Elevated Levels of Two Protein Biomarkers Indicate Potential for Development of Pre-Eclampsia
|
By LabMedica International staff writers Posted on 16 Jul 2020 |
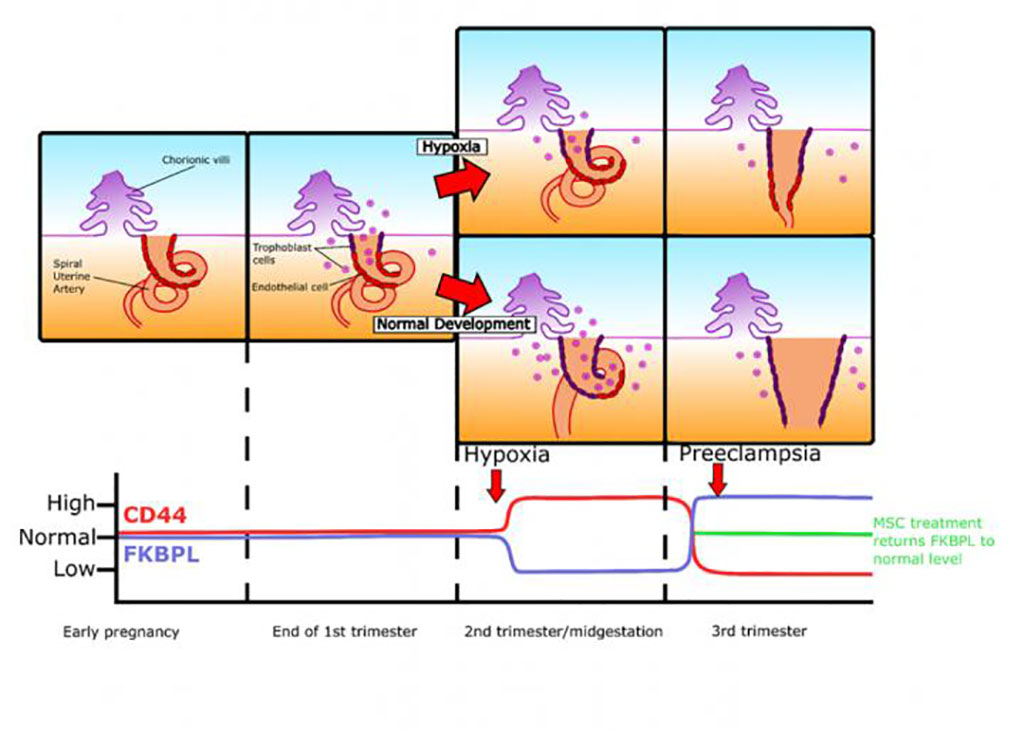
Image: Summary of changes FKBPL and CD44 changes throughout gestation in pregnancies complicated by pre-eclampsia (Photo courtesy of Oxford Press)
A pair of novel protein biomarkers has been identified in the blood of asymptomatic pregnant women that can be used to predict the potential onset of pre-eclampsia.
Pre-eclampsia (PE) is a disorder of pregnancy characterized by the onset of high blood pressure and often a significant amount of protein in the urine. When it arises, the condition begins after 20 weeks of pregnancy. In severe disease there may be red blood cell breakdown, a low blood platelet count, impaired liver function, kidney dysfunction, swelling, shortness of breath due to fluid in the lungs, or visual disturbances. Pre-eclampsia increases the risk of poor outcomes for both the mother and the baby. If left untreated, it may result in seizures at which point it is known as eclampsia. Ten to 15% of maternal mortality is associated with pre-eclampsia and eclampsia.
Development of effective prevention and treatment strategies for pre-eclampsia has been limited by the lack of accurate methods for identification of at-risk pregnancies.
In an effort to correct this lack, investigators at the University of Technology Sydney (Australia) evaluated the diagnostic and therapeutic potential for pre-eclampsia of the angiogenesis protein markers, FKBPL-CD44. FKBPL (FKBP prolyl isomerase like) has similarity to the immunophilin protein family, which play a role in immunoregulation and basic cellular processes involving protein folding and trafficking. CD44 is a cell-surface glycoprotein involved in cell-cell interactions, cell adhesion, and migration. It is a receptor for hyaluronic acid (HA) and can also interact with other ligands, such as osteopontin, collagens, and matrix metalloproteinases (MMPs). This protein participates in a wide variety of cellular functions including lymphocyte activation, recirculation and homing, hematopoiesis, and tumor metastasis.
