Leprosy Neural Impairment Diagnosis Evaluated by Tests
|
By LabMedica International staff writers Posted on 07 Jun 2018 |

Image: The ABI 7300 real-time PCR system (Photo courtesy of Applied Biosystems).
Leprosy is a chronic infectious disease caused by Mycobacterium leprae, an obligate intracellular parasite with a predilection for infecting peripheral nerves and skin. Leprosy is a current and challenging disease, because it still represents a problem for public health in developing countries.
Despite the apparent progress observed in recent years in leprosy control, early identification of cases remains one of the primary objectives of control programs. The long incubation period of leprosy, its insidious symptoms and signs make the diagnosis difficult. The predominance of multibacillary (MB) cases with neural disabilities indicates late diagnosis, reinforcing the ineffective epidemiological control in many countries.
Scientists at the Federal University of Uberlândia (UFU, Uberlândia, Brazil) and their colleagues recruited 175 seropositive and 35 seronegative household contacts from 2014 to 2016, who were subjected to an extensive protocol that included clinical, molecular (peripheral blood quantitative polymerase chain reaction (qPCR), slit-skin smear qPCR, skin biopsy qPCR) and electroneuromyographic evaluations. Enzyme-linked immunosorbent assay (ELISA) was performed on all household contacts. Serum anti-phenolic glycolipid I (PGL-I) IgM antibodies were detected by enzyme-linked immunosorbent assay (ELISA) performed against the purified native PGL-I from the M. leprae cell wall. The quantitative real-time PCR (qPCR) assay targeting M. leprae DNA was performed by targeting the bacillus-specific genomic region (RLEP) in a real-time PCR system.
The investigators found that the positivity of peripheral blood qPCR of seropositive contacts was 40.6% (71/175) whereas only 8.6% (3/35) were qPCR positive in seronegative contacts. For the slit-skin smear, only 4% (7/175) of seropositive contacts presented positive bacilloscopy, whereas the qPCR detected 47.4% (83/175) positivity in this group compared with only 17.1% (6/35) in seronegative contacts. Seropositive contacts presented a 4.04-fold higher chance of neural impairment. The peripheral blood qPCR positivity presented odds of 2.08-fold higher towards neural impairment. Contrarily, the presence of at least one BCG vaccine scar demonstrated 2.44-fold greater protection against neural impairment (OR = 0.41).
The authors concluded that ELISA anti-PGL-I is the most important test in determining the increased chance of neural impairment in asymptomatic leprosy household contacts. The combination of the two assays (ELISA anti-PGL-I and peripheral blood qPCR) and the presence of BCG scar may identify individuals with higher chances of developing leprosy neuropathy corroborating with the early diagnosis and treatment. The study was published on May 21, 2018, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Federal University of Uberlândia
Despite the apparent progress observed in recent years in leprosy control, early identification of cases remains one of the primary objectives of control programs. The long incubation period of leprosy, its insidious symptoms and signs make the diagnosis difficult. The predominance of multibacillary (MB) cases with neural disabilities indicates late diagnosis, reinforcing the ineffective epidemiological control in many countries.
Scientists at the Federal University of Uberlândia (UFU, Uberlândia, Brazil) and their colleagues recruited 175 seropositive and 35 seronegative household contacts from 2014 to 2016, who were subjected to an extensive protocol that included clinical, molecular (peripheral blood quantitative polymerase chain reaction (qPCR), slit-skin smear qPCR, skin biopsy qPCR) and electroneuromyographic evaluations. Enzyme-linked immunosorbent assay (ELISA) was performed on all household contacts. Serum anti-phenolic glycolipid I (PGL-I) IgM antibodies were detected by enzyme-linked immunosorbent assay (ELISA) performed against the purified native PGL-I from the M. leprae cell wall. The quantitative real-time PCR (qPCR) assay targeting M. leprae DNA was performed by targeting the bacillus-specific genomic region (RLEP) in a real-time PCR system.
The investigators found that the positivity of peripheral blood qPCR of seropositive contacts was 40.6% (71/175) whereas only 8.6% (3/35) were qPCR positive in seronegative contacts. For the slit-skin smear, only 4% (7/175) of seropositive contacts presented positive bacilloscopy, whereas the qPCR detected 47.4% (83/175) positivity in this group compared with only 17.1% (6/35) in seronegative contacts. Seropositive contacts presented a 4.04-fold higher chance of neural impairment. The peripheral blood qPCR positivity presented odds of 2.08-fold higher towards neural impairment. Contrarily, the presence of at least one BCG vaccine scar demonstrated 2.44-fold greater protection against neural impairment (OR = 0.41).
