Recombinant Antigen-Based ELISA Evaluated for Syphilis
|
By LabMedica International staff writers Posted on 28 Aug 2013 |
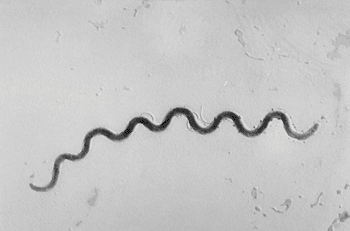
Image: Photomicrograph of Treponema pallidum (Photo courtesy of Susan Lindsley).
The diagnostic performance of the latest screening enzyme-linked immunosorbent assay (ELISA) for syphilis has been compared with the currently used treponemal tests.
The etiological agent of syphilis, Treponema pallidum, cannot be cultured and there is no single optimal alternative test. Serological testing is the most frequently used approach in laboratory diagnosis of the disease.
Scientists at Sekisui Virotech (Rüsselsheim, Germany) compared their Treponema pallidum Screen ELISA with standard tests. These tests included the fluorescent treponemal antibody absorption (FTA-ABS, Zeus Scientific; Branchburg, NJ, USA) test, which is an indirect fluorescent antibody technique; the T. pallidum particle agglutination (TPPA, Fujirebio; Hoofddorp, the Netherlands) test, which is a qualitative assay for the detection of antibodies to T. pallidum in serum or plasma. The most relevant test used for comparison was the Trep-Sure ELISA (Phoenix Bio-Tech Corporation; Mississauga, ON, Canada).
To establish the sensitivity and specificity of the Virotech Screen, 421 serum samples from different panels of infected and non-infected patients, sera from seronegative pregnant women as well as international syphilis standard sera and panels were tested. In comparison to combined TPPA/FTA-abs tests, Phoenix Trep-Sure and Virotech Screen demonstrated a sensitivity of 100% and a specificity of 93.9% and 98.3%, respectively.
All samples of a well-defined syphilis serum panel were correctly identified by the Virotech test, whereas the Phoenix test identified two Treponema negative samples as equivocal. The Trep Sure test is approved by the US Food and Drug Administration (FDA; Silver Springs, MD, USA). Results of both ELISAs highly correlated with TPPA negative and positive samples. The analytical sensitivity of the Virotech Screen with international standards was determined at 0.02 IU/mL and 0.03 IU/mL, and was slightly superior to the Phoenix Trep-Sure.
The authors concluded that the Virotech Screen ELISA demonstrated good diagnostic sensitivity and specificity when evaluated as a screening test for syphilis among various patient populations, including samples with increased rates of false positive nontreponemal test results. The Virotech ELISA may be used in automatic analyzers as an alternative to the manual TPPA. However, the use of a confirmatory test remains a must in order to avoid false-positive results. The study was published in the May/June, 2013 issue of the journal Clinical Laboratory.
Related Links:
Sekisui Virotech
Zeus Scientific
Fujirebio
The etiological agent of syphilis, Treponema pallidum, cannot be cultured and there is no single optimal alternative test. Serological testing is the most frequently used approach in laboratory diagnosis of the disease.
Scientists at Sekisui Virotech (Rüsselsheim, Germany) compared their Treponema pallidum Screen ELISA with standard tests. These tests included the fluorescent treponemal antibody absorption (FTA-ABS, Zeus Scientific; Branchburg, NJ, USA) test, which is an indirect fluorescent antibody technique; the T. pallidum particle agglutination (TPPA, Fujirebio; Hoofddorp, the Netherlands) test, which is a qualitative assay for the detection of antibodies to T. pallidum in serum or plasma. The most relevant test used for comparison was the Trep-Sure ELISA (Phoenix Bio-Tech Corporation; Mississauga, ON, Canada).
To establish the sensitivity and specificity of the Virotech Screen, 421 serum samples from different panels of infected and non-infected patients, sera from seronegative pregnant women as well as international syphilis standard sera and panels were tested. In comparison to combined TPPA/FTA-abs tests, Phoenix Trep-Sure and Virotech Screen demonstrated a sensitivity of 100% and a specificity of 93.9% and 98.3%, respectively.
All samples of a well-defined syphilis serum panel were correctly identified by the Virotech test, whereas the Phoenix test identified two Treponema negative samples as equivocal. The Trep Sure test is approved by the US Food and Drug Administration (FDA; Silver Springs, MD, USA). Results of both ELISAs highly correlated with TPPA negative and positive samples. The analytical sensitivity of the Virotech Screen with international standards was determined at 0.02 IU/mL and 0.03 IU/mL, and was slightly superior to the Phoenix Trep-Sure.
The authors concluded that the Virotech Screen ELISA demonstrated good diagnostic sensitivity and specificity when evaluated as a screening test for syphilis among various patient populations, including samples with increased rates of false positive nontreponemal test results. The Virotech ELISA may be used in automatic analyzers as an alternative to the manual TPPA. However, the use of a confirmatory test remains a must in order to avoid false-positive results. The study was published in the May/June, 2013 issue of the journal Clinical Laboratory.
