Zika Virus Test Kit Receives American Marketing Approval
|
By LabMedica International staff writers Posted on 16 Sep 2019 |
![Image: Zika virus particles (red) shown in African green monkey kidney cells (Photo courtesy of the [U.S.] National Institute of Allergy and Infectious Diseases). Image: Zika virus particles (red) shown in African green monkey kidney cells (Photo courtesy of the [U.S.] National Institute of Allergy and Infectious Diseases).](https://globetechcdn.com/mobile_labmedica/images/stories/articles/article_images/2019-09-16/GMS-151.jpg)
Image: Zika virus particles (red) shown in African green monkey kidney cells (Photo courtesy of the [U.S.] National Institute of Allergy and Infectious Diseases).
A blood test designed to confirm diagnosis of Zika virus infection has been granted 510(k) marketing approval that allows it to be sold in the United States.
The InBios International (Seattle, WA, USA) ZIKV Detect 2.0 IgM Capture ELISA Kit is intended for the presumptive detection of Zika virus IgM antibodies in human sera collected from individuals who meet the criteria of the [U.S.] Centers for Disease Control and Prevention for Zika virus clinical infection. These criteria include a history of associated clinical symptoms or travel from a geographic location with active Zika transmission.
The assay kit, which is based in part on technology licensed from Washington University (St. Louis, MO, USA), contains all the necessary reagents and controls to perform the test and obtain results in about four hours.
Performance of the InBios Zika assay was evaluated by testing 807 unique specimens from 353 subjects at endemic sites and 256 subjects at non-endemic sites. In samples from patients collected seven days or later after onset of symptoms, results correctly identified more than 90% of patients confirmed positive for Zika IgM and more than 96% of patients confirmed negative. In addition to identifying Zika-IgM positive patient samples, the assay differentiated individuals with Zika IgM from those infected with other flaviviruses (such as dengue or West Nile virus), which cross-react with Zika antibodies.
Results of the test are intended for use in conjunction with clinical observations, patient history, epidemiological information, and other laboratory evidence to make patient management decisions. Zika IgM levels are variable over the course of the infection and may be detectable near day four, post-onset of symptoms and persist up to approximately 12 weeks following initial infection.
"Pregnant women living in or visiting places where Zika is endemic will want to know if they have been exposed to the virus," said technology co-inventor Dr. Michael S. Diamond, professor of molecular microbiology, pathology, and immunology at Washington University. "This test, along with another that detects viral genetic material at very early stages of infection, will help women and their doctors make informed healthcare decisions."
The InBios International (Seattle, WA, USA) ZIKV Detect 2.0 IgM Capture ELISA Kit is intended for the presumptive detection of Zika virus IgM antibodies in human sera collected from individuals who meet the criteria of the [U.S.] Centers for Disease Control and Prevention for Zika virus clinical infection. These criteria include a history of associated clinical symptoms or travel from a geographic location with active Zika transmission.
The assay kit, which is based in part on technology licensed from Washington University (St. Louis, MO, USA), contains all the necessary reagents and controls to perform the test and obtain results in about four hours.
Performance of the InBios Zika assay was evaluated by testing 807 unique specimens from 353 subjects at endemic sites and 256 subjects at non-endemic sites. In samples from patients collected seven days or later after onset of symptoms, results correctly identified more than 90% of patients confirmed positive for Zika IgM and more than 96% of patients confirmed negative. In addition to identifying Zika-IgM positive patient samples, the assay differentiated individuals with Zika IgM from those infected with other flaviviruses (such as dengue or West Nile virus), which cross-react with Zika antibodies.
Results of the test are intended for use in conjunction with clinical observations, patient history, epidemiological information, and other laboratory evidence to make patient management decisions. Zika IgM levels are variable over the course of the infection and may be detectable near day four, post-onset of symptoms and persist up to approximately 12 weeks following initial infection.
"Pregnant women living in or visiting places where Zika is endemic will want to know if they have been exposed to the virus," said technology co-inventor Dr. Michael S. Diamond, professor of molecular microbiology, pathology, and immunology at Washington University. "This test, along with another that detects viral genetic material at very early stages of infection, will help women and their doctors make informed healthcare decisions."
