Blood Test Aids Early Detection of Rheumatoid Arthritis
|
By LabMedica International staff writers Posted on 09 Mar 2015 |
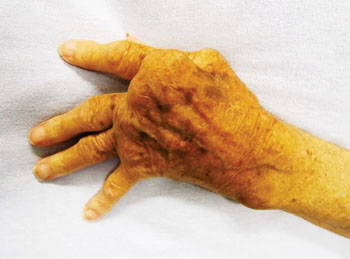
Image: Severe rheumatoid arthritis in a hand which was never treated (Photo courtesy of James Heilman, MD).
A blood test that measures the levels of a certain protein can be used in conjunction with clinical assessments to assist with the diagnosis of rheumatoid arthritis is now available in North America.
Rheumatoid arthritis (RA) is a chronic, systemic inflammatory disorder that primarily affects joints and it may result in deformed and painful joints, which can lead to loss of function. The disease may also have signs and symptoms in organs other than joints.
The JOINTstat test (Augurex Life Science; Vancouver, BC, Canada) measures 14-3-3η (eta), a protein involved in joint damage that leads to debilitation in patients with RA, a disease that affects one out of every 100 Canadians. High levels of 14-3-3η protein indicate that the patient should be referred to a rheumatologist. JOINTstat has been evaluated in clinical studies in over 3,000 patients and has been available in the USA since late 2013.
Extracellular 14-3-3η protein is a potent ligand and activator of intracellular signaling pathways that lead to the upregulation of inflammation and joint damage factors involved in RA pathogenesis. A quantitative enzyme-linked immunosorbent assay is used to assess 14-3-3η protein levels. Studies have shown that when the test is positive, greater than 0.19 ng/mL, it provides a 5 to 50 times greater likelihood of having RA versus non-RA. At high levels of greater than 80 ng/mL is an indicator of RA that will lead to more joint damage over three years and at lower levels of less than 0.4 ng/mL or negative in RA diagnosed patients indicates a higher likelihood of response to RA therapy.
Walter Maksymowych, MD, FRCPC, a professor in the department of medicine, division of rheumatology at the University of Alberta (Edmonton, AB, Canada) said, “When diagnosed early, rheumatoid arthritis can be better managed with drug therapies, resulting in better outcomes by limiting disease progression. Current diagnostic tests are limited in their ability to catch the disease early. With the availability of JOINTstat in Canada, physicians now have the opportunity to treat rheumatoid arthritis early, prior to the onset of significant joint damage. This represents an important milestone towards the goal of personalized medicine.” The study was presented at the Canadian Rheumatology Association (CRA) Annual Scientific Meeting held February 4–7, 2015, in Québec City (QC, Canada).
Related Links:
Augurex Life Science
University of Alberta
Rheumatoid arthritis (RA) is a chronic, systemic inflammatory disorder that primarily affects joints and it may result in deformed and painful joints, which can lead to loss of function. The disease may also have signs and symptoms in organs other than joints.
The JOINTstat test (Augurex Life Science; Vancouver, BC, Canada) measures 14-3-3η (eta), a protein involved in joint damage that leads to debilitation in patients with RA, a disease that affects one out of every 100 Canadians. High levels of 14-3-3η protein indicate that the patient should be referred to a rheumatologist. JOINTstat has been evaluated in clinical studies in over 3,000 patients and has been available in the USA since late 2013.
Extracellular 14-3-3η protein is a potent ligand and activator of intracellular signaling pathways that lead to the upregulation of inflammation and joint damage factors involved in RA pathogenesis. A quantitative enzyme-linked immunosorbent assay is used to assess 14-3-3η protein levels. Studies have shown that when the test is positive, greater than 0.19 ng/mL, it provides a 5 to 50 times greater likelihood of having RA versus non-RA. At high levels of greater than 80 ng/mL is an indicator of RA that will lead to more joint damage over three years and at lower levels of less than 0.4 ng/mL or negative in RA diagnosed patients indicates a higher likelihood of response to RA therapy.
Walter Maksymowych, MD, FRCPC, a professor in the department of medicine, division of rheumatology at the University of Alberta (Edmonton, AB, Canada) said, “When diagnosed early, rheumatoid arthritis can be better managed with drug therapies, resulting in better outcomes by limiting disease progression. Current diagnostic tests are limited in their ability to catch the disease early. With the availability of JOINTstat in Canada, physicians now have the opportunity to treat rheumatoid arthritis early, prior to the onset of significant joint damage. This represents an important milestone towards the goal of personalized medicine.” The study was presented at the Canadian Rheumatology Association (CRA) Annual Scientific Meeting held February 4–7, 2015, in Québec City (QC, Canada).
Related Links:
Augurex Life Science
University of Alberta
Latest Immunology News
- Point-of-Care Tests Could Expand Access to Mpox Diagnosis
- T-Cell Senescence Profiling May Predict CAR T Responses
- Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
- Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
- Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
- Study Finds Influenza Often Undiagnosed in Winter Deaths
- Combined Screening Approach Identifies Early Leprosy Cases
- Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
- FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
- Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
- Cancer Mutation ‘Fingerprints’ to Improve Prediction of Immunotherapy Response
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Blood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
- Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
Channels
Clinical Chemistry
view channel
Ultrasensitive Test Detects Key Biomarker of Frontotemporal Dementia Subtype
Dementia affects more than 57 million people worldwide and is projected to nearly double within two decades, straining health systems and families. While biomarkers now enable accurate identification of... Read more
Routine Blood Tests Years Before Pregnancy Could Identify Preeclampsia Risk
High blood pressure during pregnancy is common and can progress to pre-eclampsia, making close monitoring at antenatal visits essential. However, most risk assessment begins only after pregnancy has started.... Read moreMolecular Diagnostics
view channel
Liquid Biopsy Biomarkers Distinguish Inflammatory Breast Cancer and Support Monitoring
Inflammatory breast cancer is among the most aggressive forms of breast malignancy and remains challenging to diagnose and monitor. Obtaining tumor tissue can be difficult, and standard genome and RNA... Read more
Blood Test Maps Tumor Microenvironment to Predict Immunotherapy Response
Immunotherapy has transformed cancer care, yet durable benefit remains limited to a subset of patients, and clinicians still lack reliable tools to predict response before treatment begins.... Read more
Multiplex Respiratory Panel Integrates Automated Extraction to Streamline High-Volume Testing
Respiratory infections drive heavy testing volumes in clinical laboratories, where accurate, timely results across multiple pathogens are essential. Many labs are seeking to streamline workflows and increase... Read moreHematology
view channel
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read more
Blood Test Enables Early Detection of Multiple Myeloma Relapse
Bone marrow biopsies remain central to diagnosing and monitoring multiple myeloma, yet the procedure is painful, invasive, and often repeated over time. Older patients—who represent most new cases—can... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read more
New Platform Captures Extracellular Vesicles for Early Cancer Detection
Early diagnosis remains the most effective way to reduce cancer mortality, yet many screening tools miss disease at its earliest stages. Biomarkers shed by tumors into blood and other fluids can be scarce... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more