Exhaled Nitrous Oxide Adjunct Test Diagnoses Asthma
|
By LabMedica International staff writers Posted on 04 Jan 2018 |
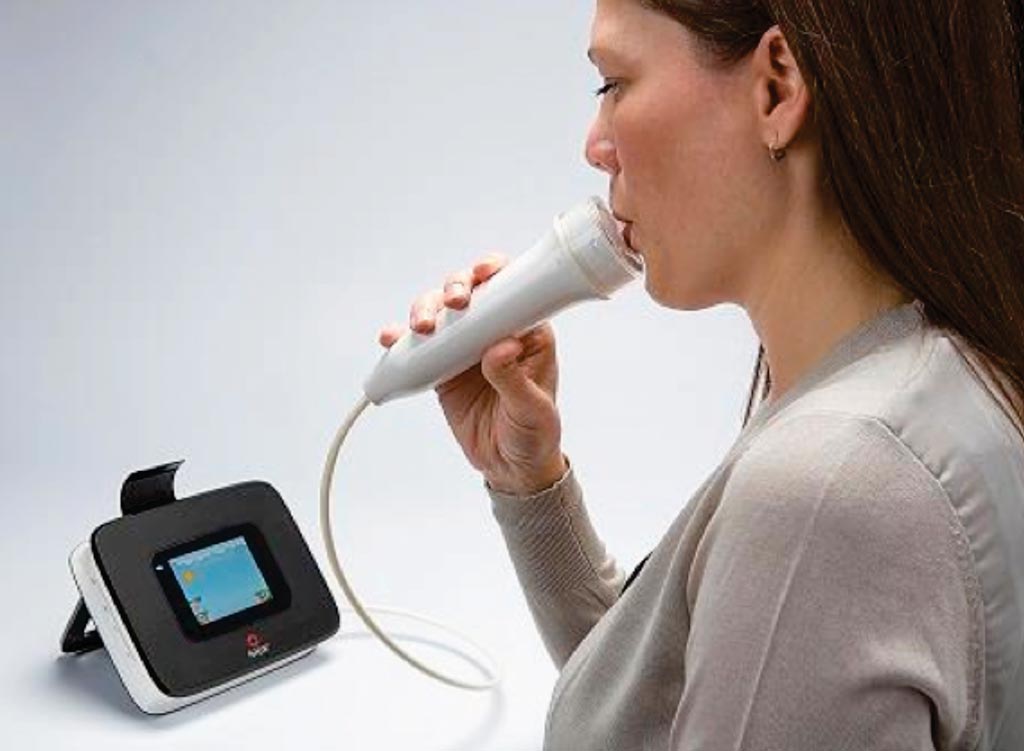
Image: The fractional exhaled nitric oxide (FeNO) concentration test is a quick and simple test and a valuable tool to assist in the diagnosis of asthma (Photo courtesy of Talkhealth Partnership).
Although about 24 million Americans are diagnosed with asthma every year, there is no single test that can diagnose the disease and common symptoms, such as shortness of breath, wheezing, and cough, are relatively nonspecific.
Various tests, including bronchodilator response and positive results on bronchial challenge, may be used by clinicians to aid in the diagnosis of asthma in the appropriate clinical context, but no single criterion standard diagnostic test exists. More recently, fractional exhaled nitric oxide (FeNO) concentration has been added to the list of tests that clinicians may use to diagnose asthma.
