Broadly Neutralizing Antibodies Protect Mice from Alphavirus Infection
|
By LabMedica International staff writers Posted on 24 Nov 2015 |
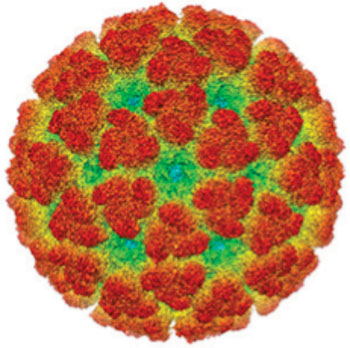
Image: Cryoelectron micrograph reconstruction of the Chikungunya virus (Photo courtesy of the Washington University School of Medicine).
While screening a panel of mouse and human monoclonal antibodies (MAbs) that had been raised against Chikungunya virus, researchers identified several with inhibitory activity against multiple other arthritogenic alphaviruses.
The arthritogenic alphaviruses, which include Chikungunya virus, Mayaro virus, and O’nyong-nyong virus among others, characteristically cause symptoms of fever followed by arthritis-like joint pain. Currently there is no vaccine or treatment for chikungunya or the other alphaviruses.
Investigators at the Washington University School of Medicine (St. Louis, MO, USA) screened 60 neutralizing mouse and human anti-chikungunya monoclonal antibodies and determined that 10 also recognized three or more different arthritogenic alphaviruses. Passive transfer of these "broadly neutralizing" MAbs protected mice against infection by chikungunya, Mayaro, and O’nyong’nyong alphaviruses.
The investigators reported in the November 6, 2015, online edition of the journal Cell that the broadly neutralizing MAbs blocked multiple steps in the viral lifecycle, including entry into host cells and egress. The antibodies were shown to bind to a conserved epitope on the B domain of the viral E2 glycoprotein. The cryoelectron microscopy structure of a Fab fragment bound to the chikungunya E2 B domain determined at 1.6 nanometer resolution showed that antibody binding caused the repositioning of the A domain of E2 so that it was able to cross-link neighboring spikes. This change in the three-dimensional structure of the proteins on the surface of the virus explained how the antibodies were able to prevent viral infection.
“There is a lot of emphasis on identifying and understanding broadly neutralizing antibodies for other viruses - HIV, Hepatitis C virus, Dengue virus, influenza virus - but most of those antibodies neutralize different strains of the same virus,” said senior author Dr. Michael Diamond, professor of medicine at the Washington University School of Medicine. “What we have identified here are antibodies that actually neutralize several different alphaviruses.”
“We have more work to do but are encouraged that targeting this epitope could be a viable strategy for developing vaccines or treatments against chikungunya and other related viruses that cause significant disease worldwide,” said Dr. Diamond. “If you can make an antibody response against this region, you may be able to protect against many viruses in the family. Our group is making proteins now that focus on this epitope, and we are planning to start immunizing animals soon to see if we generate the right kinds of antibodies.”
Related Links:
Washington University School of Medicine
The arthritogenic alphaviruses, which include Chikungunya virus, Mayaro virus, and O’nyong-nyong virus among others, characteristically cause symptoms of fever followed by arthritis-like joint pain. Currently there is no vaccine or treatment for chikungunya or the other alphaviruses.
Investigators at the Washington University School of Medicine (St. Louis, MO, USA) screened 60 neutralizing mouse and human anti-chikungunya monoclonal antibodies and determined that 10 also recognized three or more different arthritogenic alphaviruses. Passive transfer of these "broadly neutralizing" MAbs protected mice against infection by chikungunya, Mayaro, and O’nyong’nyong alphaviruses.
The investigators reported in the November 6, 2015, online edition of the journal Cell that the broadly neutralizing MAbs blocked multiple steps in the viral lifecycle, including entry into host cells and egress. The antibodies were shown to bind to a conserved epitope on the B domain of the viral E2 glycoprotein. The cryoelectron microscopy structure of a Fab fragment bound to the chikungunya E2 B domain determined at 1.6 nanometer resolution showed that antibody binding caused the repositioning of the A domain of E2 so that it was able to cross-link neighboring spikes. This change in the three-dimensional structure of the proteins on the surface of the virus explained how the antibodies were able to prevent viral infection.
“There is a lot of emphasis on identifying and understanding broadly neutralizing antibodies for other viruses - HIV, Hepatitis C virus, Dengue virus, influenza virus - but most of those antibodies neutralize different strains of the same virus,” said senior author Dr. Michael Diamond, professor of medicine at the Washington University School of Medicine. “What we have identified here are antibodies that actually neutralize several different alphaviruses.”
