Systematic Serological Screening Programme Used for Strongyloidiasis
|
By LabMedica International staff writers Posted on 13 Nov 2019 |
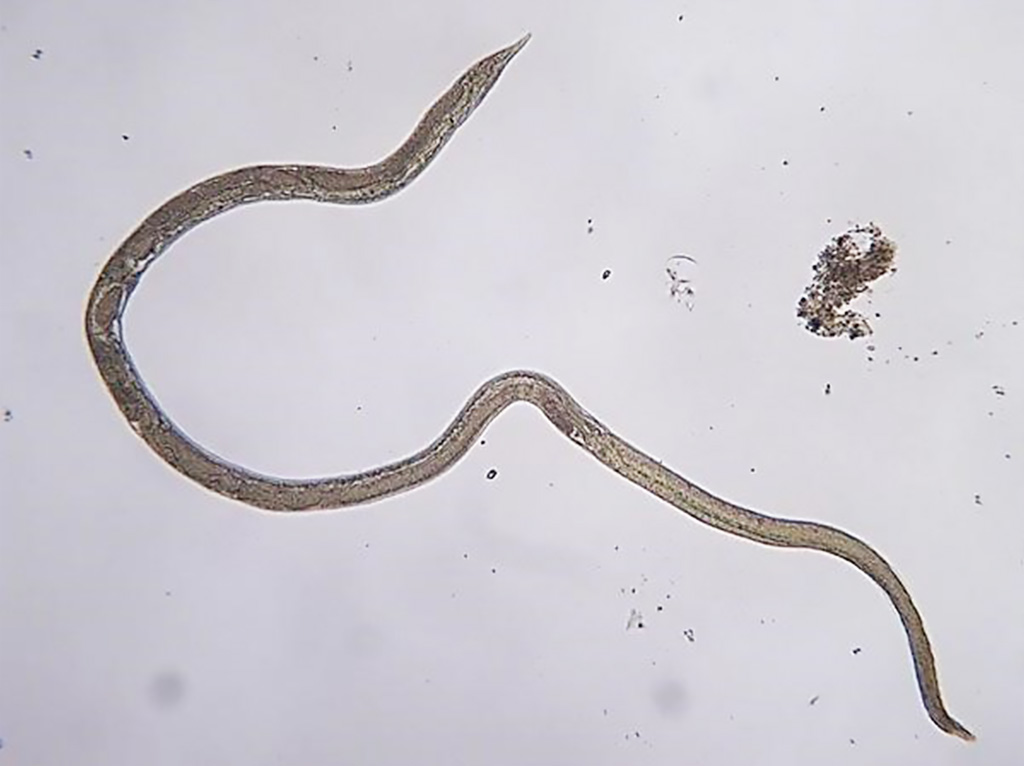
Image: Strongyloides stercoralis adult. The parasitic female lives threaded into the mucosal epithelium of the human small intestine (Photo courtesy of UVM Ecological Parasitology).
Strongyloidiasis is a soil-transmitted helminth infection caused by the nematode Strongyloides stercoralis and is endemic mainly in tropical and subtropical regions, but also in temperate regions.
This round worm infection is widely extended all over the world and it is estimated that at least 370 million individuals are infected worldwide, with a high degree of endemic burden in the tropical zones, especially Southeast Asia, Sub-Saharan Africa, and Latin America. As strongyloidiasis persists for life, it can cause serious morbidity or death long after an immigrant resettles in a new country.
Scientists at the Hospital de la Cruz Roja (Gijón, Spain) and their colleagues performed a prospective screening programme for strongyloidiasis between 2009 and 2014 for all immigrant patients attending the Tropical Medicine Unit. The patients were classified into seven groups according to their geographical area of origin (Central Africa, East Africa, West Africa, North Africa, Mexico and Central America, South America, and South Asia), following the US Centers for Disease Control and Prevention (CDC, Atlanta, GA, USA) criteria.
Three formalin-ether concentrated stool samples and an enzyme-linked immunosorbent assay (ELISA) for anti-S. stercoralis antibodies were used as the screening tools. The qualitative detection of IgG antibodies to S. stercoralis was done using an ELISA (DRG Diagnostics, Marburg, Germany) with microwells coated with Strongyloides antigen. A patient was considered as positive if at least one of the diagnostic tests used for screening (stool samples or/and ELISA) was positive. An eosinophil blood count was performed for all patients. Eosinophilia was defined as >500 eosinophils/mm3.
The team reported that of 659 patients screened, 79 (12%) were positive for S. stercoralis regardless of the diagnostic method used. The prevalence of infection was 42.9% in East African patients, 16.3% in Central African patients, 10.9% in those from South America, and 10% in the case of West Africa. Univariate analysis showed that infection by S. stercoralis was significantly more frequent in patients from Central Africa, and East Africa. Seventy-nine patients had a positive serological test for S. stercoralis; microscopic visualization of larvae of S. stercoralis by formalin-ether concentration of feces was positive for only four of them.
The authors recommended that immigrant patients from developing countries be routinely screened for S. stercoralis, especially those from East Africa. Due to the low sensitivity of stool examination for ova and parasites arising from low larval burden and intermittent shedding in the stool, serological testing is the diagnostic method of choice for screening in immigrant populations. The study was published in the November, 2019 issue of the International Journal of Infectious Diseases.