For the current study, the investigators analyzed 57 samples that were pre-diagnosis (15 and 20 weeks’ gestation) and 18 that were post-diagnosis (14 for plasma; four for placenta) of pre-eclampsia for determination of FKBPL and CD44 concentrations, compared to healthy controls.
Results revealed that the CD44/FKBPL ratio was reduced in placenta and plasma following clinical diagnosis of pre-eclampsia. At 20 weeks’ gestation, a high plasma CD44/FKBPL ratio was independently associated with the 2.5-fold increased risk of pre-eclampsia.
"There are two main types of pre-eclampsia: early-onset pre-eclampsia diagnosed before 34 weeks of a pregnancy and late-onset pre-eclampsia diagnosed from 34 weeks onwards," said senior author Dr. Lana McClements, lecturer in biotechnology at the University of Technology Sydney. "The vast majority of the current screening and monitoring strategies are focused on early-onset pre-eclampsia, which comprises only 10-15% of all pre-eclampsia cases, whereas late pre-eclampsia has been largely neglected."
"The biomarkers allow the prediction of irregular placenta or maternal vascular function, which are key underlying causes of pre-eclampsia," said Dr. McClements. "This could lead to the early diagnosis and prevention of severe pre-eclampsia and associated complications including death, therefore also giving insight into disease mechanisms and possible treatment targets."
The study was published in the July 3, 2020, online edition of the Journal of Clinical Endocrinology and Metabolism.
Related Links:
University of Technology Sydney
Pre-eclampsia (PE) is a disorder of pregnancy characterized by the onset of high blood pressure and often a significant amount of protein in the urine. When it arises, the condition begins after 20 weeks of pregnancy. In severe disease there may be red blood cell breakdown, a low blood platelet count, impaired liver function, kidney dysfunction, swelling, shortness of breath due to fluid in the lungs, or visual disturbances. Pre-eclampsia increases the risk of poor outcomes for both the mother and the baby. If left untreated, it may result in seizures at which point it is known as eclampsia. Ten to 15% of maternal mortality is associated with pre-eclampsia and eclampsia.
Development of effective prevention and treatment strategies for pre-eclampsia has been limited by the lack of accurate methods for identification of at-risk pregnancies.
In an effort to correct this lack, investigators at the University of Technology Sydney (Australia) evaluated the diagnostic and therapeutic potential for pre-eclampsia of the angiogenesis protein markers, FKBPL-CD44. FKBPL (FKBP prolyl isomerase like) has similarity to the immunophilin protein family, which play a role in immunoregulation and basic cellular processes involving protein folding and trafficking. CD44 is a cell-surface glycoprotein involved in cell-cell interactions, cell adhesion, and migration. It is a receptor for hyaluronic acid (HA) and can also interact with other ligands, such as osteopontin, collagens, and matrix metalloproteinases (MMPs). This protein participates in a wide variety of cellular functions including lymphocyte activation, recirculation and homing, hematopoiesis, and tumor metastasis.
For the current study, the investigators analyzed 57 samples that were pre-diagnosis (15 and 20 weeks’ gestation) and 18 that were post-diagnosis (14 for plasma; four for placenta) of pre-eclampsia for determination of FKBPL and CD44 concentrations, compared to healthy controls.
Results revealed that the CD44/FKBPL ratio was reduced in placenta and plasma following clinical diagnosis of pre-eclampsia. At 20 weeks’ gestation, a high plasma CD44/FKBPL ratio was independently associated with the 2.5-fold increased risk of pre-eclampsia.
"There are two main types of pre-eclampsia: early-onset pre-eclampsia diagnosed before 34 weeks of a pregnancy and late-onset pre-eclampsia diagnosed from 34 weeks onwards," said senior author Dr. Lana McClements, lecturer in biotechnology at the University of Technology Sydney. "The vast majority of the current screening and monitoring strategies are focused on early-onset pre-eclampsia, which comprises only 10-15% of all pre-eclampsia cases, whereas late pre-eclampsia has been largely neglected."