“We have more work to do but are encouraged that targeting this epitope could be a viable strategy for developing vaccines or treatments against chikungunya and other related viruses that cause significant disease worldwide,” said Dr. Diamond. “If you can make an antibody response against this region, you may be able to protect against many viruses in the family. Our group is making proteins now that focus on this epitope, and we are planning to start immunizing animals soon to see if we generate the right kinds of antibodies.”
Related Links:
Washington University School of Medicine
Latest BioResearch News
- Genome Analysis Predicts Likelihood of Neurodisability in Oxygen-Deprived Newborns
- Gene Panel Predicts Disease Progession for Patients with B-cell Lymphoma
- New Method Simplifies Preparation of Tumor Genomic DNA Libraries
- New Tool Developed for Diagnosis of Chronic HBV Infection
- Panel of Genetic Loci Accurately Predicts Risk of Developing Gout
- Disrupted TGFB Signaling Linked to Increased Cancer-Related Bacteria
- Gene Fusion Protein Proposed as Prostate Cancer Biomarker
- NIV Test to Diagnose and Monitor Vascular Complications in Diabetes
- Semen Exosome MicroRNA Proves Biomarker for Prostate Cancer
- Genetic Loci Link Plasma Lipid Levels to CVD Risk
- Newly Identified Gene Network Aids in Early Diagnosis of Autism Spectrum Disorder
- Link Confirmed between Living in Poverty and Developing Diseases
- Genomic Study Identifies Kidney Disease Loci in Type I Diabetes Patients
- Liquid Biopsy More Effective for Analyzing Tumor Drug Resistance Mutations
- New Liquid Biopsy Assay Reveals Host-Pathogen Interactions
- Method Developed for Enriching Trophoblast Population in Samples
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more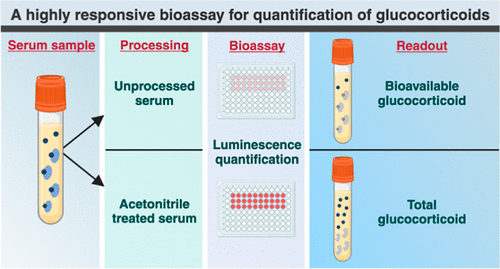
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
Novel Biomarkers to Improve Diagnosis of Renal Cell Carcinoma Subtypes
Renal cell carcinomas (RCCs) are notably diverse, encompassing over 20 distinct subtypes and generally categorized into clear cell and non-clear cell types; around 20% of all RCCs fall into the non-clear... Read more
