Urine Test Uses Tiny Worms to Detect Early Signs of Cancer
|
By LabMedica International staff writers Posted on 24 Nov 2021 |
Scientists have developed a screening test that uses tiny worms to detect early signs of cancer in urine that could help boost routine screening.
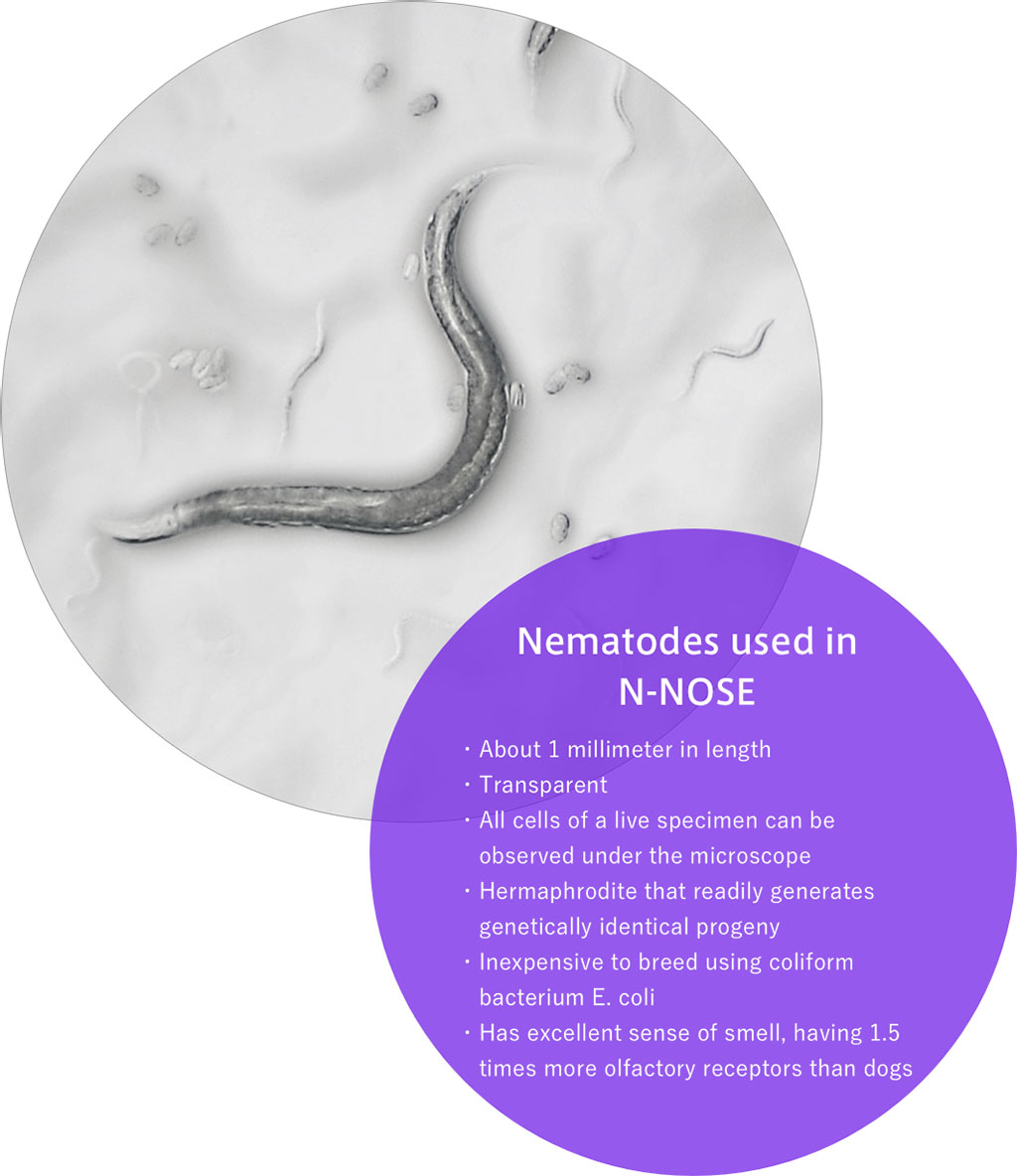
Scientists at Hirotsu Bio Science (Tokyo, Japan) have developed N-NOSE, a cancer screening test that uses highly sensitive olfactory sensory functions of genetically modified a type of worm called "C. elegans" (nematodes) with an acute sense of smell. N-NOSE, which stands for Nematode Nose, uses the chemotaxis characteristic of nematodes, i.e., attractive responses to an odor it likes and repulsive responses to an odor it does not like, as an indicator. Nematodes are known to respond to 15 types of cancer of the stomach, colorectal, lung, breast, pancreatic, liver, prostate, uterine, esophageal, gall bladder, bile duct, kidney, bladder, ovarian, and oral/pharyngeal.
