High Plasma Glucose Raises Baby Heart-Defect Risk
|
By LabMedica International staff writers Posted on 27 Dec 2017 |

Image: Testing for blood glucose levels during early pregnancy to assess the risk of congenital heart disease (Photo courtesy of Paras Hospitals).
For many years, physicians have known that women with diabetes face an increased risk of giving birth to babies with heart defects and some studies have also suggested a link between nondiabetic mothers' blood sugar levels and babies' heart defect risk.
One challenge associated with conducting such studies was the fact that maternal blood glucose is not routinely measured in nondiabetic pregnant women. Instead, women typically receive an oral glucose tolerance test halfway through pregnancy to determine whether they have gestational diabetes, but this test is performed well after the fetal heart has formed.
A team of scientists mainly from Stanford University Medical Center (Stanford, CA, USA) studied medical records from 19,107 pairs of mothers and their babies born between 2009 and 2015. The records included details of the mothers' prenatal care, including blood test results and any cardiac diagnoses made for the babies during pregnancy or after birth. The scientists analyzed blood glucose levels from any blood sample collected from the mothers between four weeks prior to the estimated date of conception and the end of the 14th gestational week, just after the completion of the first trimester of pregnancy. These early blood glucose measurements were available for 2,292, or 13%, of women in the study. They also looked at the results of oral glucose tolerance tests (OGTT) performed around 20 weeks of gestation, which were available for 9,511, or just under half, of the women in the study.
After excluding women who had diabetes before pregnancy or who developed it during pregnancy, the results showed that the risk of giving birth to a child with a congenital heart defect was elevated by 8% for every increase of 10 mg/dL in blood glucose levels in the early stages of pregnancy. The mean early glucose value was 96 mg/dL (5.3 mmol/L) in pregnancies without congenital heart disease (CHD) versus 107 mg/dL (6.0 mmol/L) in pregnancies with CHD. The respective mean 1-hour OGTT values were 117 mg/dL (6.5 mmol/L) and 122 mg/dL (6.8 mmol/L).
The findings demonstrate that higher random plasma glucose values measured during early pregnancy correlate with increased risk for congenital heart disease in offspring of mothers who do not have diabetes. Furthermore, plasma glucose measured during early pregnancy was more associated with risk for congenital heart disease in offspring, compared with the OGTT which is often used to risk-stratify pregnancies for fetal-echocardiographic screening.
James R. Priest, MD, an assistant professor of pediatric cardiology and the senior author of the study, said, “Most women who have a child with congenital heart disease are not diabetic. We found that in women who don't already have diabetes or develop diabetes during pregnancy, we can still measure risk for having a child with congenital heart disease by looking at their glucose values during the first trimester of pregnancy.” The study was published on December 15, 2017, in The Journal of Pediatrics.
Related Links:
Stanford University Medical Center
One challenge associated with conducting such studies was the fact that maternal blood glucose is not routinely measured in nondiabetic pregnant women. Instead, women typically receive an oral glucose tolerance test halfway through pregnancy to determine whether they have gestational diabetes, but this test is performed well after the fetal heart has formed.
A team of scientists mainly from Stanford University Medical Center (Stanford, CA, USA) studied medical records from 19,107 pairs of mothers and their babies born between 2009 and 2015. The records included details of the mothers' prenatal care, including blood test results and any cardiac diagnoses made for the babies during pregnancy or after birth. The scientists analyzed blood glucose levels from any blood sample collected from the mothers between four weeks prior to the estimated date of conception and the end of the 14th gestational week, just after the completion of the first trimester of pregnancy. These early blood glucose measurements were available for 2,292, or 13%, of women in the study. They also looked at the results of oral glucose tolerance tests (OGTT) performed around 20 weeks of gestation, which were available for 9,511, or just under half, of the women in the study.
After excluding women who had diabetes before pregnancy or who developed it during pregnancy, the results showed that the risk of giving birth to a child with a congenital heart defect was elevated by 8% for every increase of 10 mg/dL in blood glucose levels in the early stages of pregnancy. The mean early glucose value was 96 mg/dL (5.3 mmol/L) in pregnancies without congenital heart disease (CHD) versus 107 mg/dL (6.0 mmol/L) in pregnancies with CHD. The respective mean 1-hour OGTT values were 117 mg/dL (6.5 mmol/L) and 122 mg/dL (6.8 mmol/L).
The findings demonstrate that higher random plasma glucose values measured during early pregnancy correlate with increased risk for congenital heart disease in offspring of mothers who do not have diabetes. Furthermore, plasma glucose measured during early pregnancy was more associated with risk for congenital heart disease in offspring, compared with the OGTT which is often used to risk-stratify pregnancies for fetal-echocardiographic screening.
James R. Priest, MD, an assistant professor of pediatric cardiology and the senior author of the study, said, “Most women who have a child with congenital heart disease are not diabetic. We found that in women who don't already have diabetes or develop diabetes during pregnancy, we can still measure risk for having a child with congenital heart disease by looking at their glucose values during the first trimester of pregnancy.” The study was published on December 15, 2017, in The Journal of Pediatrics.
