Highly Sensitive Method Detects Malaria Parasites
|
By LabMedica International staff writers Posted on 18 Mar 2015 |
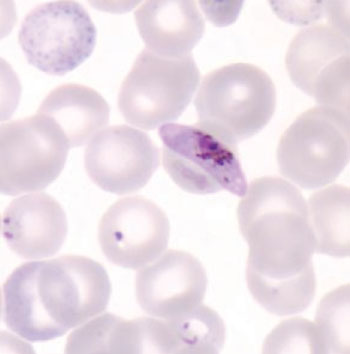
Image: A gametocyte of Plasmodium falciparum in a thin blood smear and also seen are ring-form trophozoites and a red blood cell exhibiting basophilic stippling (Photo courtesy of Dr. Mae Melvin).
A large proportion of asymptomatic malaria infections can only be identified by surveillance with molecular methods, yet these infections also contribute to onward transmission to mosquitoes.
New molecular assays that take advantage of genes with multiple copies in the parasite genome have been developed that can detect malaria parasites in human blood at very low levels and might be helpful in the campaign to eradicate malaria.
An international team of scientists led by those at the Swiss Tropical and Public Health Institute (Basel, Switzerland) compared three methods to detect malaria parasites in 498 samples randomly selected from a malaria survey in Tanzania: light microscopy, the current standard molecular assay, and the new assays. Two quantitative polymerase chain reaction (qPCR) assays were developed for ultra-sensitive detection of Plasmodium falciparum, targeting the high-copy telomere-associated repetitive element 2 (TARE-2, ~250 copies/genome) and the var gene acidic terminal sequence (varATS, 59 copies/genome).
Parasites were detected in 25% of samples by light microscopy, in 50% by the standard assay, and in 58% by the new assays. Compared to the new assays, the current molecular standard assay failed to identify 16% of infections, and at least 40% of those contained parasite gametocytes, the parasite stage that is transmitted when mosquitoes bite an infected person. Standard PCR is widely considered a molecular gold standard of malaria diagnosis complementing light microscopy, the traditional gold standard, yet these results suggest that this notion requires revision.
The new assays detect only the most common malaria parasite, P. falciparum, and while they can use very small blood samples collected in the field, the analysis itself needs to be done in a biomedical laboratory. Nonetheless, because low-density infections without disease symptoms are expected to become increasingly common as countries improve malaria control, ultra-sensitive tools such as these will likely be critical for malaria surveillance and for monitoring the progress of malaria control and elimination programs. The study was published on March 3, 2015, in the journal Public Library of Science Medicine.
Related Links:
Swiss Tropical and Public Health Institute
New molecular assays that take advantage of genes with multiple copies in the parasite genome have been developed that can detect malaria parasites in human blood at very low levels and might be helpful in the campaign to eradicate malaria.
An international team of scientists led by those at the Swiss Tropical and Public Health Institute (Basel, Switzerland) compared three methods to detect malaria parasites in 498 samples randomly selected from a malaria survey in Tanzania: light microscopy, the current standard molecular assay, and the new assays. Two quantitative polymerase chain reaction (qPCR) assays were developed for ultra-sensitive detection of Plasmodium falciparum, targeting the high-copy telomere-associated repetitive element 2 (TARE-2, ~250 copies/genome) and the var gene acidic terminal sequence (varATS, 59 copies/genome).
Parasites were detected in 25% of samples by light microscopy, in 50% by the standard assay, and in 58% by the new assays. Compared to the new assays, the current molecular standard assay failed to identify 16% of infections, and at least 40% of those contained parasite gametocytes, the parasite stage that is transmitted when mosquitoes bite an infected person. Standard PCR is widely considered a molecular gold standard of malaria diagnosis complementing light microscopy, the traditional gold standard, yet these results suggest that this notion requires revision.
The new assays detect only the most common malaria parasite, P. falciparum, and while they can use very small blood samples collected in the field, the analysis itself needs to be done in a biomedical laboratory. Nonetheless, because low-density infections without disease symptoms are expected to become increasingly common as countries improve malaria control, ultra-sensitive tools such as these will likely be critical for malaria surveillance and for monitoring the progress of malaria control and elimination programs. The study was published on March 3, 2015, in the journal Public Library of Science Medicine.
