Hodgkin Lymphoma Therapy Response Linked to T-Cell Receptor Repertoire
|
By LabMedica International staff writers Posted on 27 Aug 2020 |
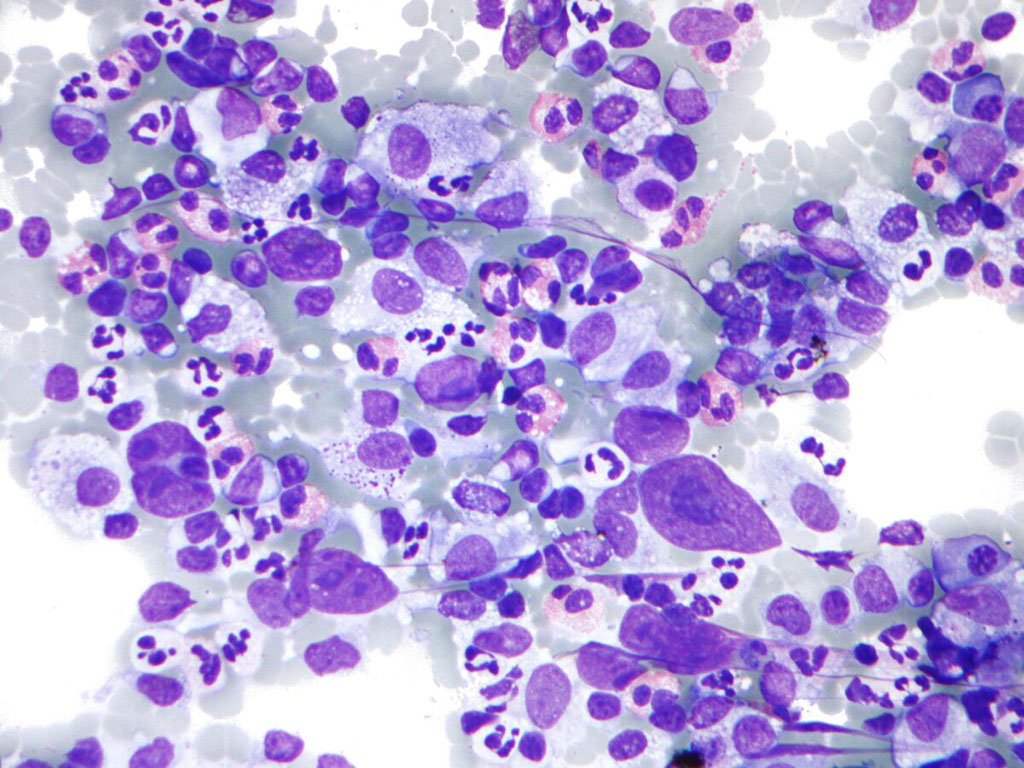
Image: Photomicrograph of Hodgkin lymphoma (HL), from a fine needle aspiration of a lymph node, which shows a mixture of cells common in HL: Eosinophils, Reed-Sternberg cells, Plasma cells, and Histocytes (Photo courtesy of Nephron).
Hodgkin lymphoma (HL) is a type of lymphoma in which cancer originates from a specific type of white blood cells called lymphocytes. Symptoms may include fever, night sweats, and weight loss. Often there will be non-painful enlarged lymph nodes in the neck, under the arm, or in the groin.
Hodgkin lymphoma may be treated with chemotherapy, radiation therapy, and stem cell transplant. The choice of treatment often depends on how advanced the cancer has become and whether or not it has favorable features. In early disease, a cure is often possible. PD-1 blockade is highly effective in classical Hodgkin lymphomas (cHLs), which exhibit frequent copy-number gains of CD274 (PD-L1) and PDC1LG2 (PD-L2) on chromosome 9p24.1.
A team of medical oncologists collaborating with the Dana-Farber Cancer Institute (Boston, MA, USA) used T cell receptor (TCR) sequencing and cytometry by time-of-flight (CyTOF) analysis to obtain a peripheral immune signature of responsiveness to PD-1 blockade in 56 patients treated with nivolumab in the CheckMate 205 phase II clinical trial. The TCR sequences for the study were processed through the immunoSEQ platform (Adaptive Biotechnologies, Seattle, WA, USA).
The scientists reported that anti-PD-1 therapy was most effective in patients with a diverse baseline TCR repertoire and an associated expansion of singleton clones during treatment. They also noted that CD4+ TCR diversity significantly increased during therapy, most prominently in patients who had achieved complete responses, but did not make the same observation for CD8+ TCR diversity. Further, the scientists said, patients who responded to therapy had an increased abundance of activated natural killer (NK) cells and a newly identified subset of CD3- CD68+ CD4+ GrB+ cells.
The investigators also found the trial patients had no significant differences in ratios of input CD4+ and CD8+ T cells or total detected CD4+ and CD8+ TCR sequences at baseline, but that peripheral CD4+ TCR repertoire diversity was significantly higher in healthy donors than in patients with newly diagnosed or relapsed or refractory cHL. While baseline CD4+ TCR diversity was not significantly different in newly diagnosed patients and those with relapsed or refractory disease who had complete responses to nivolumab, it was significantly lower in patients with partial responses or progressive disease. They saw similar patterns in baseline CD8+ TCR diversity.
The authors concluded that their studies highlighted the roles of recently expanded, clonally diverse CD4+ T cells and innate effectors in the efficacy of PD-1 blockade in cHL. The study was published on August 10, 2020 in the journal Nature Medicine.
Related Links:
Dana-Farber Cancer Institute
Adaptive Biotechnologies
Hodgkin lymphoma may be treated with chemotherapy, radiation therapy, and stem cell transplant. The choice of treatment often depends on how advanced the cancer has become and whether or not it has favorable features. In early disease, a cure is often possible. PD-1 blockade is highly effective in classical Hodgkin lymphomas (cHLs), which exhibit frequent copy-number gains of CD274 (PD-L1) and PDC1LG2 (PD-L2) on chromosome 9p24.1.