"The biomarkers allow the prediction of irregular placenta or maternal vascular function, which are key underlying causes of pre-eclampsia," said Dr. McClements. "This could lead to the early diagnosis and prevention of severe pre-eclampsia and associated complications including death, therefore also giving insight into disease mechanisms and possible treatment targets."
The study was published in the July 3, 2020, online edition of the Journal of Clinical Endocrinology and Metabolism.
Related Links:
University of Technology Sydney
Latest Molecular Diagnostics News
- Position Statements Outline Evidence Standards for Multi-Cancer Detection Tests
- Ultrasensitive MRD Blood Test Detects Early Breast Cancer Recurrence
- Gene Fusion Patterns May Flag High Risk Solitary Fibrous Tumors
- New RNA Origami Method Supports Faster Targeted Testing for Repeat Expansion Disorders
- FDA Approves Expanded Liquid Biopsy Panel for Advanced Cancer Profiling
- Microbial Saliva Test Could Help Triage Esophageal Cancer Risk
- Expanded DPYD Genotyping Test Supports Safer Chemotherapy Dosing
- Blood Test Detects Early Nonresponse in Metastatic Prostate Cancer
- Multi-Omics Profiling Helps Predict BCG Response and Recurrence in Bladder Cancer
- New Computational Tool Reveals Genetic Driver of Idiopathic Neuropathy
- Breast Cancer-Specific Signatures Link Genome Instability to Outcomes
- FDA-Cleared Genomic Profiling Assay Guides Treatment Selection in Solid Tumors
- ctDNA Blood Test Could Help Guide Radiotherapy in Patients with Limited Metastases
- FDA-Approved MRD Blood Test Guides Adjuvant Bladder Cancer Therapy
- Whole-Genome MRD Test Detects Early Recurrence in Muscle-Invasive Bladder Cancer
- Tumor Genomic Profiling Identifies High-Risk Gallbladder Cancer
Channels
Clinical Chemistry
view channel
Liquid Biopsy Biomarkers May Improve Childhood Epilepsy Diagnosis
Childhood epilepsy remains a major neurological disorder with unmet needs for accurate, non-invasive biomarkers, as conventional tests such as electroencephalography and neuroimaging can have limited sensitivity... Read more
Blood-Based Sensor Detects Early Signs of Alzheimer’s and Parkinson’s
Alzheimer’s disease and Parkinson’s disease are increasing as populations age, yet diagnosis remains largely symptom-driven and often occurs after irreversible brain damage has begun. Earlier detection,... Read moreHematology
view channel
Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
Iron deficiency in school-age children can affect brain development, learning, growth, and physical performance, yet early deficiency may be missed when screening focuses mainly on anemia.... Read more
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read moreImmunology
view channel
Immune Enzyme Linked to Treatment-Resistant Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) affects nearly 3 million people in the United States and its prevalence continues to rise. Medications that target tumor necrosis factor (TNF)-alpha are widely used, but... Read more
Simple Blood Test Could Replace Biopsies for Lung Transplant Rejection Monitoring
Lung transplant recipients face some of the highest rates of acute cellular rejection, and routine surveillance often relies on repeated surgical biopsies. These procedures can cause complications such... Read moreMicrobiology
view channel
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read more
New AMR Assay Supports Rapid Infection Control Screening in Hospitals
As antimicrobial resistance spreads worldwide, healthcare-associated infections are placing a growing burden on hospitals, increasing the need for faster and broader diagnostic solutions.... Read morePathology
view channel
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read more
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read moreTechnology
view channel
AI-Enabled Assistant Unifies Molecular Workflow Planning and Support
Clinical laboratories and research groups face increasingly complex molecular workflows and expanding technical documentation spread across multiple systems. Fragmented digital tools can slow experiment... Read more
AI Tool Automates Validation of Laboratory Software Configuration Changes
Regulated laboratories face heavy documentation and requalification demands when software configurations change, slowing improvements and discouraging beneficial updates. A new capability now automates... Read moreIndustry
view channel
New Distribution Agreement Expands Access to CE-Marked Precision Oncology Assays
Eurobio Scientific (Les Ulis, France) has signed a distribution agreement with Canhelp Genomics (Hangzhou, China) to broaden availability of the Canhelp‑UCa and Canhelp‑Origin assays. The agreement extends... Read more