Latest Technology News
- Robotic Technology Unveiled for Automated Diagnostic Blood Draws
- ADLM Launches First-of-Its-Kind Data Science Program for Laboratory Medicine Professionals
- Aptamer Biosensor Technology to Transform Virus Detection
- AI Models Could Predict Pre-Eclampsia and Anemia Earlier Using Routine Blood Tests
- AI-Generated Sensors Open New Paths for Early Cancer Detection
- Pioneering Blood Test Detects Lung Cancer Using Infrared Imaging
- AI Predicts Colorectal Cancer Survival Using Clinical and Molecular Features
- Diagnostic Chip Monitors Chemotherapy Effectiveness for Brain Cancer
- Machine Learning Models Diagnose ALS Earlier Through Blood Biomarkers
- Artificial Intelligence Model Could Accelerate Rare Disease Diagnosis
Channels
Clinical Chemistry
view channel
New PSA-Based Prognostic Model Improves Prostate Cancer Risk Assessment
Prostate cancer is the second-leading cause of cancer death among American men, and about one in eight will be diagnosed in their lifetime. Screening relies on blood levels of prostate-specific antigen... Read more
Extracellular Vesicles Linked to Heart Failure Risk in CKD Patients
Chronic kidney disease (CKD) affects more than 1 in 7 Americans and is strongly associated with cardiovascular complications, which account for more than half of deaths among people with CKD.... Read moreHematology
view channel
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read more
Fast and Easy Test Could Revolutionize Blood Transfusions
Blood transfusions are a cornerstone of modern medicine, yet red blood cells can deteriorate quietly while sitting in cold storage for weeks. Although blood units have a fixed expiration date, cells from... Read more
Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
High-volume hemostasis sections must sustain rapid turnaround while managing reruns and reflex testing. Manual tube handling and preanalytical checks can strain staff time and increase opportunities for error.... Read more
High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
Blood clotting is essential for preventing bleeding, but even small imbalances can lead to serious conditions such as thrombosis or dangerous hemorrhage. In cardiovascular disease, clinicians often struggle... Read moreImmunology
view channelBlood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
Small cell lung cancer (SCLC) is an aggressive disease with limited treatment options, and even newly approved immunotherapies do not benefit all patients. While immunotherapy can extend survival for some,... Read more
Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
Targeted cancer therapies such as PARP inhibitors can be highly effective, but only for patients whose tumors carry specific DNA repair defects. Identifying these patients accurately remains challenging,... Read more
Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
Immunotherapy has transformed cancer treatment, but only a small proportion of patients experience lasting benefit, with response rates often remaining between 10% and 20%. Clinicians currently lack reliable... Read moreMicrobiology
view channel
Comprehensive Review Identifies Gut Microbiome Signatures Associated With Alzheimer’s Disease
