Personal Lab Offers Rapid Detection of Food Allergens
|
By LabMedica International staff writers Posted on 06 Nov 2017 |

Image: The iEAT system for onsite antigen detection consists of a pocket-sized detector, an electrode chip, and a disposable kit for allergen extraction. The detector connects with a smartphone for system control and data upload to a cloud server (Photo courtesy of Lin, et al. ACS Nano, August 2017).
Researchers have developed a small, low cost device for individual use that can accurately detect food allergens in less than ten minutes.
Adverse food reactions, including food allergies, food sensitivities, and autoimmune reaction (e.g., celiac disease) affect 5-15% of the population of the USA and remain a considerable public health problem requiring stringent food avoidance and epinephrine availability for emergency events. Avoiding problematic foods is difficult in practical terms, given current reliance on prepared foods and out-of-home meals.
In response to the food allergy problem, investigators at Harvard Medical School (Boston, MA, USA) developed a portable, point-of-use detection technology, that they called "integrated exogenous antigen testing" (iEAT).
The iEAT device consists of three components: (1) a small plastic test tube, (2) a small electronic detection module, and (3) the electronic keychain reader. To perform the test, the user dissolves a small sample of the food in the plastic test tube and then adds magnetic beads that capture the food allergen of interest. A bit of this mixture is loaded onto electrode strips attached to a small module that is then inserted into the electronic keychain reader. The keychain reader has a small display that indicates whether the allergen is present, and if so, in what concentration.
The prototype iEAT system was optimized to detect five major food antigens in peanuts, hazelnuts, wheat, milk, and eggs. Antigen extraction and detection with iEAT required less than 10 minutes and achieved high-detection sensitivities (e.g., 0.1 milligram per kilogram for gluten, 200 times lower than regulatory limits of 20 milligram per kilogram).
The investigators also developed a dedicated cell phone application, which allows the user to compile and store the data collected by testing different foods for various allergens at different restaurants or in packaged foods. The application is set up to share this information online with both time and location stamps indicating when, where, and in what food or dish an allergen reading was taken.
“High accuracy built into a compact system was the key goals of the project,” said contributing author Dr. Ralph Weissleder, professor of radiology and systems biology at Harvard Medical School. “Users can be confident that even if they are sensitive to very low levels, iEAT will be able to give them exact concentrations. Armed with accurate concentration levels they will not have to completely avoid potentially problematic foods, but will know whether an allergen is at a dangerous level for them or a concentration that is safe for them to eat.”
The iEAT device was described in the August 2017 issue of the journal ACS Nano.
Related Links:
Harvard Medical School
Adverse food reactions, including food allergies, food sensitivities, and autoimmune reaction (e.g., celiac disease) affect 5-15% of the population of the USA and remain a considerable public health problem requiring stringent food avoidance and epinephrine availability for emergency events. Avoiding problematic foods is difficult in practical terms, given current reliance on prepared foods and out-of-home meals.
In response to the food allergy problem, investigators at Harvard Medical School (Boston, MA, USA) developed a portable, point-of-use detection technology, that they called "integrated exogenous antigen testing" (iEAT).
The iEAT device consists of three components: (1) a small plastic test tube, (2) a small electronic detection module, and (3) the electronic keychain reader. To perform the test, the user dissolves a small sample of the food in the plastic test tube and then adds magnetic beads that capture the food allergen of interest. A bit of this mixture is loaded onto electrode strips attached to a small module that is then inserted into the electronic keychain reader. The keychain reader has a small display that indicates whether the allergen is present, and if so, in what concentration.
The prototype iEAT system was optimized to detect five major food antigens in peanuts, hazelnuts, wheat, milk, and eggs. Antigen extraction and detection with iEAT required less than 10 minutes and achieved high-detection sensitivities (e.g., 0.1 milligram per kilogram for gluten, 200 times lower than regulatory limits of 20 milligram per kilogram).
The investigators also developed a dedicated cell phone application, which allows the user to compile and store the data collected by testing different foods for various allergens at different restaurants or in packaged foods. The application is set up to share this information online with both time and location stamps indicating when, where, and in what food or dish an allergen reading was taken.
“High accuracy built into a compact system was the key goals of the project,” said contributing author Dr. Ralph Weissleder, professor of radiology and systems biology at Harvard Medical School. “Users can be confident that even if they are sensitive to very low levels, iEAT will be able to give them exact concentrations. Armed with accurate concentration levels they will not have to completely avoid potentially problematic foods, but will know whether an allergen is at a dangerous level for them or a concentration that is safe for them to eat.”
