonant AuNSs performed better than the non-resonant AuNPs. Furthermore, the results showed that PET imaging could be reliably used to monitor early treatment response of photothermal treatment.
In the mouse study, 150 nanometer gold-covered glass nanoparticles were injected into tumors and then illuminated with near-infrared laser light, which penetrated through the tissue. In contrast to conventional radiation therapy, the near-infrared laser light caused no burn damage to the tissue that it passed through. Within an hour following the treatment, the investigators could detect cancer cell death on PET scans and determined that the effect continued for at least two days.
"The treatment involves injecting tiny nanoparticles directly into the cancer. Then you heat up the nanoparticles from outside using lasers. It is a strong interaction between the nanoparticles and the laser light, which causes the particles to heat up. What then happens is that the heated particles damage or kill the cancer cells," said senior author Dr. Lene Oddershede, professor of biophysics at the University of Copenhagen. "As physicists we have great expertise in the interaction between light and nanoparticles and we can very accurately measure the temperature of the heated nanoparticles. The effectiveness depends on the right combination between the structure and material of the particles, their physical size, and the wavelength of the light."
"Now we have proven that the method works. In the longer term, we would like the method to work by injecting the nanoparticles into the bloodstream, where they end up in the tumors that may have metastasized. With the PET scans we can see where the tumors are and irradiate them with lasers, while also effectively assessing how well the treatment has worked shortly after the irradiation. In addition, we will coat the particles with chemotherapy, which is released by the heat and which will also help kill the cancer cells," said Dr. Oddershede.
Related Links:
University of Copenhagen
Latest BioResearch News
- Hidden 'Jumping Gene' Variant Linked to Higher Pancreatic Cancer Risk
- Common White Blood Cells Produce Schizophrenia-Linked Protein
- Nanopore Method Captures RNA Folding at Single-Molecule Resolution
- Tumor Microenvironment Marker Linked to Worse Survival in Solid Tumors
- Hidden Immune Gene Defect May Explain Kaposi Sarcoma Susceptibility
- Genetic Markers May Help Predict Amputation Risk in Peripheral Artery Disease
- Gene Signature Shows Promise for Depression Biomarker Testing
- AI-Driven Tumor Profiling Initiative Targets Precision Therapy Development
- Researchers Map Protein and Glycosylation Across 15 Human Body Fluids
- Telomere Length Abnormalities Linked to Lymphoma Development
- Biomarker Signals Chemotherapy Resistance in Relapsed Small Cell Lung Cancer
- Inflammatory Gene Signature Links Metabolic Disease to Pancreatic Cancer Recurrence
- Study Links Abnormal Gene Splicing to Treatment Response in Metastatic Kidney Cancer
- Research Reveals How Some Aplastic Anemia Patients Recover Bone Marrow Function
- New Molecular Insights Support Diagnosis of Hodgkin Lymphoma
- Epigenetic Signals and Blood Markers Aid Chronic Fatigue Syndrome Diagnosis
Channels
Clinical Chemistry
view channel
International Experts Recommend Ending Routine 'Corrected' Calcium Reporting
Interpreting serum calcium can be clinically challenging when albumin levels vary, especially in patients with chronic illness or kidney disease. For decades, laboratories have used formulas to adjust... Read more
Long-Term Data Show PSA Screening Modestly Reduces Prostate Cancer Deaths
Prostate cancer is among the most common cancers in men, and the role of population screening has remained controversial because of overdiagnosis and overtreatment. Health systems have sought clearer,... Read moreMolecular Diagnostics
view channel
Microbial Saliva Test Could Help Triage Esophageal Cancer Risk
Esophageal squamous cell carcinoma (ESCC) is highly lethal, partly because many patients are diagnosed only after swallowing becomes difficult and treatment options are largely palliative.... Read more
Expanded DPYD Genotyping Test Supports Safer Chemotherapy Dosing
Fluoropyrimidines such as 5-fluorouracil (5-FU) are chemotherapy drugs prescribed to more than two million cancer patients each year, but 10–20% of patients can experience severe, and sometimes fatal,... Read more
Multi-Omics Profiling Helps Predict BCG Response and Recurrence in Bladder Cancer
High-risk non–muscle-invasive bladder cancer frequently recurs after therapy, with about 30% of patients relapsing and roughly 10% dying within two years despite tumor resection, surveillance, and Bacillus... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read more
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read morePathology
view channel
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read more
AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
Non–muscle invasive bladder cancer has highly variable outcomes, complicating surveillance and treatment planning. Risk assessment typically relies on stage, grade, and tumor size, leaving uncertainty... Read moreTechnology
view channel
AI-Enabled Assistant Unifies Molecular Workflow Planning and Support
Clinical laboratories and research groups face increasingly complex molecular workflows and expanding technical documentation spread across multiple systems. Fragmented digital tools can slow experiment... Read more
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read moreIndustry
view channel
Strategic Collaboration Advances RNA Foundation Models for Precision Oncology
Bulk RNA sequencing is increasingly used to study tumor biology, but standard analyses often reduce results to gene-level summaries that miss important transcript variants and mutation patterns.... Read more