The authors concluded that ELISA anti-PGL-I is the most important test in determining the increased chance of neural impairment in asymptomatic leprosy household contacts. The combination of the two assays (ELISA anti-PGL-I and peripheral blood qPCR) and the presence of BCG scar may identify individuals with higher chances of developing leprosy neuropathy corroborating with the early diagnosis and treatment. The study was published on May 21, 2018, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Federal University of Uberlândia
Latest Immunology News
- Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
- FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
- Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
- Cancer Mutation ‘Fingerprints’ to Improve Prediction of Immunotherapy Response
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Blood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
- Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
- Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
- Blood Test Could Identify Colon Cancer Patients to Benefit from NSAIDs
- Blood Test Could Detect Adverse Immunotherapy Effects
- Routine Blood Test Can Predict Who Benefits Most from CAR T-Cell Therapy
- New Test Distinguishes Vaccine-Induced False Positives from Active HIV Infection
- Gene Signature Test Predicts Response to Key Breast Cancer Treatment
- Chip Captures Cancer Cells from Blood to Help Select Right Breast Cancer Treatment
Channels
Clinical Chemistry
view channel
AI-Based Blood Test Diagnose Multiple Brain Disorders from Blood Sample
Diagnosing the cause of age-related cognitive symptoms remains challenging because clinical presentations of neurodegenerative diseases often overlap, and multiple pathologies can co-occur... Read more
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read moreMolecular Diagnostics
view channel
RNA Profiling Uncovers Therapeutic Targets in Solid Tumors
Many patients with advanced solid tumors exhaust broad DNA panel testing yet still lack biomarkers that match guideline-recommended therapies, limiting access to targeted options. Expanding molecular profiling... Read more
Whole Genome Sequencing in Routine Care Expands Rare Disease Detection
Rare diseases often involve prolonged diagnostic journeys that delay clinical decision-making and complicate family planning. As phenotypes become more heterogeneous, sequencing-based methods are increasingly... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
Active tuberculosis (TB) remains a leading cause of death and illness worldwide, yet distinguishing contagious disease from latent infection continues to challenge clinicians. Standard screening tools... Read more
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read morePathology
view channel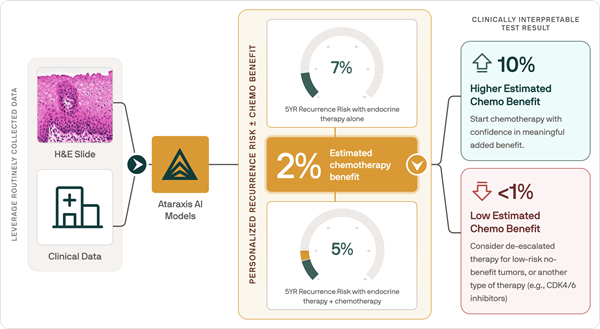
AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer is typically guided by recurrence risk and population-level averages rather than patient-specific benefit. However, existing clinicopathologic... Read more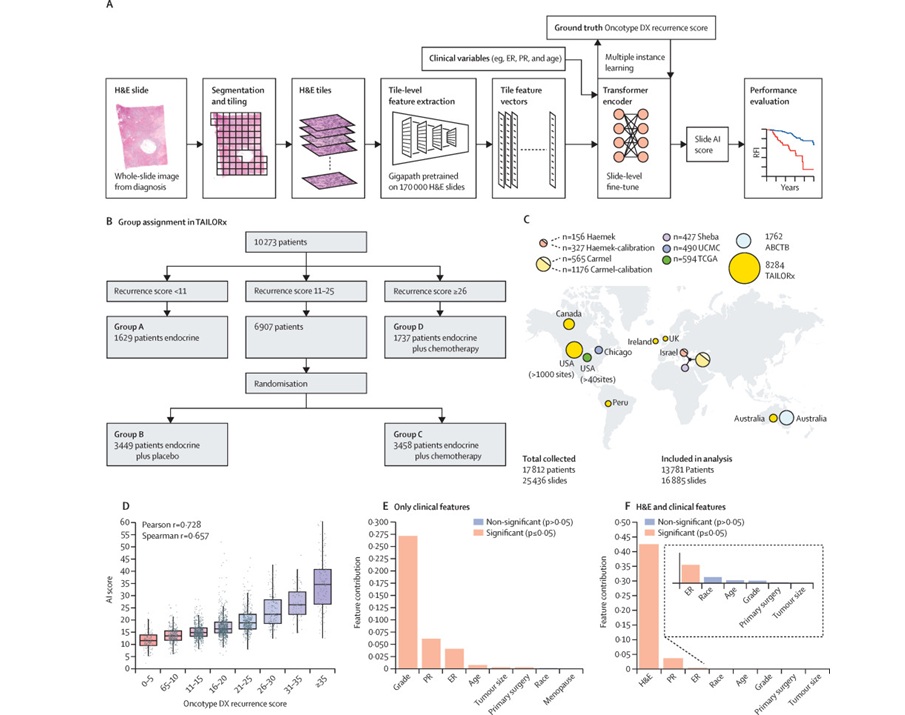
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Takara Bio USA and Hamilton Partner Partner to Automate NGS Library Preparation
Takara Bio USA, Inc. (San Jose, CA, USA), a wholly owned subsidiary of Takara Bio Inc., and Hamilton Company (Reno, NV, USA) announced a development and co-marketing agreement to deliver integrated, automated... Read more