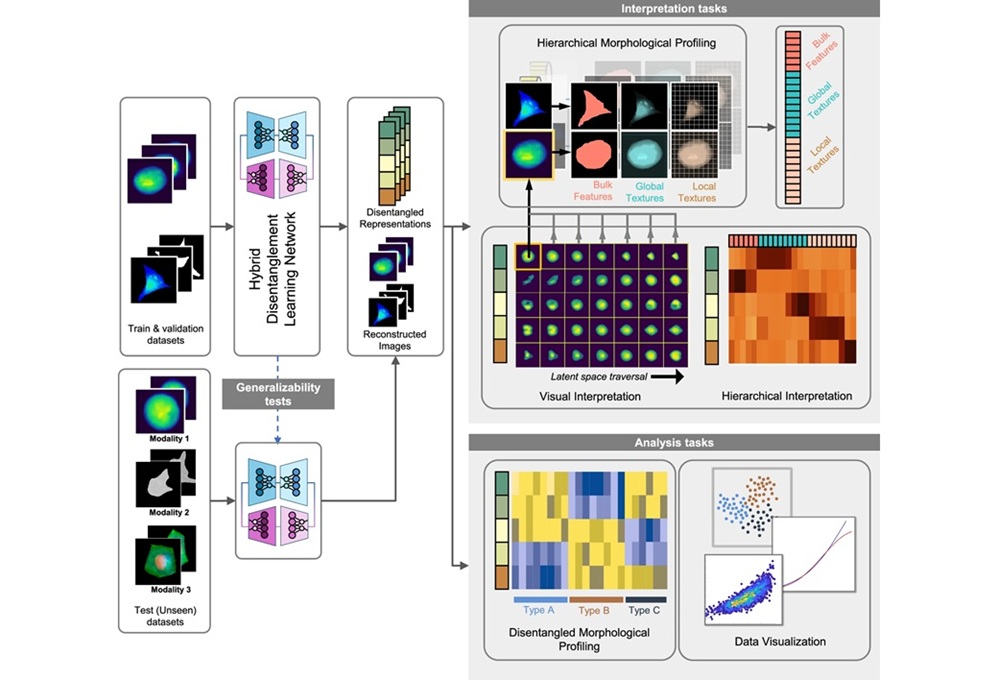
A team of scientists from the Mayo Clinic (Rochester, MN, USA) conducted a comprehensive literature search of six databases. The search included randomized clinical trials and observational studies that (1) enrolled patients aged five years and older with suspected asthma, (2) compared FeNO testing (diagnostic test) to standard diagnostic testing of asthma by health care professionals based on history, clinical course, or other diagnostic tests (clinical diagnosis, bronchodilator response, and positive results on bronchial challenge) (reference test), and (3) reported FeNO diagnostic accuracy.
The team included 43 studies with a total of 13,747 patients. In adults, using FeNO cutoffs of less than 20, 20 to 29, 30 to 39, and 40 or more parts per billion (ppb), FeNO testing had sensitivities of 0.80, 0.69, 0.53, and 0.41, respectively, and specificities of 0.64, 0.78, 0.85, and 0.93, respectively. In children, using FeNO cutoffs of less than 20 and 20 to 29 ppb, FeNO testing had sensitivities of 0.78 and 0.61, respectively, and specificities of 0.79 and 0.89, respectively. Depending on the FeNO cutoff, the posttest odds of having asthma with a positive FeNO test result increased by 2.80-fold to 7.00-fold. Diagnostic accuracy was modestly better in corticosteroid-naive asthmatics, children, and nonsmokers than in the overall population.
M. Hassan Murad, MD, MPH, the lead investigator of the study, said, “Asthma can sometimes be difficult to diagnose, and FeNO can be helpful to make therapeutic decisions more evidence based. In addition to a patient's history, the initial test is usually spirometry with an assessment of bronchodilator response. If this test does not confirm the diagnosis, but the index of suspicion for asthma is still high, measurement of FeNO may be helpful to rule in disease; although will still miss some patients with asthma.” The study was published on December 20, 2017, in the journal Mayo Clinic Proceedings.
Related Links:
Mayo Clinic
Various tests, including bronchodilator response and positive results on bronchial challenge, may be used by clinicians to aid in the diagnosis of asthma in the appropriate clinical context, but no single criterion standard diagnostic test exists. More recently, fractional exhaled nitric oxide (FeNO) concentration has been added to the list of tests that clinicians may use to diagnose asthma.
A team of scientists from the Mayo Clinic (Rochester, MN, USA) conducted a comprehensive literature search of six databases. The search included randomized clinical trials and observational studies that (1) enrolled patients aged five years and older with suspected asthma, (2) compared FeNO testing (diagnostic test) to standard diagnostic testing of asthma by health care professionals based on history, clinical course, or other diagnostic tests (clinical diagnosis, bronchodilator response, and positive results on bronchial challenge) (reference test), and (3) reported FeNO diagnostic accuracy.
The team included 43 studies with a total of 13,747 patients. In adults, using FeNO cutoffs of less than 20, 20 to 29, 30 to 39, and 40 or more parts per billion (ppb), FeNO testing had sensitivities of 0.80, 0.69, 0.53, and 0.41, respectively, and specificities of 0.64, 0.78, 0.85, and 0.93, respectively. In children, using FeNO cutoffs of less than 20 and 20 to 29 ppb, FeNO testing had sensitivities of 0.78 and 0.61, respectively, and specificities of 0.79 and 0.89, respectively. Depending on the FeNO cutoff, the posttest odds of having asthma with a positive FeNO test result increased by 2.80-fold to 7.00-fold. Diagnostic accuracy was modestly better in corticosteroid-naive asthmatics, children, and nonsmokers than in the overall population.
M. Hassan Murad, MD, MPH, the lead investigator of the study, said, “Asthma can sometimes be difficult to diagnose, and FeNO can be helpful to make therapeutic decisions more evidence based. In addition to a patient's history, the initial test is usually spirometry with an assessment of bronchodilator response. If this test does not confirm the diagnosis, but the index of suspicion for asthma is still high, measurement of FeNO may be helpful to rule in disease; although will still miss some patients with asthma.” The study was published on December 20, 2017, in the journal Mayo Clinic Proceedings.