Alzheimer’s disease affects approximately 6.7 million people in the United States and nearly 50 million worldwide, yet early cognitive decline remains difficult to characterize. Increasing evidence suggests... Read moreAI-Powered Platform Enables Rapid Detection of Drug-Resistant C. Auris Pathogens
Infections caused by the pathogenic yeast Candida auris pose a significant threat to hospitalized patients, particularly those with weakened immune systems or those who have invasive medical devices.... Read morePathology
view channel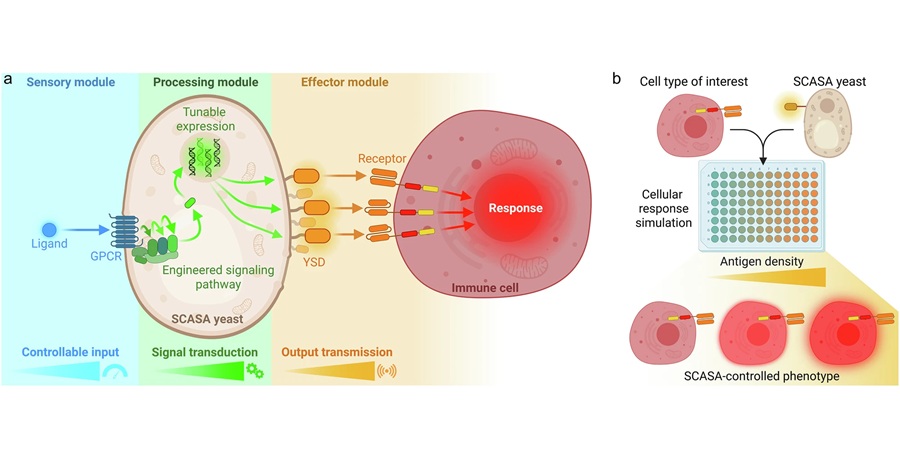
Engineered Yeast Cells Enable Rapid Testing of Cancer Immunotherapy
Developing new cancer immunotherapies is a slow, costly, and high-risk process, particularly for CAR T cell treatments that must precisely recognize cancer-specific antigens. Small differences in tumor... Read more
First-Of-Its-Kind Test Identifies Autism Risk at Birth
Autism spectrum disorder is treatable, and extensive research shows that early intervention can significantly improve cognitive, social, and behavioral outcomes. Yet in the United States, the average age... Read moreTechnology
view channel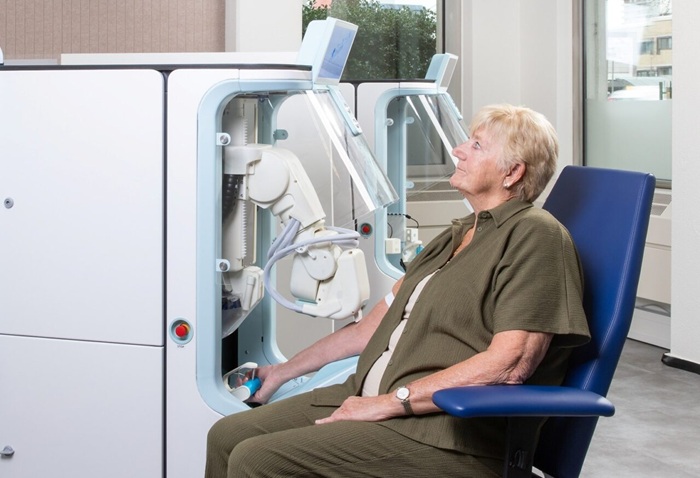
Robotic Technology Unveiled for Automated Diagnostic Blood Draws
Routine diagnostic blood collection is a high‑volume task that can strain staffing and introduce human‑dependent variability, with downstream implications for sample quality and patient experience.... Read more
ADLM Launches First-of-Its-Kind Data Science Program for Laboratory Medicine Professionals
Clinical laboratories generate billions of test results each year, creating a treasure trove of data with the potential to support more personalized testing, improve operational efficiency, and enhance patient care.... Read moreAptamer Biosensor Technology to Transform Virus Detection
Rapid and reliable virus detection is essential for controlling outbreaks, from seasonal influenza to global pandemics such as COVID-19. Conventional diagnostic methods, including cell culture, antigen... Read more
AI Models Could Predict Pre-Eclampsia and Anemia Earlier Using Routine Blood Tests
Pre-eclampsia and anemia are major contributors to maternal and child mortality worldwide, together accounting for more than half a million deaths each year and leaving millions with long-term health complications.... Read moreIndustry
view channelNew Collaboration Brings Automated Mass Spectrometry to Routine Laboratory Testing
Mass spectrometry is a powerful analytical technique that identifies and quantifies molecules based on their mass and electrical charge. Its high selectivity, sensitivity, and accuracy make it indispensable... Read more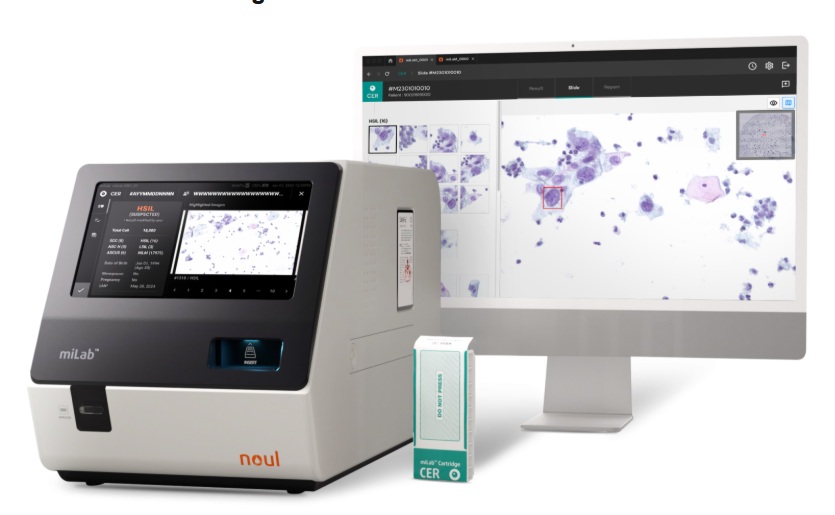
AI-Powered Cervical Cancer Test Set for Major Rollout in Latin America
Noul Co., a Korean company specializing in AI-based blood and cancer diagnostics, announced it will supply its intelligence (AI)-based miLab CER cervical cancer diagnostic solution to Mexico under a multi‑year... Read more
Diasorin and Fisher Scientific Enter into US Distribution Agreement for Molecular POC Platform
Diasorin (Saluggia, Italy) has entered into an exclusive distribution agreement with Fisher Scientific, part of Thermo Fisher Scientific (Waltham, MA, USA), for the LIAISON NES molecular point-of-care... Read more