Randox Promotes Evidence MultiSTAT Automated Benchtop Immunoassay Analyzer
|
By Theresa Herman, Regional Director Posted on 31 Jan 2019 |

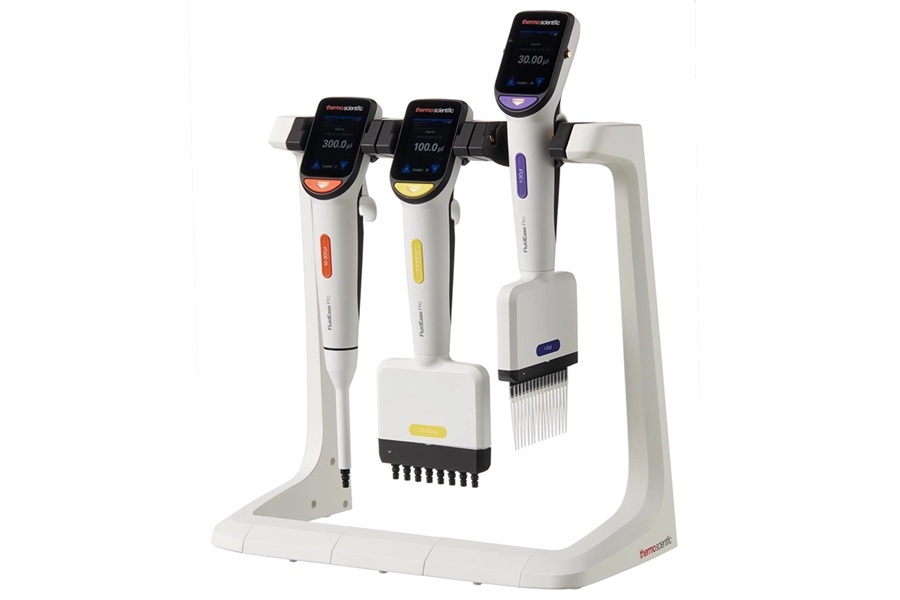
Image: The Evidence MultiSTAT analyzer (Photo courtesy of Randox Laboratories).
Randox Laboratories (Crumlin, UK) participated in MedLab Middle East 2019 held in Dubai, UAE, from February 4-7, 2019, where it offered live product demonstrations of the Evidence MultiSTAT automated benchtop immunoassay analyzer.
The Evidence MultiSTAT simultaneously detects up to 21 classical, prescription and synthetic drugs from a single patient sample. Utilizing revolutionary Biochip Array Technology, it uses a chemiluminescence reaction as a measuring principle to ensure consistently accurate results. Moving away from traditional single analyte assays, Biochip Array Technology boasts of cutting-edge multiplex testing capabilities providing rapid and accurate drug detection from a single sample. Biochip Array Technology has a proven high standard of accurate test results with CVs typically <10% with a 98% agreement with confirmatory methods.
Based on ELISA principles, the biochip is a solid-state device with discrete test sites onto which antibodies specific to different drug compounds are immobilized and stabilized. Competitive chemiluminescent immunoassays are then employed, offering a highly sensitive screen. Testing panels are available for blood and urine samples to accommodate various testing requirements, with a minimal 200ul sample volume required, ensuring more sample is available for confirmatory testing.
Randox has the world’s largest toxicology test menu, with the ability to detect over 500 drugs and drug metabolites. Randox also has the world’s most advanced drug screening test menu spanning a wide range of classical, prescription and synthetic drugs of abuse. The Evidence Multistat provides a complete drug profile in one simple process, reducing the amount of time and labor spent on individual tests. The reliability and accuracy of results also reduces the financial burden of re-testing. With multiple results obtained in less than 20 minutes, the Evidence MultiSTAT is an ideal analyzer for emergency drugs of abuse screening.
The Evidence MultiSTAT simultaneously detects up to 21 classical, prescription and synthetic drugs from a single patient sample. Utilizing revolutionary Biochip Array Technology, it uses a chemiluminescence reaction as a measuring principle to ensure consistently accurate results. Moving away from traditional single analyte assays, Biochip Array Technology boasts of cutting-edge multiplex testing capabilities providing rapid and accurate drug detection from a single sample. Biochip Array Technology has a proven high standard of accurate test results with CVs typically <10% with a 98% agreement with confirmatory methods.
