Thermo Fisher and Oncocyte to Jointly Develop IVD Test Kits and Companion Diagnostic Tests in Precision Oncology
|
By LabMedica International staff writers Posted on 20 Jan 2022 |

Oncocyte Corporation (Irvine, CA, USA) has entered into a development and co-marketing agreement for two distributed in vitro diagnostic (IVD) assays on Thermo Fisher Scientific’s (Waltham, MA, USA) Ion Torrent Genexus System. The agreement also grants Oncocyte rights to develop future companion diagnostics on the Genexus System.
The first-of-its-kind Genexus System is designed to be used in any lab and delivers comprehensive next-generation sequencing (NGS) results in as little as a single day – the same timeframe as single-gene tests, such as immunohistochemistry (IHC) assays. The Genexus system’s flexible batching makes it an attractive solution for hospital labs with low and medium testing volume by reducing the need to wait several days until a lab has a sufficient number of samples to test. The integrated purification system and sequencer enable sample preparation and processing with only 20 minutes of hands-on time and minimal user intervention, allowing for the democratization of NGS to community and regional cancer centers globally.
Under the terms of the collaboration, Oncocyte will clinically validate Thermo Fisher’s existing Oncomine Comprehensive Assay Plus on the Genexus System, paving the way toward IVD clearance for use in tumor profiling and future submissions as a companion diagnostic. As an IVD, the >500-gene assay will initially be able to provide physicians with information about patients’ tumors in accordance with established clinical evidence, applicable clinical trials, and future approval may assist with the selection of targeted therapies. Oncocyte will also develop its 27-gene expression DetermaIO test as a distributed kit on the Genexus. DetermaIO may predict response to immuno-oncology therapies based on data demonstrating potential pan-cancer utility and improvement over current standard-of-care tests.
“As many as 44% of newly diagnosed cancer patients may be eligible for immuno-oncology therapies, with additional patients potentially benefiting from other precision medicines, yet many patients’ tumors are never sequenced to determine if they may benefit from these targeted treatments,1” said Ron Andrews, President and Chief Executive Officer of Oncocyte. “In order to increase the number of patients benefiting from precision medicine, we need to expand the number of IVDs and develop these tests on instruments that are designed to make sequencing-based testing simple and more accessible. We have developed our Determa menu to ultimately become regulated kits for distribution ex-US on an IVD instrument. Our collaboration with Thermo Fisher using the Genexus System’s end-to-end automation enables that vision.”
“Leveraging Thermo Fisher’s proven global capabilities and installed base will enable us to expand the availability of IVD assays beyond the US. market. The collaboration with Thermo Fisher, along with the resulting IVD test kits and potential companion diagnostic tests that we will seek to develop and market together, will play a key role in helping us improve patient outcomes worldwide,” added Andrews.
“Genomic testing can have a dramatic impact on patient care, especially when results are available quickly to support early clinical decision-making,” said Garret Hampton, President, clinical next-generation sequencing and oncology at Thermo Fisher Scientific. “Today, access to comprehensive testing depends on where you are treated. Our goal is to democratize genomic profiling so it is available to patients right away in more clinical settings – ultimately, everywhere patients receive treatment. By partnering with Oncocyte to validate and co-develop new IVD assays, we are working to expand access to genomic profiling and spread the benefits of precision medicine to more patients.”