Related Links:
Stanford University Medical Center
Latest Clinical Chem. News
- Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
- Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
- AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
- Next Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
- Blood Metabolite Test Detects Early Cognitive Decline
- AI-Based Blood Test Diagnose Multiple Brain Disorders from Blood Sample
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
- New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
- CSF Biomarker Improves Diagnosis of Parkinson’s Disease and Lewy Body Dementia
- Simple Urine Home Test Kit Could Detect Early-Stage Breast Cancer
- Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
- New Tool Tracks Biomarker Changes to Predict Myeloma Progression
- New Plasma Tau Assay Improves Prediction of Alzheimer’s Progression
- First IVD Immunoassay to Detect Alzheimer’s Risk Gene Variant Receives CE Mark
- Routine Blood Markers Predict Heart Failure Risk in Prediabetes
Channels
Molecular Diagnostics
view channel
Blood-Based Epigenetic Signals Enable Osteosarcoma Disease Monitoring
Osteosarcoma is a rare but aggressive pediatric bone cancer where recurrence and metastasis remain difficult to detect early. Imaging-based surveillance can miss small lesions and exposes children to repeated... Read more
Host–Virus Genetic Interactions Drive Nasopharyngeal Cancer Risk
Epstein–Barr virus (EBV) infects more than 95% of adults worldwide, yet only a small fraction develops EBV‑associated cancers such as nasopharyngeal carcinoma. Explaining this divergence requires understanding... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read moreMicrobiology
view channel
Rapid Color Test Stratifies Virulent and Resistant Staph Strains
Staphylococcus aureus (golden staph) remains a leading cause of infection-related mortality worldwide, responsible for more than a million deaths each year. Rapidly distinguishing highly virulent or a... Read more
Syndromic Panel Enables Rapid Identification of Bloodstream Infections
Bloodstream infections require rapid identification of causative pathogens and resistance determinants to guide therapy, yet laboratories often face pressure to deliver clinically relevant results quickly... Read more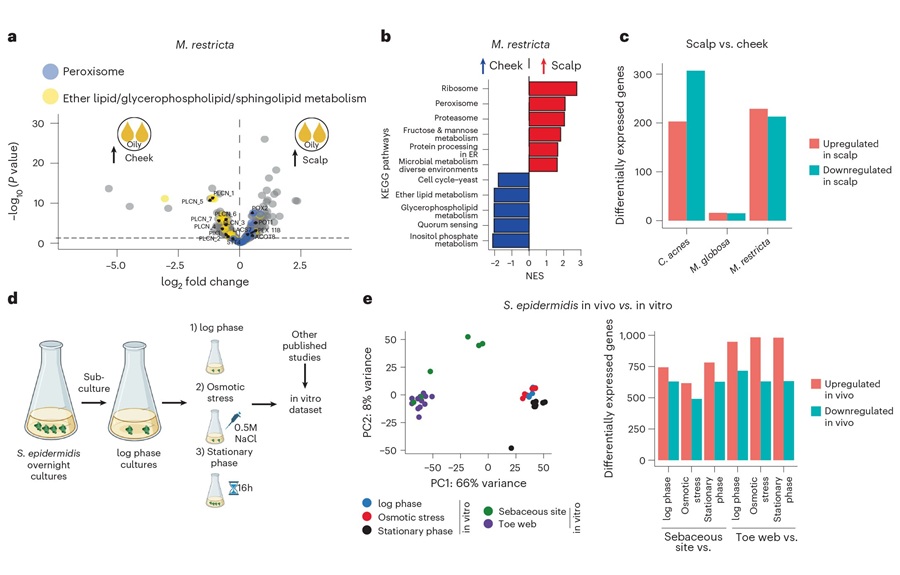
RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
Human skin carries diverse microbial communities that influence barrier function and inflammation, yet identifying which organisms are metabolically active has been challenging. DNA-based surveys catalog... Read more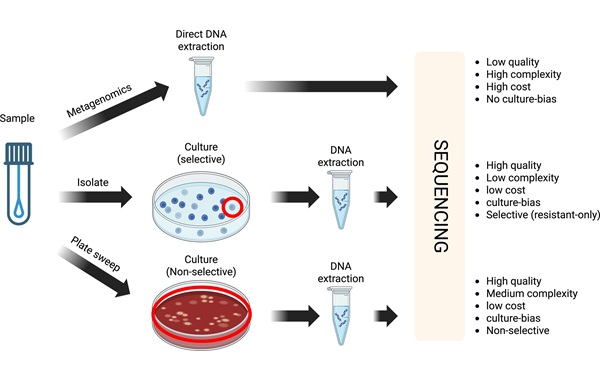
Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
Intensive care units face persistent threats from hospital-acquired infections, increasingly driven by drug-resistant bacteria. Rapidly pinpointing environmental reservoirs and transmission hotspots remains... Read morePathology
view channel
Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
Colorectal cancer is common and often lethal, and therapeutic decision-making is complicated by heterogeneous tumor microenvironments. Immunotherapy benefits only a small subset of patients, around 5%,... Read more
Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
Antibody-drug conjugates (ADC) are reshaping oncology, yet scalable biomarkers that reliably predict which patients will benefit remain limited as treatment regimens and combinations grow more complex.... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
Partnership Expands Ultrasensitive WGS Assay for for Hematologic Malignancies and MRD Monitoring
Tempus AI and Predicta Biosciences announced the commercial expansion of a co-branded whole‑genome sequencing assay GenoPredicta, which is intended for comprehensive genomic characterization of hematologic... Read more