A team of medical oncologists collaborating with the Dana-Farber Cancer Institute (Boston, MA, USA) used T cell receptor (TCR) sequencing and cytometry by time-of-flight (CyTOF) analysis to obtain a peripheral immune signature of responsiveness to PD-1 blockade in 56 patients treated with nivolumab in the CheckMate 205 phase II clinical trial. The TCR sequences for the study were processed through the immunoSEQ platform (Adaptive Biotechnologies, Seattle, WA, USA).
The scientists reported that anti-PD-1 therapy was most effective in patients with a diverse baseline TCR repertoire and an associated expansion of singleton clones during treatment. They also noted that CD4+ TCR diversity significantly increased during therapy, most prominently in patients who had achieved complete responses, but did not make the same observation for CD8+ TCR diversity. Further, the scientists said, patients who responded to therapy had an increased abundance of activated natural killer (NK) cells and a newly identified subset of CD3- CD68+ CD4+ GrB+ cells.
The investigators also found the trial patients had no significant differences in ratios of input CD4+ and CD8+ T cells or total detected CD4+ and CD8+ TCR sequences at baseline, but that peripheral CD4+ TCR repertoire diversity was significantly higher in healthy donors than in patients with newly diagnosed or relapsed or refractory cHL. While baseline CD4+ TCR diversity was not significantly different in newly diagnosed patients and those with relapsed or refractory disease who had complete responses to nivolumab, it was significantly lower in patients with partial responses or progressive disease. They saw similar patterns in baseline CD8+ TCR diversity.
The authors concluded that their studies highlighted the roles of recently expanded, clonally diverse CD4+ T cells and innate effectors in the efficacy of PD-1 blockade in cHL. The study was published on August 10, 2020 in the journal Nature Medicine.
Related Links:
Dana-Farber Cancer Institute
Adaptive Biotechnologies
Latest Immunology News
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
- Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
- Cancer Mutation ‘Fingerprints’ to Improve Prediction of Immunotherapy Response
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
- Blood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
- Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
- Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
- Blood Test Could Identify Colon Cancer Patients to Benefit from NSAIDs
- Blood Test Could Detect Adverse Immunotherapy Effects
- Routine Blood Test Can Predict Who Benefits Most from CAR T-Cell Therapy
- New Test Distinguishes Vaccine-Induced False Positives from Active HIV Infection
- Gene Signature Test Predicts Response to Key Breast Cancer Treatment
- Chip Captures Cancer Cells from Blood to Help Select Right Breast Cancer Treatment
- Blood-Based Liquid Biopsy Model Analyzes Immunotherapy Effectiveness
- Signature Genes Predict T-Cell Expansion in Cancer Immunotherapy
Channels
Clinical Chemistry
view channel
Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
Colorectal cancer (CRC) is a leading cause of cancer-related death worldwide, with more than 60% of cases still diagnosed at a late stage. Uptake of existing screening tools remains suboptimal,... Read more
Automated NfL Assay Supports Monitoring of Neurological Disorders
Neuroaxonal injury occurs across a wide range of neurological disorders and remains difficult to monitor noninvasively over time. Blood-based measurement of neurofilament light chain (NfL) provides a biologically... Read moreMolecular Diagnostics
view channel
New Respiratory Panel Expands Pathogen Detection to 25 Targets
Respiratory infections often present with overlapping symptoms, complicating differential diagnosis in acute and community settings. The stakes are higher for older adults, young children, and people with... Read more
Simple Nasal Swab May Reveal Early Signs of Alzheimer’s Disease
Alzheimer’s disease affects millions worldwide but remains difficult to detect at its earliest, pre-symptomatic stage. Clinicians need tools that can identify biological changes before cognitive symptoms... Read moreImmunology
view channel
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read more
Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
Early identification of treatment response and relapse remains a major challenge in solid tumors, where minimal residual disease is difficult to detect with routine imaging and blood tests.... Read moreMicrobiology
view channel
Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Accurate and rapid identification of bacterial infections remains challenging in acute care, where delays can hinder timely, targeted therapy. Infectious diseases are a major cause of mortality worldwide,... Read more
Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
Direct-to-consumer gut microbiome kits promise personalized insights by profiling fecal bacteria and generating health readouts, but their analytical accuracy remains uncertain. A new study shows that... Read more
WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
Tuberculosis (TB) remains one of the world’s leading infectious disease killers, yet millions of cases go undiagnosed or are detected too late. Barriers such as reliance on sputum samples, limited laboratory... Read morePathology
view channel
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read more
AI-Powered Tool to Transform Dermatopathology Workflow
Skin cancer accounts for the largest number of cancer diagnoses in the United States, placing sustained pressure on pathology services. Diagnostic interpretation can be variable for challenging melanocytic... Read moreTechnology
view channel
Online Tool Supports Family Screening for Inherited Cancer Risk
Genetic test results in oncology often have implications for relatives who may share inherited cancer risk. Many health systems lack structured processes to help patients alert family members, limiting... Read more
Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
Pneumonia is commonly confirmed with chest X-rays or laboratory assays that can take hours, delaying clinical decisions in acute and outpatient settings. Breath-based diagnostics promise faster answers... Read moreIndustry
view channel
Integrated DNA Technologies Expands into Clinical Diagnostics
Integrated DNA Technologies (IDT; Coralville, Iowa, USA) has announced the launch of Archer FUSIONPlex-HT Dx and VARIANTPlex-HT Dx. This launch marks the company’s first in vitro diagnostic (IVD) offerings... Read more