RNA-Powered Molecular Test to Help Combat Early-Age Onset Colorectal Cancer
Colorectal cancer (CRC) ranks as the second most lethal cancer in the United States. Nevertheless, many Americans eligible for screening do not undergo testing due to limited access or reluctance towards... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more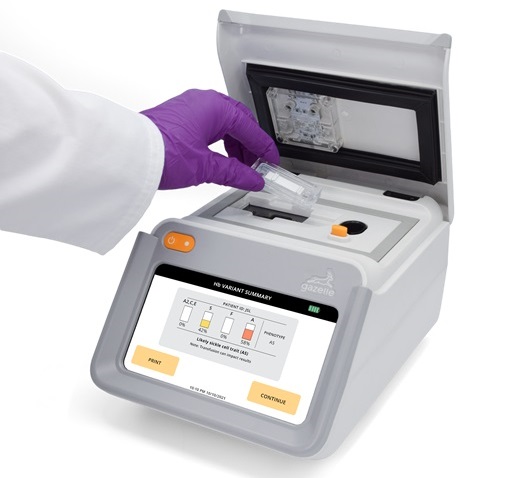
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read moreMicrobiology
view channel
Integrated Solution Ushers New Era of Automated Tuberculosis Testing
Tuberculosis (TB) is responsible for 1.3 million deaths every year, positioning it as one of the top killers globally due to a single infectious agent. In 2022, around 10.6 million people were diagnosed... Read more
Automated Sepsis Test System Enables Rapid Diagnosis for Patients with Severe Bloodstream Infections
Sepsis affects up to 50 million people globally each year, with bacteraemia, formerly known as blood poisoning, being a major cause. In the United States alone, approximately two million individuals are... Read moreEnhanced Rapid Syndromic Molecular Diagnostic Solution Detects Broad Range of Infectious Diseases
GenMark Diagnostics (Carlsbad, CA, USA), a member of the Roche Group (Basel, Switzerland), has rebranded its ePlex® system as the cobas eplex system. This rebranding under the globally renowned cobas name... Read more
Clinical Decision Support Software a Game-Changer in Antimicrobial Resistance Battle
Antimicrobial resistance (AMR) is a serious global public health concern that claims millions of lives every year. It primarily results from the inappropriate and excessive use of antibiotics, which reduces... Read morePathology
view channel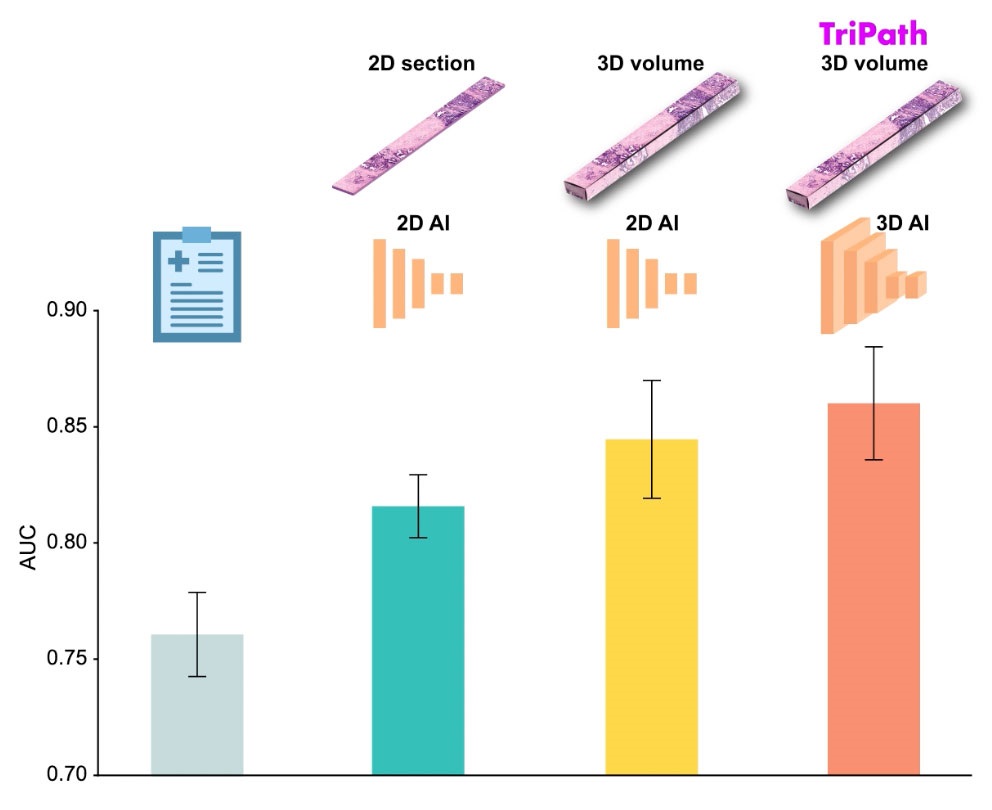
AI Advancements Enable Leap into 3D Pathology
Human tissue is complex, intricate, and naturally three-dimensional. However, the thin two-dimensional tissue slices commonly used by pathologists to diagnose diseases provide only a limited view of the... Read more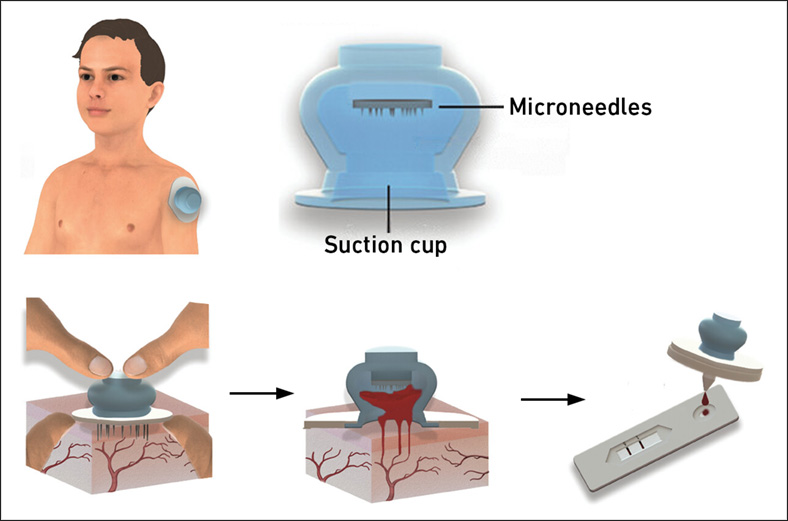