The iEAT device was described in the August 2017 issue of the journal ACS Nano.
Related Links:
Harvard Medical School
Latest Technology News
- Microfluidic Single-Cell Assay Predicts Breast Cancer Risk
- AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
- Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
- Noninvasive Sputum Test Detects Early Lung Cancer
- New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
- Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
- Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
- Online Tool Supports Family Screening for Inherited Cancer Risk
- Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
- New Electronic Pipette Enhances Workflows with Touchscreen Control
- AI Model Outperforms Clinicians in Rare Disease Detection
- AI-Driven Diagnostic Demonstrates High Accuracy in Detecting Periprosthetic Joint Infection
- Blood Test “Clocks” Predict Start of Alzheimer’s Symptoms
- AI-Powered Biomarker Predicts Liver Cancer Risk
Channels
Clinical Chemistry
view channel
Blood Test Detects Testicular Cancer Missed by Standard Markers
Testicular cancer most often affects adolescents and young adults and is highly treatable when found early. Diagnosis can be difficult when tumors do not produce sufficient levels of standard blood-based... Read more
Routine Blood Tests Identify Biomarkers Linked to PTSD
Post-traumatic stress disorder (PTSD) is associated with a range of chronic physical health conditions and affects multiple organ systems. Clinical laboratories routinely measure blood analytes that reflect... Read moreHematology
view channel
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read more
Blood Test Enables Early Detection of Multiple Myeloma Relapse
Bone marrow biopsies remain central to diagnosing and monitoring multiple myeloma, yet the procedure is painful, invasive, and often repeated over time. Older patients—who represent most new cases—can... Read moreImmunology
view channel
Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
Sepsis remains a time-critical condition in which rapid risk assessment is often hindered by reliance on centralized laboratory testing. The global burden is substantial, with an estimated 166 million... Read more
Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
Accurate laboratory diagnosis of early Lyme disease remains challenging because serologic responses may be limited soon after infection. Missed detection at this stage can delay evaluation and management... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read morePathology
view channel
New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
Diabetic kidney disease is a leading cause of chronic kidney disease and end-stage kidney disease, affecting 20%–40% of people with diabetes and more than 107 million individuals worldwide as of 2021.... Read more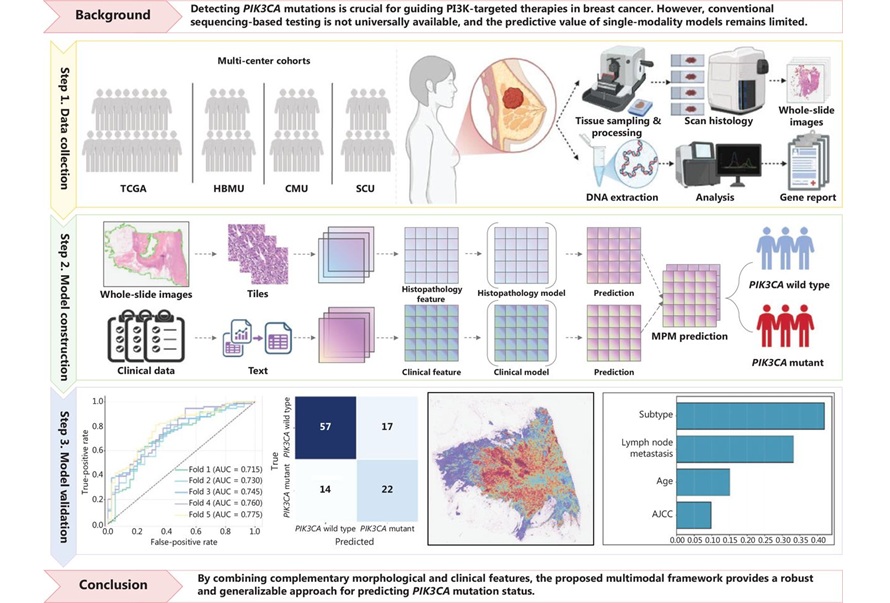
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read moreTechnology
view channel
Microfluidic Single-Cell Assay Predicts Breast Cancer Risk
Risk stratification for breast cancer remains imprecise, as population-based models and breast density can over- or underestimate individual risk, potentially leading to over- or under-screening.... Read more