Developing POC Tests for Alzheimer’s to Improve Monitoring and Management of the Disease
|
By LabMedica International staff writers Posted on 22 Mar 2016 |
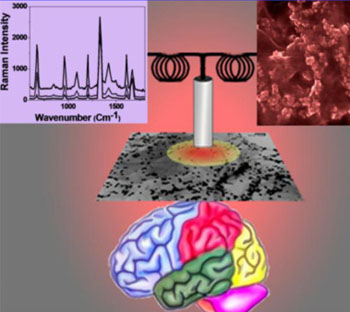
Image: Illustration of a protocol showing a pathway to develop core-shell nanoparticle/hybrid graphene oxide based multi-functional platform label-free SERS detection of β-amyloid toward developing a portable point-of-care blood test to monitor Alzheimer’s disease progression (Figure courtesy of Teresa Demeritt et al., 2015, ACS Appl. Mater. Interfaces; Copyright ACS-2015).
Toward development of a portable point-of-care (POC) biosensor for Alzheimer’s disease (AD), a new review explores recent advancements in nano-enabling electrochemical beta-amyloid (β-A) -sensing technologies. A simple, rapid POC biomarker test could greatly improve AD management and personalized treatment, also in developing countries.
The authors of the review, from the College of Medicine at Florida International University (Miami, FL, USA), are taking a new approach to diagnosing AD: measuring β-A in the blood with a POC test. AD is caused by high levels of β-A in the brain that lead to degeneration of brain cells. Various types of scans and immunoassays, such as MRI and ELISA, are available to estimate β-A levels in the brain. But the peptide can also be found at lower levels in blood, making it a useful biomarker for a simple test.
Currently there is no sensitive or inexpensive way to measure β-A levels in blood samples. The authors of the new review plan to change that. “We want to develop a point-of-care system where a small drop of blood plasma can reveal their β-A level immediately so that a doctor can tailor a patient’s therapy immediately,” said lead author Dr. Ajeet Kaushik, “The drugs used to treat AD can have side effects, so it’s better for patients not to overdose. With the right data, doctors can respond quickly to changes in a patient’s brain by reducing or increasing their dose.”
In the review, Dr. Kaushik and colleagues looked at each of the methods available to measure β-A concentration in brain tissue and in blood. None of the existing tests can be done at the bedside and all need special expertise and large samples. They also take a long time to generate a useful result—the main existing test, an ELISA, takes 6–8 hours. In comparison, the cheap, simple biosensor Dr. Kaushik and colleagues describe can measure β-A in the blood at low (pico molar) concentrations in just 30 minutes.
“Even though existing technologies are well established, we need to move towards small sample, high accuracy tests that can be used in all environments, from developed countries to rural settings. Our goal is to develop a test that’s sensitive, small, and affordable,” said Dr. Kaushik. To develop the new biosensor, the team will need many bio-fluid samples taken at different stages of the disease. Finding these samples will be challenging, but the review demonstrates that a biosensor is achievable. Such a test would also “show if and when the disease reaches an untreatable level. In the future we hope a rapid biosensor test for AD will help scientists study disease progression and help clinicians deliver personalized therapy to patients.”
The study, by Kaushik A et al., was published online ahead of print January 28, 2016, in the journal Biosensors and Bioelectronics.
Related Links:
Florida International University
The authors of the review, from the College of Medicine at Florida International University (Miami, FL, USA), are taking a new approach to diagnosing AD: measuring β-A in the blood with a POC test. AD is caused by high levels of β-A in the brain that lead to degeneration of brain cells. Various types of scans and immunoassays, such as MRI and ELISA, are available to estimate β-A levels in the brain. But the peptide can also be found at lower levels in blood, making it a useful biomarker for a simple test.
Currently there is no sensitive or inexpensive way to measure β-A levels in blood samples. The authors of the new review plan to change that. “We want to develop a point-of-care system where a small drop of blood plasma can reveal their β-A level immediately so that a doctor can tailor a patient’s therapy immediately,” said lead author Dr. Ajeet Kaushik, “The drugs used to treat AD can have side effects, so it’s better for patients not to overdose. With the right data, doctors can respond quickly to changes in a patient’s brain by reducing or increasing their dose.”
