Multiplex Immunoassays Enable Quantification of Malaria Antigens
|
By LabMedica International staff writers Posted on 16 Jun 2022 |
The standard of care for malaria diagnosis is blood smear microscopy and antigen detection through rapid diagnostic test (RDT). Microscopy has limitations in terms of difficulty in identifying mixed infections and RDTs are more amenable for the diagnosis of malaria in settings with limited laboratory infrastructure.
Immunoassay platforms that simultaneously detect malaria antigens including histidine-rich protein 2 (HRP2)/HRP3 and Plasmodium lactate dehydrogenase (pLDH), are useful epidemiological tools for rapid diagnostic test evaluation. The gold standard for malaria detection is confirmation of the presence of parasite DNA or RNA in whole blood by polymerase chain reaction (PCR) testing.
A team of medical scientists led by those at the Diagnostic Group, PATH (Seattle, WA, USA) studied the comparative evaluation of two multiplex platforms in identifying Plasmodium falciparum with presence or absence of HRP2/HRP3 expression as being indicative of hrp2/hrp3 deletions and other Plasmodium species.
The team used a 77-member panel of specimens composed of the World Health Organization (WHO, Geneva, Switzerland) international Plasmodium antigen standards, cultured parasites for P. falciparum and Plasmodium knowlesi, and clinical specimens with mono-infections for P. falciparum, Plasmodium vivax, and Plasmodium malariae was generated as both whole blood and dried blood spot (DBS) specimens.
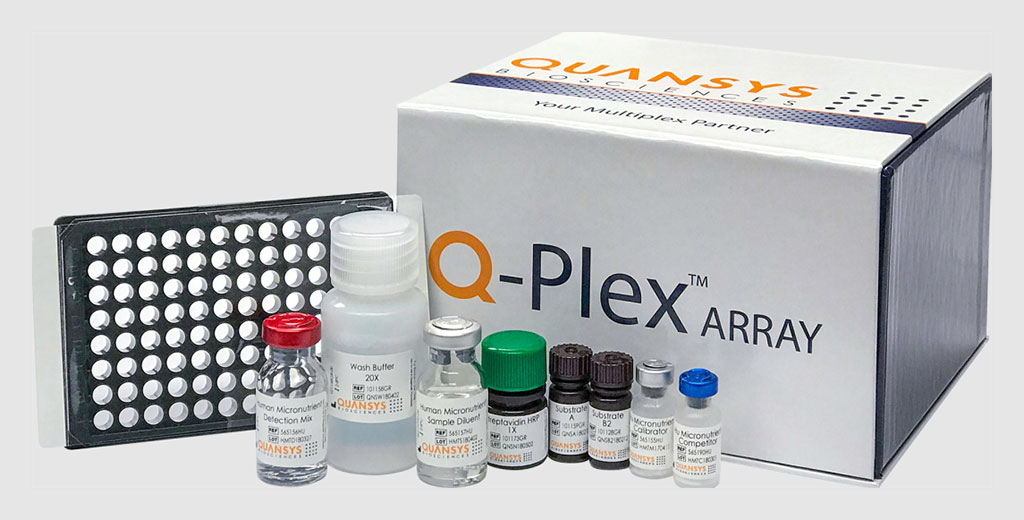
Assays for HRP2, P. falciparum–specific pLDH (PfLDH), P. vivax–specific pLDH (PvLDH), and all human Plasmodium species Pan malaria pLDH (PanLDH) on the Human Malaria Array Q-Plex (Quansys Biosciences, Logan, UT, USA) and the xMAP platforms (Luminex, Austin, TX, USA) were evaluated with these panels.
The investigators reported that the xMAP showed a higher percent positive agreement for identification of hrp2-deleted P. falciparum and Plasmodium species in whole blood and DBS than the Q-Plex. For whole blood samples, there was a highly positive correlation between the two platforms for PfLDH and PvLDH, moderate positive correlation for HRP2, and poor correlation for PanLDH. The xMAP HRP2 assay appeared to cross-react with HRP3, while the Q-Plex did not. The Q-Plex PfLDH assay cross-reacted with P. malariae, while the xMAP did not. For both platforms, P. knowlesi was detected on the PvLDH assay. The WHO international standards allowed normalization across both platforms on their HRP2, PfLDH, and PvLDH assays in whole blood and DBS.