Related Links:
Hospital de la Cruz Roja
US Centers for Disease Control and Prevention
DRG Diagnostics
This round worm infection is widely extended all over the world and it is estimated that at least 370 million individuals are infected worldwide, with a high degree of endemic burden in the tropical zones, especially Southeast Asia, Sub-Saharan Africa, and Latin America. As strongyloidiasis persists for life, it can cause serious morbidity or death long after an immigrant resettles in a new country.
Scientists at the Hospital de la Cruz Roja (Gijón, Spain) and their colleagues performed a prospective screening programme for strongyloidiasis between 2009 and 2014 for all immigrant patients attending the Tropical Medicine Unit. The patients were classified into seven groups according to their geographical area of origin (Central Africa, East Africa, West Africa, North Africa, Mexico and Central America, South America, and South Asia), following the US Centers for Disease Control and Prevention (CDC, Atlanta, GA, USA) criteria.
Three formalin-ether concentrated stool samples and an enzyme-linked immunosorbent assay (ELISA) for anti-S. stercoralis antibodies were used as the screening tools. The qualitative detection of IgG antibodies to S. stercoralis was done using an ELISA (DRG Diagnostics, Marburg, Germany) with microwells coated with Strongyloides antigen. A patient was considered as positive if at least one of the diagnostic tests used for screening (stool samples or/and ELISA) was positive. An eosinophil blood count was performed for all patients. Eosinophilia was defined as >500 eosinophils/mm3.
The team reported that of 659 patients screened, 79 (12%) were positive for S. stercoralis regardless of the diagnostic method used. The prevalence of infection was 42.9% in East African patients, 16.3% in Central African patients, 10.9% in those from South America, and 10% in the case of West Africa. Univariate analysis showed that infection by S. stercoralis was significantly more frequent in patients from Central Africa, and East Africa. Seventy-nine patients had a positive serological test for S. stercoralis; microscopic visualization of larvae of S. stercoralis by formalin-ether concentration of feces was positive for only four of them.
The authors recommended that immigrant patients from developing countries be routinely screened for S. stercoralis, especially those from East Africa. Due to the low sensitivity of stool examination for ova and parasites arising from low larval burden and intermittent shedding in the stool, serological testing is the diagnostic method of choice for screening in immigrant populations. The study was published in the November, 2019 issue of the International Journal of Infectious Diseases.
Related Links:
Hospital de la Cruz Roja
US Centers for Disease Control and Prevention
DRG Diagnostics
Latest Immunology News
- FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
- Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
- Cancer Mutation ‘Fingerprints’ to Improve Prediction of Immunotherapy Response
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Blood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
- Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
- Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
- Blood Test Could Identify Colon Cancer Patients to Benefit from NSAIDs
- Blood Test Could Detect Adverse Immunotherapy Effects
- Routine Blood Test Can Predict Who Benefits Most from CAR T-Cell Therapy
- New Test Distinguishes Vaccine-Induced False Positives from Active HIV Infection
- Gene Signature Test Predicts Response to Key Breast Cancer Treatment
- Chip Captures Cancer Cells from Blood to Help Select Right Breast Cancer Treatment
- Blood-Based Liquid Biopsy Model Analyzes Immunotherapy Effectiveness
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreMolecular Diagnostics
view channel
First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
Accurate identification of clonal immunoglobulin heavy chain (IGH) gene rearrangements is central to evaluating suspected B-cell lymphoproliferative disorders, where a single B-cell clone yields a defining... Read more
Plasma ctDNA Testing Predicts Breast Cancer Recurrence After Neoadjuvant Therapy
Accurate identification of breast cancer patients at risk of relapse after pre-surgery treatment is central to guiding adjuvant decisions, particularly in aggressive disease. Circulating fragments of tumor... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreMicrobiology
view channel
WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Tuberculosis (TB) remains a leading infectious killer, with more than 3,300 deaths and 29,000 new illnesses every day. Diagnostic delays and dependence on centralized laboratory networks continue to impede... Read more
Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Accurate and rapid identification of bacterial infections remains challenging in acute care, where delays can hinder timely, targeted therapy. Infectious diseases are a major cause of mortality worldwide,... Read more
Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
Direct-to-consumer gut microbiome kits promise personalized insights by profiling fecal bacteria and generating health readouts, but their analytical accuracy remains uncertain. A new study shows that... Read more
WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
Tuberculosis (TB) remains one of the world’s leading infectious disease killers, yet millions of cases go undiagnosed or are detected too late. Barriers such as reliance on sputum samples, limited laboratory... Read morePathology
view channel
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read more
AI-Powered Tool to Transform Dermatopathology Workflow
Skin cancer accounts for the largest number of cancer diagnoses in the United States, placing sustained pressure on pathology services. Diagnostic interpretation can be variable for challenging melanocytic... Read moreTechnology
view channel
Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
Chemotherapy selection for breast cancer is challenged by heterogeneous tumor responses. Conventional chemosensitivity assays can be slow, require large sample volumes, and struggle with complex biological... Read more
Online Tool Supports Family Screening for Inherited Cancer Risk
Genetic test results in oncology often have implications for relatives who may share inherited cancer risk. Many health systems lack structured processes to help patients alert family members, limiting... Read moreIndustry
view channel
Integrated DNA Technologies Expands into Clinical Diagnostics
Integrated DNA Technologies (IDT; Coralville, Iowa, USA) has announced the launch of Archer FUSIONPlex-HT Dx and VARIANTPlex-HT Dx. This launch marks the company’s first in vitro diagnostic (IVD) offerings... Read more