QuidelOrtho Demonstrates Automated, Sample-To-Result, Multiplex RT-PCR Testing Platform at Medlab Middle East 2023
|
By LabMedica International staff writers Posted on 08 Feb 2023 |

At Medlab Middle East 2023, QuidelOrtho Corporation (San Diego, CA, USA) is showcasing for the first time the benefits of the combined organization’s consolidated portfolio of world-class technologies and platforms, through expanded access to clinical chemistry, immunoassay, molecular diagnostics, immunohematology, donor screening, and point-of-care diagnostics offerings.
At the QuidelOrtho booth, the focus is on the Savanna automated, sample-to-result, multiplex RT-PCR testing platform that performs real-time Polymerization Chain Reaction (PCR) tests by using the Savanna instrument and Savanna assay cartridges. This evolutionary multiplex molecular platform enables professional customers to analyze up to 12 pathogens or targets, plus controls, from a single assay run in less than 30 minutes. After inserting the assay cartridge, the instrument performs sample and reagent preparation, nucleic acid extraction and amplification, real-time detection of RNA or DNA target sequence, and qualitative or quantitative result interpretation from a variety of sample types. The Savanna system is intended to aid in the diagnosis of the related diseases.
QuidelOrtho is also demonstrating the Savanna RVP4 Assay (Respiratory Viral Panel-4), a rapid, multiplexed nucleic acid test intended for use with the Savanna instrument for the simultaneous qualitative detection and differentiation of influenza A (Flu A), influenza B (Flu B), respiratory syncytial virus (RSV), and SARS-CoV-2 RNA isolated from human nasal or nasopharyngeal swabs in media obtained from patients with signs and symptoms of respiratory tract infection. The in vitro diagnostic test is intended to aid in the differential diagnosis of infections with these viruses. The Savanna RVP4 Assay comes in a fully integrated cartridge that is stable at room temperature.
Related Links:
QuidelOrtho Corporation
Latest Medlab 2023 News
- Fapon Biotech Presents Latest IVD Innovations at Medlab Middle East 2023
- SNIBE Brings Latest Innovations in Immunoassay, Clinical Biochemistry and Molecular Diagnostics to Medlab Middle East 2023
- Sansure Exhibits Portable Molecular Workstation at Medlab Middle East 2023
- QIAGEN Demonstrates Its Revolutionary Molecular Diagnostic Solution at Medlab Middle East 2023
- Erba Introduces XL Range of Fully Automated Clinical Chemistry Systems at Medlab Middle East 2023
- BioPerfectus Presents Total PCR Solution at Medlab Middle East 2023
- 77 Elektronika Highlights Latest Urinanalysis Solutions at Medlab Middle East 2023
- Sysmex Introduces Automated Urinalysis Testing Solutions that Revolutionize Workflow at Medlab Middle East 2023
- Abbott Demonstrates ID NOW Rapid Molecular Respiratory Testing Platform at Medlab Middle East 2023
- Horiba Showcases HELO Global Hematology Solution at Medlab Middle East 2023
- LGC Clinical Diagnostics Demonstrates How Labs Can Enhance QC Efficiencies at Medlab Middle East 2023
- DiaSys Highlights Comprehensive Portfolio of New Assays at Medlab Middle East 2023
- Illumina Introduces Its Most Powerful and Sustainable Sequencer Yet at Medlab Middle East 2023
- BioVendor Presents Latest Innovations in Immunodiagnostics and Molecular Diagnostics at Medlab Middle East 2023
- Seegene Highlights Cutting-Edge '3 Ct' PCR Assay at Medlab Middle East 2023
- Mindray Presents Holistic IVD Solutions for Laboratories at Medlab Middle East 2023