Related Links:
Sekisui Virotech
Zeus Scientific
Fujirebio
Latest Immunology News
- Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
- Study Points to Autoimmune Pathway Behind Long COVID Symptoms
- Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
- Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
- Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
- Biomarkers and Molecular Testing Advance Precision Allergy Care
- Point-of-Care Tests Could Expand Access to Mpox Diagnosis
- T-Cell Senescence Profiling May Predict CAR T Responses
- Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
- Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
- Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
- Study Finds Influenza Often Undiagnosed in Winter Deaths
- Combined Screening Approach Identifies Early Leprosy Cases
- Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
- FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Channels
Clinical Chemistry
view channel
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read more
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read moreMolecular Diagnostics
view channel
Tumor Mutation Marker Helps Refine Lung Cancer Prognosis and Guide Therapy Selection
Lung cancer remains the leading cause of cancer death worldwide, while heterogeneous tumor genetics continue to complicate treatment decisions. Although molecular testing is increasingly used to match... Read more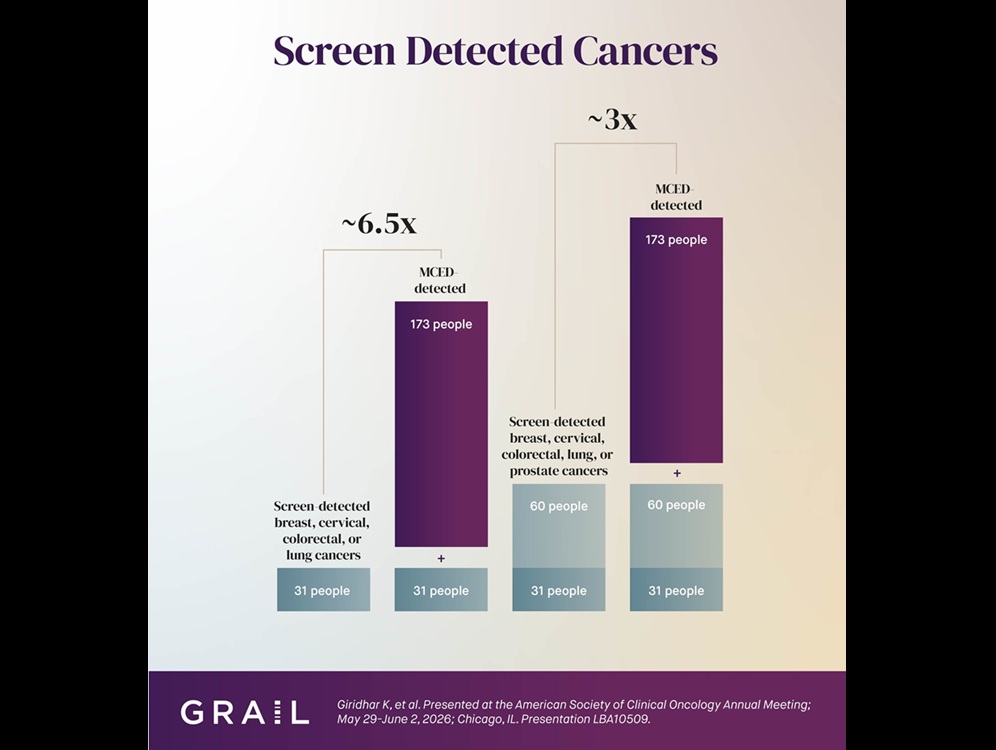
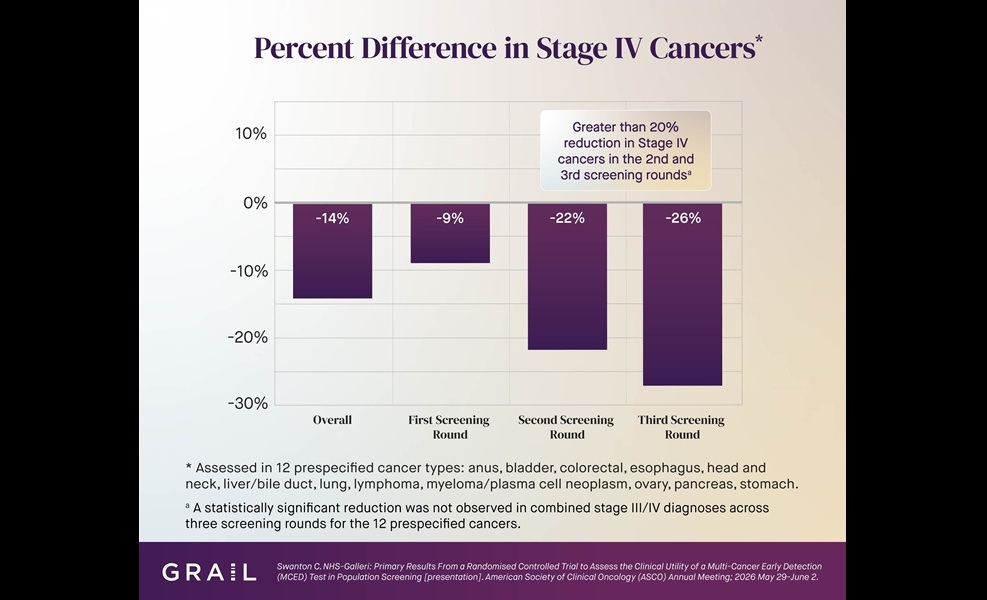
Multi-Cancer Test Boosts Detection When Added to Standard Screening
Most cancers are still diagnosed outside organ-specific screening, often at advanced stages. In the United States, guideline-recommended screening detects only 14% of cancers, underscoring the need for... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channelPartnership Expands Access to Alzheimer’s Blood Tests in Latin America and Caribbean
Alzheimer’s disease assessment remains challenging in many regions where aging populations are increasing demand for care, but access to dementia specialists and advanced imaging remains limited.... Read more




.jpg)