New Blood Test Device Modeled on Leeches to Help Diagnose Malaria
Many individuals have a fear of needles, making the experience of having blood drawn from their arm particularly distressing. An alternative method involves taking blood from the fingertip or earlobe,... Read more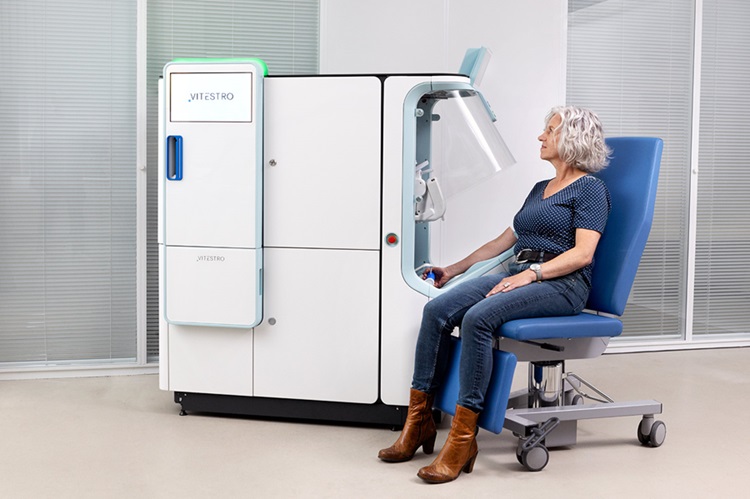
Robotic Blood Drawing Device to Revolutionize Sample Collection for Diagnostic Testing
Blood drawing is performed billions of times each year worldwide, playing a critical role in diagnostic procedures. Despite its importance, clinical laboratories are dealing with significant staff shortages,... Read more.jpg)
Use of DICOM Images for Pathology Diagnostics Marks Significant Step towards Standardization
Digital pathology is rapidly becoming a key aspect of modern healthcare, transforming the practice of pathology as laboratories worldwide adopt this advanced technology. Digital pathology systems allow... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more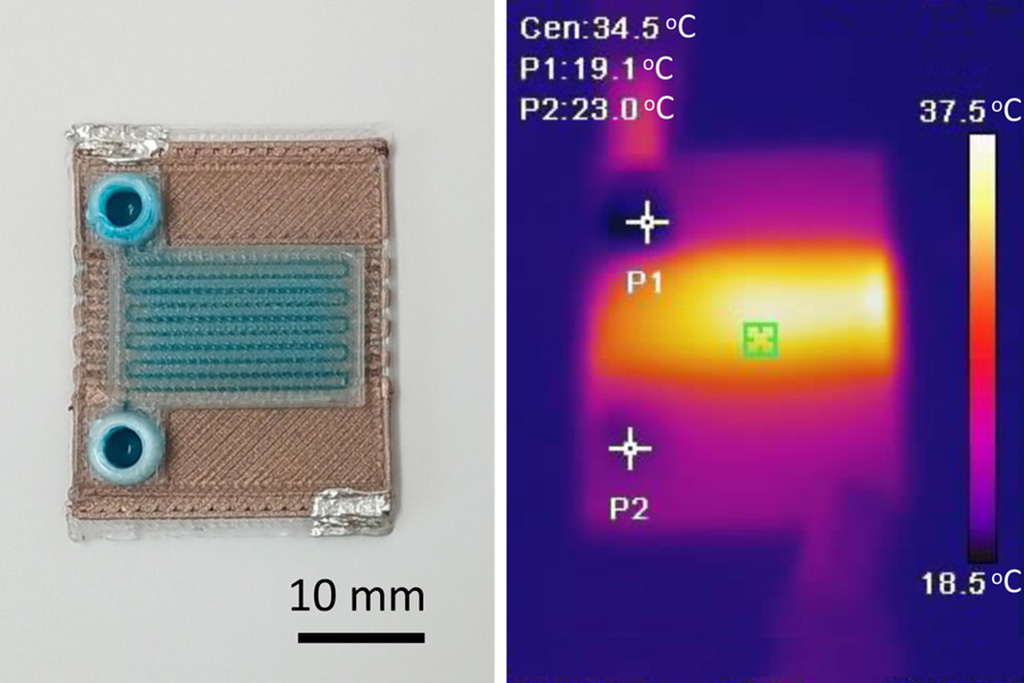
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel
Beckman Coulter and MeMed Expand Host Immune Response Diagnostics Partnership
Beckman Coulter Diagnostics (Brea, CA, USA) and MeMed BV (Haifa, Israel) have expanded their host immune response diagnostics partnership. Beckman Coulter is now an authorized distributor of the MeMed... Read more_1.jpg)
Thermo Fisher and Bio-Techne Enter Into Strategic Distribution Agreement for Europe
Thermo Fisher Scientific (Waltham, MA USA) has entered into a strategic distribution agreement with Bio-Techne Corporation (Minneapolis, MN, USA), resulting in a significant collaboration between two industry... Read more
ECCMID Congress Name Changes to ESCMID Global
Over the last few years, the European Society of Clinical Microbiology and Infectious Diseases (ESCMID, Basel, Switzerland) has evolved remarkably. The society is now stronger and broader than ever before... Read more