Channels
Clinical Chemistry
view channel
Urinary Biomarker Assay Predicts Kidney Disease Progression Beyond Standard Measures
Many patients with type 2 diabetes and chronic kidney disease continue to experience progressive renal decline, yet conventional markers such as albuminuria and estimated glomerular filtration rate (eGFR)... Read more
Saliva-Based Test Detects Biochemical Signs of Sleep Loss
Acute sleep loss impairs cognition and motor skills, raising safety risks that resemble alcohol intoxication. Clinicians currently lack an objective biochemical test to determine when someone is dangerously... Read more
Simple Dual-Tau Blood Test Detects and Stages Alzheimer’s Disease
Alzheimer’s disease is typically confirmed and staged with positron emission tomography scans and cerebrospinal fluid testing, procedures that are costly and invasive. Broader access to minimally invasive... Read more
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read moreMolecular Diagnostics
view channel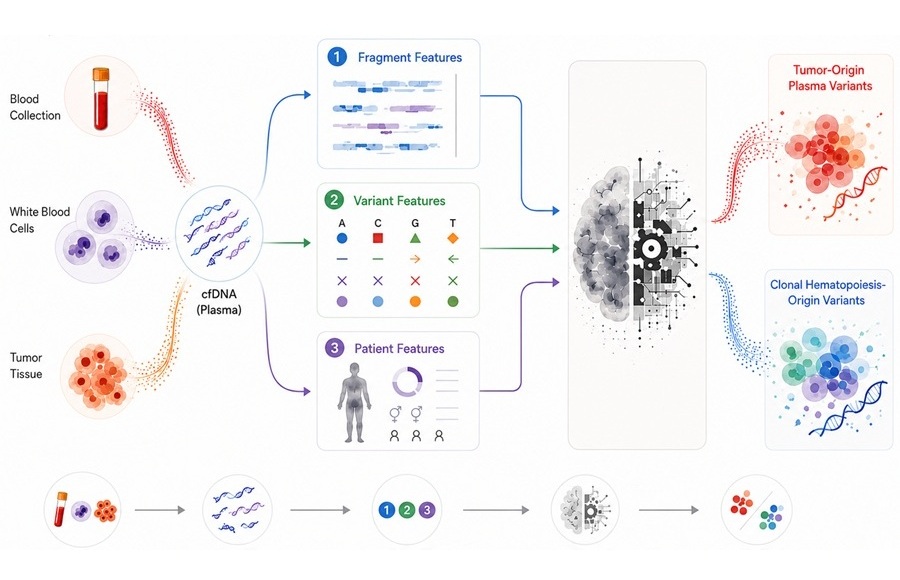
AI Tool Improves Accuracy of Cancer Liquid Biopsy for Therapy Selection
Liquid biopsy is increasingly used to guide targeted therapy by detecting tumor-derived mutations in blood, but distinguishing true tumor signals from background noise remains challenging.... Read more
Study Highlights Inherited Breast Cancer Risk Genes in Young Black Women
Early-onset breast cancer remains a significant concern, with some cases presenting as triple-negative disease, one of its most aggressive forms. Black women face disproportionately elevated risks of developing... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channelAptamer-Based Biosensor Enables Mutation-Resilient SARS-CoV-2 Detection
Rapid evolution of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can undermine existing molecular diagnostics, especially when assays target small viral components. Double-antibody sandwich... Read more
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
TORCH Infection Trends Point to Need for Tailored Screening in Pregnancy
Congenital TORCH infections can be asymptomatic during pregnancy yet cause stillbirth, birth defects, and lifelong disability in infants. Many regions still lack robust surveillance to guide testing and... Read more
New Culture Medium Speeds C. difficile Resistance Detection and Reduces Costs
Clostridioides difficile infections remain a persistent threat in hospitals and communities, affecting about 500,000 people in the United States each year. Severe cases can be fatal within 30 days of diagnosis,... Read morePathology
view channel
AI Pathology Tool Predicts Meningioma Recurrence from Routine Slides
Meningiomas are the most common primary brain tumors in adults, yet their course ranges from indolent to highly recurrent disease. Estimating an individual patient’s recurrence risk often requires advanced... Read more
3D Spatial Multi-Omics Maps Intra-Tumor Diversity in Colorectal Cancer
Colorectal cancer remains a leading cause of cancer death, and clinical decision-making is complicated by marked intra-tumor heterogeneity. Conventional bulk sequencing averages molecular signals across... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel
Genetic Testing Program Expands Detection of Alpha-1 Antitrypsin Deficiency
Alpha-1 Antitrypsin Deficiency (AATD) is a progressive genetic condition, the leading known genetic risk factor for chronic obstructive pulmonary disease (COPD), and a cause of liver disease in both children... Read more
