The authors concluded that Q-Plex and xMAP show good agreement for identification of P. falciparum mutants with hrp2/hrp3 deletions, and other Plasmodium species. Quantitative results from both platforms, normalized into international units for HRP2, PfLDH, and PvLDH, showed good agreement and should allow comparison and analysis of results generated by either platform. The study was published on June 7, 2022 in the Malaria Journal.
Related Links:
Diagnostic Group, PATH
World Health Organization
Quansys Biosciences
Luminex
Latest Technology News
- New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
- Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
- Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
- Online Tool Supports Family Screening for Inherited Cancer Risk
- Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
- New Electronic Pipette Enhances Workflows with Touchscreen Control
- AI Model Outperforms Clinicians in Rare Disease Detection
- AI-Driven Diagnostic Demonstrates High Accuracy in Detecting Periprosthetic Joint Infection
- Blood Test “Clocks” Predict Start of Alzheimer’s Symptoms
- AI-Powered Biomarker Predicts Liver Cancer Risk
- Robotic Technology Unveiled for Automated Diagnostic Blood Draws
- ADLM Launches First-of-Its-Kind Data Science Program for Laboratory Medicine Professionals
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreMolecular Diagnostics
view channel
New Molecular Test Boosts Accuracy of Bile Duct Cancer Diagnosis
Bile duct strictures can arise from cancer or benign disease, but their location within ducts connecting the liver, gallbladder, and intestines complicates evaluation. Standard biopsy and cytology may... Read moreAdaptive PCR Platform Improves Consistency in Small-Batch NGS Workflows
PCR amplification during next-generation sequencing (NGS) library preparation can introduce variability, often requiring manual quantification and risking over-cycling artifacts. The issue is especially... Read more
First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
Accurate identification of clonal immunoglobulin heavy chain (IGH) gene rearrangements is central to evaluating suspected B-cell lymphoproliferative disorders, where a single B-cell clone yields a defining... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read morePathology
view channel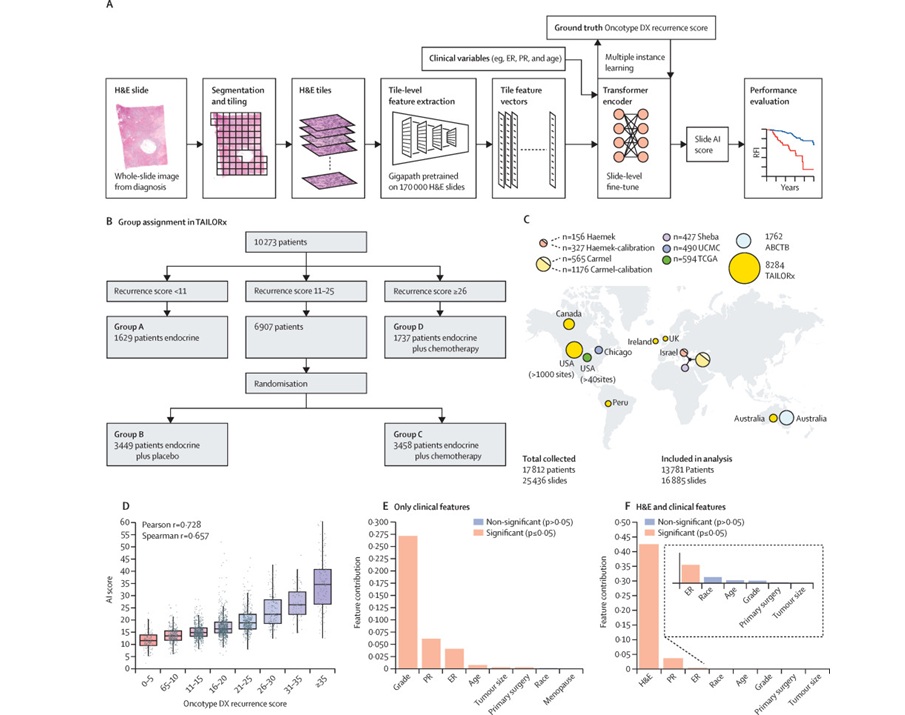
AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
Selecting adjuvant chemotherapy for early-stage breast cancer remains a difficult decision because only a subset benefits and many undergo toxicity without gain. Genomic assays can help but are costly,... Read more
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read moreTechnology
view channel
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Lunit and CellCarta Collaborate to Expand AI Pathology in CDx Development
Lunit (Seoul, South Korea), a leading provider of AI for cancer diagnostics and precision oncology, and CellCarta (Montreal, QC, Canada), a global contract research organization (CRO) laboratory serving... Read more
