Related Links:
Mayo Clinic
Latest Clinical Chem. News
- Blood Test Detects Testicular Cancer Missed by Standard Markers
- Routine Blood Tests Identify Biomarkers Linked to PTSD
- Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
- Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
- Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
- Rapid Biosensor Detects Pancreatic Cancer Biomarker for Early Detection
- Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
- Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
- Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
- AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
- Next Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
- Blood Metabolite Test Detects Early Cognitive Decline
- AI-Based Blood Test Diagnose Multiple Brain Disorders from Blood Sample
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
- New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
Channels
Molecular Diagnostics
view channel
CRISPR-Based Test Identifies Multiple Respiratory Viruses Simultaneously
Respiratory virus co-circulation complicates differential diagnosis, as overlapping symptoms can obscure etiology. Multiplex testing typically depends on multiple enzymes or fluorophores and multistep... Read more
Multi-Omic Assay Predicts Recurrence and Radiation Benefit in Early Breast Cancer
Early-stage invasive breast cancer is frequently managed with breast-conserving surgery followed by adjuvant radiation therapy, but the magnitude of benefit from radiation varies among patients.... Read more
Portable Test Detects Tuberculosis from Tongue Swabs in 30 Minutes
Despite decades of effective drug regimens, tuberculosis remains the leading cause of death from an infectious disease, driven in part by limited access to accurate, rapid testing. Conventional diagnostics... Read more
Blood Test Receives FDA Breakthrough Status to Differentiate Schizophrenia and Bipolar Disorder
Differentiating schizophrenia from bipolar I disorder in symptomatic patients is challenging because early presentations often overlap. Current diagnostic pathways rely heavily on subjective assessments... Read moreHematology
view channel
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read more
Blood Test Enables Early Detection of Multiple Myeloma Relapse
Bone marrow biopsies remain central to diagnosing and monitoring multiple myeloma, yet the procedure is painful, invasive, and often repeated over time. Older patients—who represent most new cases—can... Read moreImmunology
view channel
Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
Sepsis remains a time-critical condition in which rapid risk assessment is often hindered by reliance on centralized laboratory testing. The global burden is substantial, with an estimated 166 million... Read more
Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
Accurate laboratory diagnosis of early Lyme disease remains challenging because serologic responses may be limited soon after infection. Missed detection at this stage can delay evaluation and management... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
Diabetic kidney disease is a leading cause of chronic kidney disease and end-stage kidney disease, affecting 20%–40% of people with diabetes and more than 107 million individuals worldwide as of 2021.... Read more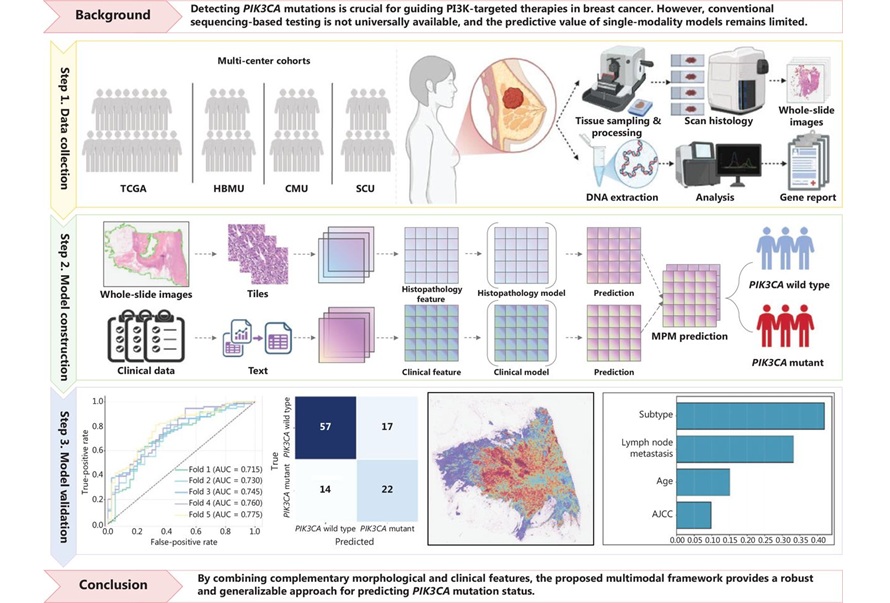
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read moreTechnology
view channel
Microfluidic Single-Cell Assay Predicts Breast Cancer Risk
Risk stratification for breast cancer remains imprecise, as population-based models and breast density can over- or underestimate individual risk, potentially leading to over- or under-screening.... Read more