Related Links:
Thermo Fisher Scientific
Oncocyte Corporation
Latest Industry News
- CareDx Expands Precision Oncology Portfolio with Naveris Acquisition
- Thermo Fisher Scientific to Sell Microbiology Business to Astorg
- Collaboration Expands Access to Rapid Metagenomic Diagnostics for Complex Infections
- Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
- QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
- QIAGEN Showcases Integrated Sample-to-Insight Oncology Solutions at AACR 2026
- Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
- Partnership Expands Ultrasensitive WGS Assay for for Hematologic Malignancies and MRD Monitoring
- Beckman Coulter Gains CE Mark for Rapid Assay Distinguishing Bacterial vs Viral Infections
- Junction, GRAIL Enable Scalable Access to Multi-Cancer Detection Test
- GRAIL Partners with Epic to Integrate Multi-Cancer Test into EHR
- Global Partnership Aims to Streamline NGS Tumor Profiling in Oncology Trials
- Takara Bio USA and Hamilton Partner to Automate NGS Library Preparation
- Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
- Integrated DNA Technologies Expands into Clinical Diagnostics
- Co-Diagnostics Agreement Expands Commercial and Distribution Reach in South Asia
Channels
Clinical Chemistry
view channel
Blood Test Detects Testicular Cancer Missed by Standard Markers
Testicular cancer most often affects adolescents and young adults and is highly treatable when found early. Diagnosis can be difficult when tumors do not produce sufficient levels of standard blood-based... Read more
Routine Blood Tests Identify Biomarkers Linked to PTSD
Post-traumatic stress disorder (PTSD) is associated with a range of chronic physical health conditions and affects multiple organ systems. Clinical laboratories routinely measure blood analytes that reflect... Read moreMolecular Diagnostics
view channel
CRISPR-Based Test Identifies Multiple Respiratory Viruses Simultaneously
Respiratory virus co-circulation complicates differential diagnosis, as overlapping symptoms can obscure etiology. Multiplex testing typically depends on multiple enzymes or fluorophores and multistep... Read more
Multi-Omic Assay Predicts Recurrence and Radiation Benefit in Early Breast Cancer
Early-stage invasive breast cancer is frequently managed with breast-conserving surgery followed by adjuvant radiation therapy, but the magnitude of benefit from radiation varies among patients.... Read more
Portable Test Detects Tuberculosis from Tongue Swabs in 30 Minutes
Despite decades of effective drug regimens, tuberculosis remains the leading cause of death from an infectious disease, driven in part by limited access to accurate, rapid testing. Conventional diagnostics... Read more
Blood Test Receives FDA Breakthrough Status to Differentiate Schizophrenia and Bipolar Disorder
Differentiating schizophrenia from bipolar I disorder in symptomatic patients is challenging because early presentations often overlap. Current diagnostic pathways rely heavily on subjective assessments... Read moreHematology
view channel
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read more
Blood Test Enables Early Detection of Multiple Myeloma Relapse
Bone marrow biopsies remain central to diagnosing and monitoring multiple myeloma, yet the procedure is painful, invasive, and often repeated over time. Older patients—who represent most new cases—can... Read moreImmunology
view channel
Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
Sepsis remains a time-critical condition in which rapid risk assessment is often hindered by reliance on centralized laboratory testing. The global burden is substantial, with an estimated 166 million... Read more
Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
Accurate laboratory diagnosis of early Lyme disease remains challenging because serologic responses may be limited soon after infection. Missed detection at this stage can delay evaluation and management... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
Diabetic kidney disease is a leading cause of chronic kidney disease and end-stage kidney disease, affecting 20%–40% of people with diabetes and more than 107 million individuals worldwide as of 2021.... Read more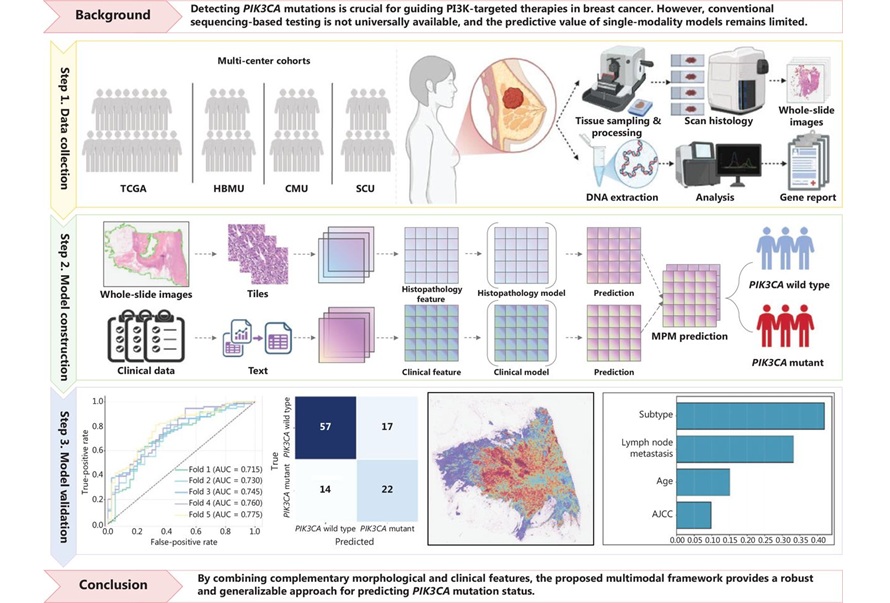
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read moreTechnology
view channel
Microfluidic Single-Cell Assay Predicts Breast Cancer Risk
Risk stratification for breast cancer remains imprecise, as population-based models and breast density can over- or underestimate individual risk, potentially leading to over- or under-screening.... Read more




.jpg)