C. elegans nematode is the first multicellular organism to have its entire genome sequenced and is a very popular model organism in biological research. The idea of using nematodes, live creatures, in cancer testing stems from the fact that they have superior olfactory sensors that no man-made device can come close to. Because it has no eyes or ears, it relies on smell to navigate its environment and detect food. C. elegans therefore has a well-developed sense of smell and about 1,200 types of olfactory receptor genes that are able to detect minute odors that machines cannot detect. The novel N-NOSE test that employs the worm’s natural capabilities is not invasive, causes no pain and can be used for early detection of cancer.
The test requires a small amount of urine and is inexpensive since nematodes are inexpensive to breed. Each nematode lays as many as 300 eggs and feeds on coliform bacterium E. coli, making it inexpensive to breed C. elegans populations. Currently available cancer diagnostic tests often do not detect incipient cancer and it has been thought very difficult to know the existence of cancer at an early stage. However, nematodes respond to the minute odor of cancer, which is undetectable with existing tests, and thus make it possible to predict risks. Early detection of cancers as early as Stages 0 and 1 is possible with N-NOSE according to clinical studies, and one test is able to detect cancers regardless of their locations. The simple, painless urine test is suitable as a cancer test for all generations, including children. The company believes that in future, children will take regular cancer screening tests to detect pre-symptomatic tumors.
Related Links:
Hirotsu Bio Science
Latest Clinical Chem. News
- International Experts Recommend Ending Routine 'Corrected' Calcium Reporting
- Long-Term Data Show PSA Screening Modestly Reduces Prostate Cancer Deaths
- Urine-Based Nanosensor Tracks Lung Cancer and Fibrosis Noninvasively
- FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
- CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
- Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
- Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
- At-Home Blood and Cognitive Tests Support Dementia Risk Stratification
- Ultrasensitive Test Detects Key Biomarker of Frontotemporal Dementia Subtype
- Routine Blood Tests Years Before Pregnancy Could Identify Preeclampsia Risk
- Blood Test Detects Testicular Cancer Missed by Standard Markers
- Routine Blood Tests Identify Biomarkers Linked to PTSD
- Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
- Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
- Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
- Rapid Biosensor Detects Pancreatic Cancer Biomarker for Early Detection
Channels
Molecular Diagnostics
view channel
Multi-Omics Profiling Helps Predict BCG Response and Recurrence in Bladder Cancer
High-risk non–muscle-invasive bladder cancer frequently recurs after therapy, with about 30% of patients relapsing and roughly 10% dying within two years despite tumor resection, surveillance, and Bacillus... Read more
New Computational Tool Reveals Genetic Driver of Idiopathic Neuropathy
Peripheral neuropathy is a common neurological disorder that causes pain, sensory loss, imbalance, and weakness, affecting an estimated 12%–20% of people in the U.S. and nearly 30% of adults over age 65.... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read morePathology
view channel
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read more
AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
Non–muscle invasive bladder cancer has highly variable outcomes, complicating surveillance and treatment planning. Risk assessment typically relies on stage, grade, and tumor size, leaving uncertainty... Read moreTechnology
view channel
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read more
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read moreIndustry
view channel
Partnership Brings Single-Cell Analysis into Clinical Oncology Workflows
Selecting treatments for advanced cancer remains difficult when bulk analyses mask the functional diversity of tumor cells and mechanisms of resistance that emerge over time. Clinicians increasingly need... Read more