Enzyme Signpost Points to Better Bowel Cancer Test
|
By LabMedica International staff writers Posted on 20 Mar 2016 |
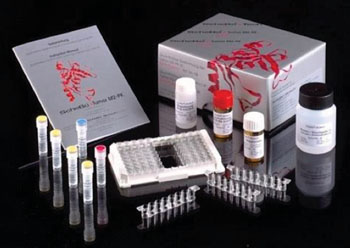
Image: The ScheBo Tumor M2-PK Stool sandwich enzyme-linked immunosorbent assay (Photo courtesy of ScheBo Biotech AG).
A sensitive screening tool for colorectal cancer that detects a special isoenzyme of pyruvate kinase, termed M2-PK, which leaks from the cancerous tissue into the bowel, can then be found in feces.
One alteration consistently found during tumor formation, including gastrointestinal tumors, is the upregulation of glycolytic enzymes. This upregulation takes place at the ribonucleic acid (RNA) and protein level, as well as at the level of enzymatic activities.
The UK Government is currently considering the introduction of a national bowel-screening program. One of the screening tests under consideration is based on the work done several years ago by scientists at the Giessen University Hospital, (Germany) and their colleagues. The team asked patients, given appointments for colonoscopy for various reasons, to provide one stool sample for measuring fecal Tumor M2-PK. Endoscopies were carried out as standard investigations. Histology was obtained from the routine biopsies and/or from surgery. In all, 60 patients with colorectal cancer have been evaluated.
Stool samples of patients with colorectal cancer and patients without pathological findings were tested. Tumor M2-PK was measured with a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) (ScheBo Biotech AG; Giessen, Germany). The ELISA plate is coated with a monoclonal antibody against Tumor M2-PK. Tumor M2-PK from stool samples or standards binds to the antibody. A second monoclonal antibody, which is biotinylated, binds to Tumor M2-PK during the next incubation. Both monoclonal antibodies against Tumor M2-PK specifically react with Tumor M2-PK (dimeric form of M2-PK) and do not cross-react with the other isoforms of pyruvate kinase (type L, R, M1 and tetrameric M2-PK).
There was a highly significant difference between tumor patients and controls. At a cutoff level of 4 UmL-1 the sensitivity was calculated to be 73% and the specificity as 78%. The intra-assay variance was evaluated by 18-fold determination of five samples (5–66 UmL-1), giving an average coefficient of variance (CV) of 7.9% (3.5%–13.6%). The interassay variance was calculated with five samples between 4 and 73 UmL-1, tested on 10 different days. The average CV was 7.3% (3.8%–12.6%).
Robert Souhami, CBE, FMedSci, Director of Policy and Communication for Cancer Research UK, said, “There is currently much interest in this area of investigation. We hope that enzymes such as this one will eventually offer not only useful screening tools, but also an effective method of monitoring bowel cancer patients in remission, so that any return of disease can be quickly detected and acted upon.”
Related Links:
Giessen University Hospital
ScheBo Biotech AG
One alteration consistently found during tumor formation, including gastrointestinal tumors, is the upregulation of glycolytic enzymes. This upregulation takes place at the ribonucleic acid (RNA) and protein level, as well as at the level of enzymatic activities.
The UK Government is currently considering the introduction of a national bowel-screening program. One of the screening tests under consideration is based on the work done several years ago by scientists at the Giessen University Hospital, (Germany) and their colleagues. The team asked patients, given appointments for colonoscopy for various reasons, to provide one stool sample for measuring fecal Tumor M2-PK. Endoscopies were carried out as standard investigations. Histology was obtained from the routine biopsies and/or from surgery. In all, 60 patients with colorectal cancer have been evaluated.
Stool samples of patients with colorectal cancer and patients without pathological findings were tested. Tumor M2-PK was measured with a commercially available sandwich enzyme-linked immunosorbent assay (ELISA) (ScheBo Biotech AG; Giessen, Germany). The ELISA plate is coated with a monoclonal antibody against Tumor M2-PK. Tumor M2-PK from stool samples or standards binds to the antibody. A second monoclonal antibody, which is biotinylated, binds to Tumor M2-PK during the next incubation. Both monoclonal antibodies against Tumor M2-PK specifically react with Tumor M2-PK (dimeric form of M2-PK) and do not cross-react with the other isoforms of pyruvate kinase (type L, R, M1 and tetrameric M2-PK).
There was a highly significant difference between tumor patients and controls. At a cutoff level of 4 UmL-1 the sensitivity was calculated to be 73% and the specificity as 78%. The intra-assay variance was evaluated by 18-fold determination of five samples (5–66 UmL-1), giving an average coefficient of variance (CV) of 7.9% (3.5%–13.6%). The interassay variance was calculated with five samples between 4 and 73 UmL-1, tested on 10 different days. The average CV was 7.3% (3.8%–12.6%).
Robert Souhami, CBE, FMedSci, Director of Policy and Communication for Cancer Research UK, said, “There is currently much interest in this area of investigation. We hope that enzymes such as this one will eventually offer not only useful screening tools, but also an effective method of monitoring bowel cancer patients in remission, so that any return of disease can be quickly detected and acted upon.”
Related Links:
Giessen University Hospital
ScheBo Biotech AG
Latest Immunology News
- Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
- Biomarkers and Molecular Testing Advance Precision Allergy Care
- Point-of-Care Tests Could Expand Access to Mpox Diagnosis
- T-Cell Senescence Profiling May Predict CAR T Responses
- Finger-Prick Lateral Flow Test Detects Sepsis Biomarkers at Point of Care
- Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
- Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
- Study Finds Influenza Often Undiagnosed in Winter Deaths
- Combined Screening Approach Identifies Early Leprosy Cases
- Antibody Blood Test Identifies Active TB and Distinguishes Latent Infection
- FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
- Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
- Microfluidic Chip Detects Cancer Recurrence from Immune Response Signals
- Cancer Mutation ‘Fingerprints’ to Improve Prediction of Immunotherapy Response
- Immune Signature Identified in Treatment-Resistant Myasthenia Gravis
- New Biomarker Predicts Chemotherapy Response in Triple-Negative Breast Cancer
Channels
Clinical Chemistry
view channel
Urine-Based Nanosensor Tracks Lung Cancer and Fibrosis Noninvasively
Lung cancer remains difficult to monitor for early progression and treatment resistance, while pulmonary fibrosis continues to pose major challenges for early diagnosis. Clinicians need repeatable, noninvasive... Read more
Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
Alzheimer’s disease is the most common cause of dementia, yet confirmatory testing remains invasive and hard to access. Diagnosis currently takes an average of 3.5 years, and about 75% of people with dementia... Read moreMolecular Diagnostics
view channel
ctDNA Blood Test Could Help Guide Radiotherapy in Patients with Limited Metastases
Selecting the right therapy for patients whose solid tumors have begun to spread remains a major clinical challenge. Clinicians often count metastatic lesions on X-ray, computed tomography (CT), or magnetic... Read more
FDA-Approved MRD Blood Test Guides Adjuvant Bladder Cancer Therapy
Muscle-invasive bladder cancer (MIBC) carries a substantial risk of recurrence after radical cystectomy, and selecting which patients require adjuvant therapy remains challenging. Approximately 30,000... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read moreTechnology
view channel
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read more
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read moreIndustry
view channel
AI-Powered Multi-Functional Analyzer Wins German Innovation Award
Hematology services are increasingly delivered across distributed care settings, where limited staffing and complex workflows can extend turnaround times. Advanced morphology review still often depends... Read more