Hepatitis C Assay Receives European In Vitro Diagnostic Test Certification
|
By LabMedica International staff writers Posted on 27 Apr 2015 |
Laboratories that adhere to the European Directive on In Vitro Diagnostic Medical Devices will now be able to work with a quantitative test that provides on-demand molecular testing for confirmation of infection and monitoring of Hepatitis C virus (HCV).
The Cepheid (Sunnyvale, CA, USA) Xpert HCV Viral Load assay was recently awarded CE IVD mark status under the European Directive. The test was designed to run on the Cepheid GeneXpert System, one of the world's leading molecular diagnostic platforms with over 8,000 systems deployed globally in both developed and emerging market countries.
Cepheid's GeneXpert System is a closed, self-contained, fully-integrated, and automated platform that combines on-board sample preparation with real-time PCR (polymerase chain reaction) amplification and detection functions for fully integrated and automated nucleic acid analysis. The system is designed to purify, concentrate, detect, and identify targeted nucleic acid sequences thereby delivering answers directly from unprocessed samples.
The Xpert HCV Viral Load assay is intended for use as an aid in the management of HCV infected patients undergoing antiviral therapy. The test can be used to measure HCV RNA levels at baseline and during treatment, and to help predict virological responses to HCV therapy. In addition, the test can be used for confirmation of HCV serologic test results.
"Existing HCV viral load tests run on complex systems that require highly-trained technicians to operate, and generally lack the flexibility to provide on-demand answers when required," said John Bishop, chairman and CEO of Cepheid. "Xpert HCV Viral Load, our 21st CE-IVD Xpert test, provides reliable results in less than two hours, making it a valuable tool for clinicians in the management of HCV infected patients on antiviral therapy."
"The Xpert HCV Viral Load test incorporates a number of novel technologies including our "BigTube" design, which allows us to take full advantage of the efficiency of integrated GeneXpert cartridge fluidics," said Dr. David H. Persing, CMO and CTO at Cepheid. "This helps us to avoid dead volume losses incurred with competitive first generation systems and to achieve unprecedented sensitivity levels for detection of HCV RNA."
Related Links:
Cepheid
The Cepheid (Sunnyvale, CA, USA) Xpert HCV Viral Load assay was recently awarded CE IVD mark status under the European Directive. The test was designed to run on the Cepheid GeneXpert System, one of the world's leading molecular diagnostic platforms with over 8,000 systems deployed globally in both developed and emerging market countries.
Cepheid's GeneXpert System is a closed, self-contained, fully-integrated, and automated platform that combines on-board sample preparation with real-time PCR (polymerase chain reaction) amplification and detection functions for fully integrated and automated nucleic acid analysis. The system is designed to purify, concentrate, detect, and identify targeted nucleic acid sequences thereby delivering answers directly from unprocessed samples.
The Xpert HCV Viral Load assay is intended for use as an aid in the management of HCV infected patients undergoing antiviral therapy. The test can be used to measure HCV RNA levels at baseline and during treatment, and to help predict virological responses to HCV therapy. In addition, the test can be used for confirmation of HCV serologic test results.
"Existing HCV viral load tests run on complex systems that require highly-trained technicians to operate, and generally lack the flexibility to provide on-demand answers when required," said John Bishop, chairman and CEO of Cepheid. "Xpert HCV Viral Load, our 21st CE-IVD Xpert test, provides reliable results in less than two hours, making it a valuable tool for clinicians in the management of HCV infected patients on antiviral therapy."
"The Xpert HCV Viral Load test incorporates a number of novel technologies including our "BigTube" design, which allows us to take full advantage of the efficiency of integrated GeneXpert cartridge fluidics," said Dr. David H. Persing, CMO and CTO at Cepheid. "This helps us to avoid dead volume losses incurred with competitive first generation systems and to achieve unprecedented sensitivity levels for detection of HCV RNA."
Related Links:
Cepheid
Read the full article by registering today, it's FREE! 

Register now for FREE to LabMedica.com and get access to news and events that shape the world of Clinical Laboratory Medicine. 
- Free digital version edition of LabMedica International sent by email on regular basis
- Free print version of LabMedica International magazine (available only outside USA and Canada).
