Whole Blood Controls Monitor Immunosuppressant Drug Test Precision
|
By LabMedica International staff writers Posted on 29 Oct 2012 |
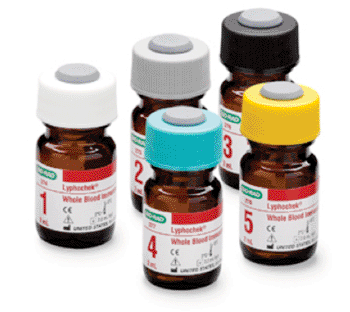
Image: Five levels of Lyphocheck Whole Blood Immunosuppressant Control (Photo courtesy of Bio-Rad).
A lyophilized, third party, human whole blood based control offers five individual levels and includes Cyclosporine, Tacrolimus, and Sirolimus.
The control can be used with immunoassay or chromatographic techniques. Called the Lyphochek Whole Blood Immunosuppressant Control, it was designed for use as an assayed quality control material to monitor the precision of immunosuppressant drug test procedures in the clinical laboratory from low to very high concentrations. Immunosuppressant drugs are typically used in transplant therapy to help minimize organ rejection. Lyphochek Control is supplied as a whole blood based material that can be processed in the same manner as patient samples.
Bio-Rad Laboratories, Inc. (Hercules, CA, USA), a manufacturer and distributor of life science clinical diagnostics and research products, released the Lyphochek Whole Blood Immunosuppressant Control.
The company offers technical support and troubleshooting assistance for the control. The Unity Interlaboratory Program and the Unity Real Time, a data management-software solution designed to improve the effectiveness of quality control processes, are available for use with the Lyphochek Control.
Related Links:
Bio-Rad Laboratories - TDM controls
Bio-Rad Laboratories, Inc.
The control can be used with immunoassay or chromatographic techniques. Called the Lyphochek Whole Blood Immunosuppressant Control, it was designed for use as an assayed quality control material to monitor the precision of immunosuppressant drug test procedures in the clinical laboratory from low to very high concentrations. Immunosuppressant drugs are typically used in transplant therapy to help minimize organ rejection. Lyphochek Control is supplied as a whole blood based material that can be processed in the same manner as patient samples.
Bio-Rad Laboratories, Inc. (Hercules, CA, USA), a manufacturer and distributor of life science clinical diagnostics and research products, released the Lyphochek Whole Blood Immunosuppressant Control.
The company offers technical support and troubleshooting assistance for the control. The Unity Interlaboratory Program and the Unity Real Time, a data management-software solution designed to improve the effectiveness of quality control processes, are available for use with the Lyphochek Control.
Related Links:
Bio-Rad Laboratories - TDM controls
Bio-Rad Laboratories, Inc.
Latest Hematology News
- Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
- Advanced CBC-Derived Indices Integrated into Hematology Platforms
- Blood Test Enables Early Detection of Multiple Myeloma Relapse
- Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
- Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
- Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
- Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
- New Platelet Function Assay Enables Monitoring of Antiplatelet Therapy
- Open Multi-Omics Platform Identifies Prognostic Subtypes in Blood Cancers
- AI-Powered Digital Workflow Standardizes Bone Marrow Aspirate Morphology
- Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
- Fast and Easy Test Could Revolutionize Blood Transfusions
- High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
- AI Algorithm Effectively Distinguishes Alpha Thalassemia Subtypes
Channels
Clinical Chemistry
view channel
Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
Delays in diagnosing serious psychiatric conditions can leave patients without timely support and complicate treatment planning. For bipolar disorder, average time to diagnosis can exceed nine years, and... Read moreAt-Home Blood and Cognitive Tests Support Dementia Risk Stratification
Dementia places a substantial burden on patients and health systems, and identifying individuals at elevated risk remains difficult at scale. In the UK, almost a million people are estimated to be living... Read moreMolecular Diagnostics
view channel
Emerging Biomarkers Advance Early Detection of MASLD and Liver Cancer Risk
Metabolic dysfunction-associated steatotic liver disease (MASLD) affects about 30% of people worldwide and can advance to metabolic dysfunction-associated steatohepatitis (MASH), fibrosis, cirrhosis, and... Read more
Patented Isothermal Amplification Chemistry Advances Decentralized Testing
Molecular diagnostics offer high sensitivity for pathogen detection but typically rely on thermal cycling and specialized instruments, limiting their use outside centralized laboratories.... Read more
Finger-Prick Blood Test Aids Early Tuberculosis Detection and Risk Stratification
Household contacts of people with tuberculosis face an estimated 2% risk of developing disease, yet most are asymptomatic at the time of screening. Early-stage cases are often missed because symptom checks... Read more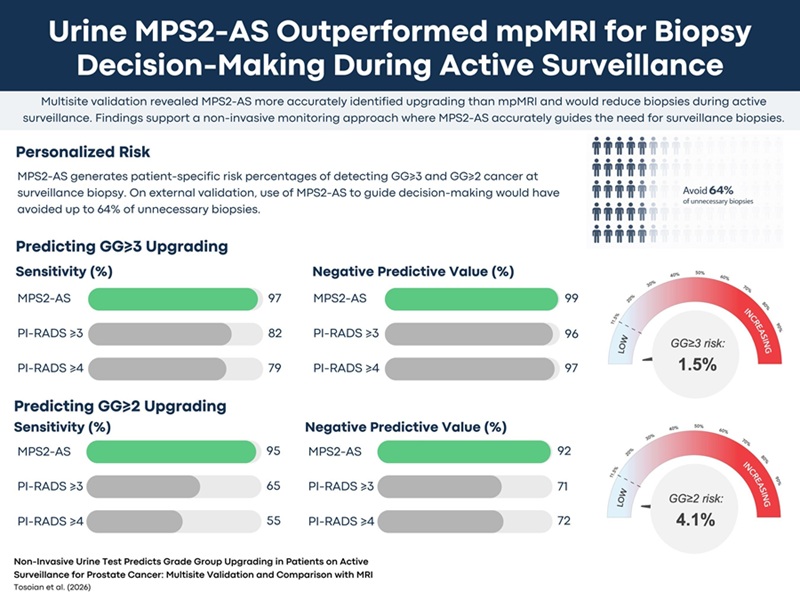
Urine Test Beats MRI in Identifying Prostate Cancer Upgrading During Active Surveillance
Active surveillance is common for men with low-risk prostate cancer, yet deciding when to repeat biopsy remains challenging. Prostate-specific antigen (PSA) testing and magnetic resonance imaging (MRI)... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more