Based on ELISA principles, the biochip is a solid-state device with discrete test sites onto which antibodies specific to different drug compounds are immobilized and stabilized. Competitive chemiluminescent immunoassays are then employed, offering a highly sensitive screen. Testing panels are available for blood and urine samples to accommodate various testing requirements, with a minimal 200ul sample volume required, ensuring more sample is available for confirmatory testing.
Randox has the world’s largest toxicology test menu, with the ability to detect over 500 drugs and drug metabolites. Randox also has the world’s most advanced drug screening test menu spanning a wide range of classical, prescription and synthetic drugs of abuse. The Evidence Multistat provides a complete drug profile in one simple process, reducing the amount of time and labor spent on individual tests. The reliability and accuracy of results also reduces the financial burden of re-testing. With multiple results obtained in less than 20 minutes, the Evidence MultiSTAT is an ideal analyzer for emergency drugs of abuse screening.
Latest MEDLAB 2019 News
- ERBA Group Launches New Hematology Analyzers at MEDLAB 2019
- Telstar Promotes Latest Generation of Freeze Dryers in Dubai
- Siemens Healthineers Showcases In-Vitro Diagnostics Products at MedLab 2019
- Mindray Displays New Generation Benchtop Cellular Analysis Line
- HORIBA Medical Shows Latest Yumizen G Range at Lab Trade Show
- Greiner Bio-One Displays Range of Safety Products at MedLab
- EKF Diagnostics Presents Latest Diabetes Care Analyzers
- BioSystems Highlights New iFOB Integral System at MedLab 2019
- Beckman Coulter Exhibits New Hematology Analyzer in Middle East
- Agappe Diagnostics Displays Analyzers and Solutions at Middle East Congress
- Abbott Highlights Point of Care Management Solutions
Channels
Clinical Chemistry
view channel
New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Steroid hormone measurement is a core application of clinical mass spectrometry, which is widely regarded as a diagnostic gold standard. Access to these high-specificity methods has often been constrained... Read more
Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
Alzheimer’s disease develops slowly, and biological changes can appear in blood many years before symptoms. While plasma assays for phosphorylated tau offer earlier detection, discerning whether these... Read moreMolecular Diagnostics
view channel
First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
Accurate identification of clonal immunoglobulin heavy chain (IGH) gene rearrangements is central to evaluating suspected B-cell lymphoproliferative disorders, where a single B-cell clone yields a defining... Read more
Plasma ctDNA Testing Predicts Breast Cancer Recurrence After Neoadjuvant Therapy
Accurate identification of breast cancer patients at risk of relapse after pre-surgery treatment is central to guiding adjuvant decisions, particularly in aggressive disease. Circulating fragments of tumor... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Tuberculosis (TB) remains a leading infectious killer, with more than 3,300 deaths and 29,000 new illnesses every day. Diagnostic delays and dependence on centralized laboratory networks continue to impede... Read more
Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Accurate and rapid identification of bacterial infections remains challenging in acute care, where delays can hinder timely, targeted therapy. Infectious diseases are a major cause of mortality worldwide,... Read more
Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
Direct-to-consumer gut microbiome kits promise personalized insights by profiling fecal bacteria and generating health readouts, but their analytical accuracy remains uncertain. A new study shows that... Read more
WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
Tuberculosis (TB) remains one of the world’s leading infectious disease killers, yet millions of cases go undiagnosed or are detected too late. Barriers such as reliance on sputum samples, limited laboratory... Read morePathology
view channel
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read more
AI-Powered Tool to Transform Dermatopathology Workflow
Skin cancer accounts for the largest number of cancer diagnoses in the United States, placing sustained pressure on pathology services. Diagnostic interpretation can be variable for challenging melanocytic... Read moreTechnology
view channel
Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
Chemotherapy selection for breast cancer is challenged by heterogeneous tumor responses. Conventional chemosensitivity assays can be slow, require large sample volumes, and struggle with complex biological... Read more
Online Tool Supports Family Screening for Inherited Cancer Risk
Genetic test results in oncology often have implications for relatives who may share inherited cancer risk. Many health systems lack structured processes to help patients alert family members, limiting... Read moreIndustry
view channel
Integrated DNA Technologies Expands into Clinical Diagnostics
Integrated DNA Technologies (IDT; Coralville, Iowa, USA) has announced the launch of Archer FUSIONPlex-HT Dx and VARIANTPlex-HT Dx. This launch marks the company’s first in vitro diagnostic (IVD) offerings... Read more