- Free and unlimited access to back issues of LabMedica International in digital format
- Free LabMedica International Newsletter sent every week containing the latest news
- Free breaking news sent via email
- Free access to Events Calendar
- Free access to LinkXpress new product services
- REGISTRATION IS FREE AND EASY!
Sign in: Registered website members
Sign in: Registered magazine subscribers
Latest Microbiology News
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
- Rapid Sequencing Could Transform Tuberculosis Care
- Blood-Based Viral Signature Identified in Crohn’s Disease
- Hidden Gut Viruses Linked to Colorectal Cancer Risk
- Three-Test Panel Launched for Detection of Liver Fluke Infections
Channels
Clinical Chemistry
view channel
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read more
Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
Routine blood tests that measure low-density lipoprotein (LDL), commonly known as “bad” cholesterol, are widely used to guide lipid-lowering therapy, but they do not always provide a complete picture of... Read more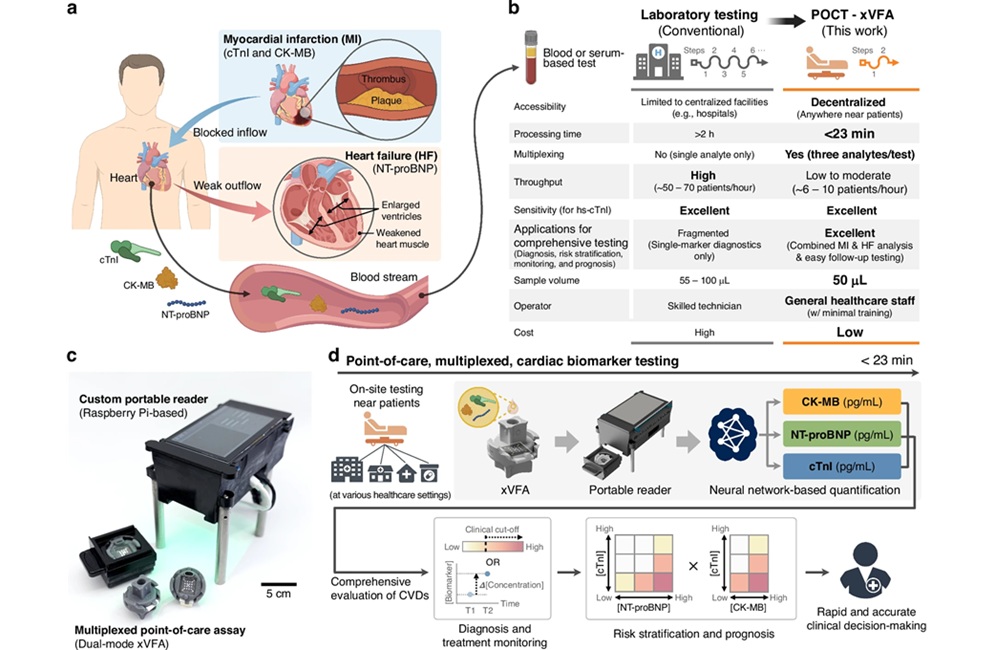
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel
Blood-Based Epigenetic Signals Enable Osteosarcoma Disease Monitoring
Osteosarcoma is a rare but aggressive pediatric bone cancer where recurrence and metastasis remain difficult to detect early. Imaging-based surveillance can miss small lesions and exposes children to repeated... Read more
Host–Virus Genetic Interactions Drive Nasopharyngeal Cancer Risk
Epstein–Barr virus (EBV) infects more than 95% of adults worldwide, yet only a small fraction develops EBV‑associated cancers such as nasopharyngeal carcinoma. Explaining this divergence requires understanding... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read morePathology
view channel
Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
Colorectal cancer is common and often lethal, and therapeutic decision-making is complicated by heterogeneous tumor microenvironments. Immunotherapy benefits only a small subset of patients, around 5%,... Read more
Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
Antibody-drug conjugates (ADC) are reshaping oncology, yet scalable biomarkers that reliably predict which patients will benefit remain limited as treatment regimens and combinations grow more complex.... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
Partnership Expands Ultrasensitive WGS Assay for for Hematologic Malignancies and MRD Monitoring
Tempus AI and Predicta Biosciences announced the commercial expansion of a co-branded whole‑genome sequencing assay GenoPredicta, which is intended for comprehensive genomic characterization of hematologic... Read more