In the review, Dr. Kaushik and colleagues looked at each of the methods available to measure β-A concentration in brain tissue and in blood. None of the existing tests can be done at the bedside and all need special expertise and large samples. They also take a long time to generate a useful result—the main existing test, an ELISA, takes 6–8 hours. In comparison, the cheap, simple biosensor Dr. Kaushik and colleagues describe can measure β-A in the blood at low (pico molar) concentrations in just 30 minutes.
“Even though existing technologies are well established, we need to move towards small sample, high accuracy tests that can be used in all environments, from developed countries to rural settings. Our goal is to develop a test that’s sensitive, small, and affordable,” said Dr. Kaushik. To develop the new biosensor, the team will need many bio-fluid samples taken at different stages of the disease. Finding these samples will be challenging, but the review demonstrates that a biosensor is achievable. Such a test would also “show if and when the disease reaches an untreatable level. In the future we hope a rapid biosensor test for AD will help scientists study disease progression and help clinicians deliver personalized therapy to patients.”
The study, by Kaushik A et al., was published online ahead of print January 28, 2016, in the journal Biosensors and Bioelectronics.
Related Links:
Florida International University
Latest Pathology News
- AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
- Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
- AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
- FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
- New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
- AI System Analyzes Routine Pathology Slides to Predict Cancer Outcomes
- New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
- Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
- Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
- Tumor Immune Structure Predicts Response to Immunotherapy in Melanoma
- Plug-and-Play AI Pathology System Classifies Multiple Cancers from Few Slides
- AI-Based Assays Support Risk Stratification in Prostate and Breast Cancer
- AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
- Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
- AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
- Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
Channels
Clinical Chemistry
view channel
Fluid Biomarker Improves Diagnosis and Monitoring of Primary CNS Lymphoma
Primary central nervous system lymphoma (PCNSL) is a rare malignancy of the brain, spinal cord, and eyes with delayed diagnosis and poor outcomes. Current fluid-based testing using interleukin measurements... Read more
New CA19-9 Cutoff Value Helps Identify High-Risk Pancreatic Cancer Patients
Pancreatic ductal adenocarcinoma (PDAC) is frequently diagnosed at an advanced stage and remains one of the most lethal solid tumors. Clinicians commonly use serum carbohydrate antigen 19-9 (CA19-9) to... Read moreMolecular Diagnostics
view channel
Gene Fusion Patterns May Flag High Risk Solitary Fibrous Tumors
Solitary fibrous tumor (SFT) is a rare subtype of sarcoma, a cancer that develops in connective tissues such as fat, muscle, blood vessels, and fibrous tissue. SFTs most commonly arise in the chest cavity... Read more
New RNA Origami Method Supports Faster Targeted Testing for Repeat Expansion Disorders
Repeat expansion disorders drive conditions such as myotonic dystrophy, Huntington’s disease, and amyotrophic lateral sclerosis (ALS), yet accurately sizing the mutated sequences remains difficult.... Read moreHematology
view channel
Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
Iron deficiency in school-age children can affect brain development, learning, growth, and physical performance, yet early deficiency may be missed when screening focuses mainly on anemia.... Read more
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read moreMicrobiology
view channel
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read more
New AMR Assay Supports Rapid Infection Control Screening in Hospitals
As antimicrobial resistance spreads worldwide, healthcare-associated infections are placing a growing burden on hospitals, increasing the need for faster and broader diagnostic solutions.... Read morePathology
view channel
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read more
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read moreTechnology
view channel
AI-Enabled Assistant Unifies Molecular Workflow Planning and Support
Clinical laboratories and research groups face increasingly complex molecular workflows and expanding technical documentation spread across multiple systems. Fragmented digital tools can slow experiment... Read more
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read moreIndustry
view channel
Natera to Present Data on MRD-Guided Cancer Care at ASCO 2026
Natera, Inc. (Austin, TX, USA), a company focused on cell-free DNA testing and precision medicine, announced an oncology data program for the 2026 American Society of Clinical Oncology (ASCO) Annual Meeting,... Read more