Related Links:
Swiss Tropical and Public Health Institute
Latest Microbiology News
- Label-Free Microscopy Methodd Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
Channels
Clinical Chemistry
view channel
Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
Serum proteins underpin many routine tests used to detect inflammation, hormonal imbalance, cardiovascular disease, and metabolic disorders. Yet pediatric interpretation often relies on adult reference... Read more
Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
Alzheimer’s disease and related dementias develop over years, making it difficult to identify at-risk patients before symptoms appear. Clinicians therefore need widely available laboratory markers that... Read more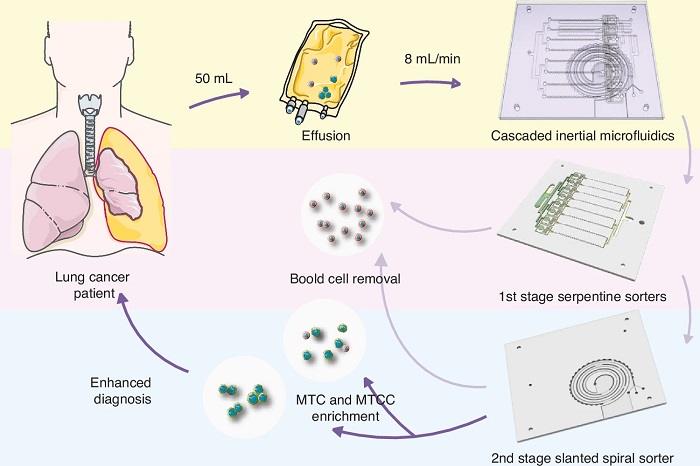
Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
Diagnosing malignancy from pleural effusion remains challenging because tumor cells are rare and clusters are easily disrupted during processing. Conventional cytology can miss malignant tumor cells and... Read moreMolecular Diagnostics
view channel
AI Blood Test Enhances Monitoring of Liver Cirrhosis Progression
Monitoring chronic liver disease remains difficult because clinicians rely on tools that can be inconsistent and may miss early progression. Standard approaches often combine ultrasound imaging with blood-based... Read more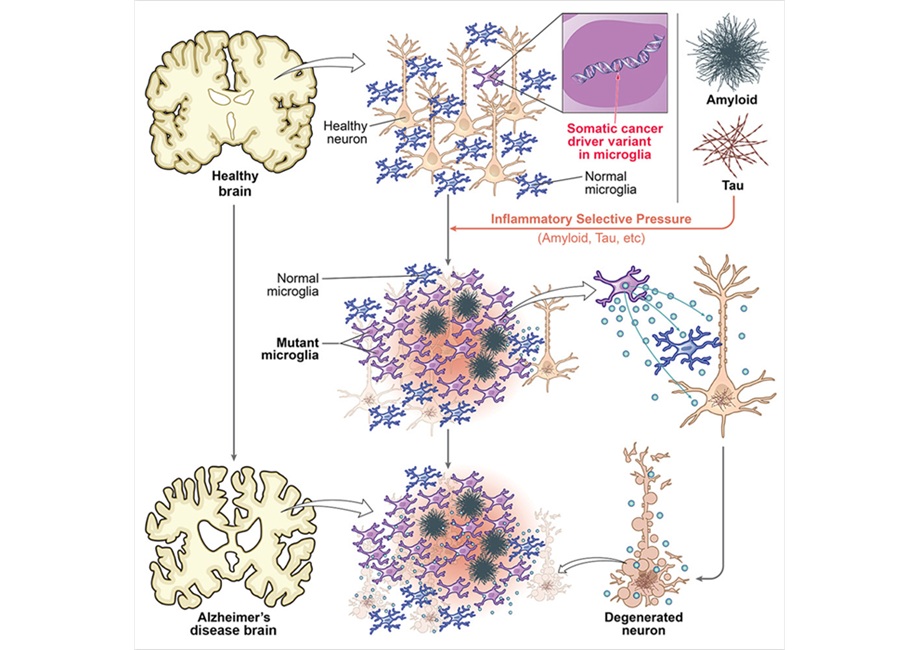
Cancer-Related Mutations in Immune Cells Linked to Alzheimer’s
Alzheimer’s disease is marked by protein aggregation and inflammatory changes in the brain’s immune system, yet its molecular drivers remain incompletely understood. With aging, human cells accumulate... Read more
Composite Blood Biomarkers Enable Early Detection of Common Cancers
Early diagnosis of colorectal, lung, and ovarian cancers remains challenging, with many patients identified only after tumors have begun to spread. A scalable blood test could expand access to screening,... Read more
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read moreHematology
view channel
Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
CareDx (Brisbane, CA, USA) has introduced AlloSeq Nano, a nanopore‑based HLA (human leukocyte antigen) and ABO genotyping solution unveiled at the European Federation for Immunogenetics (EFI) Conference 2026.... Read more
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read morePathology
view channel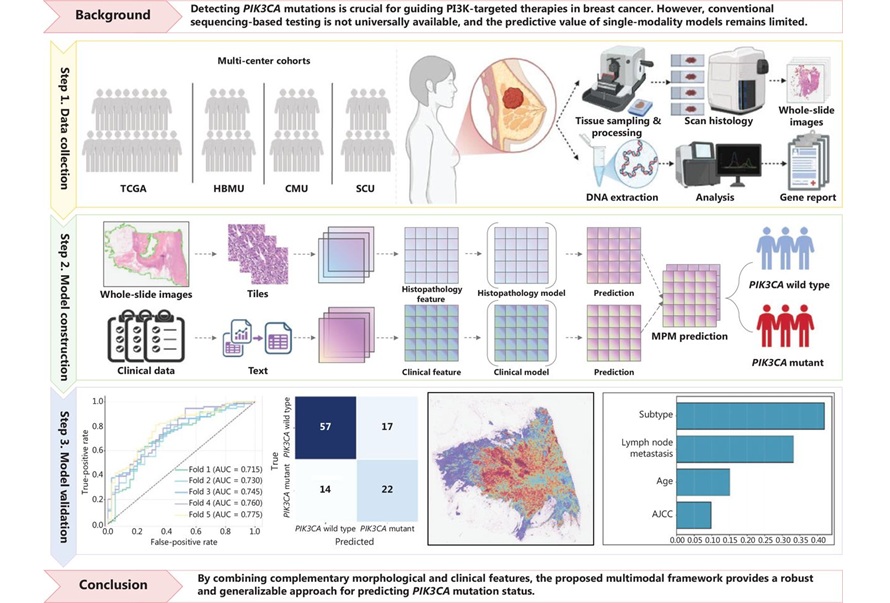
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read more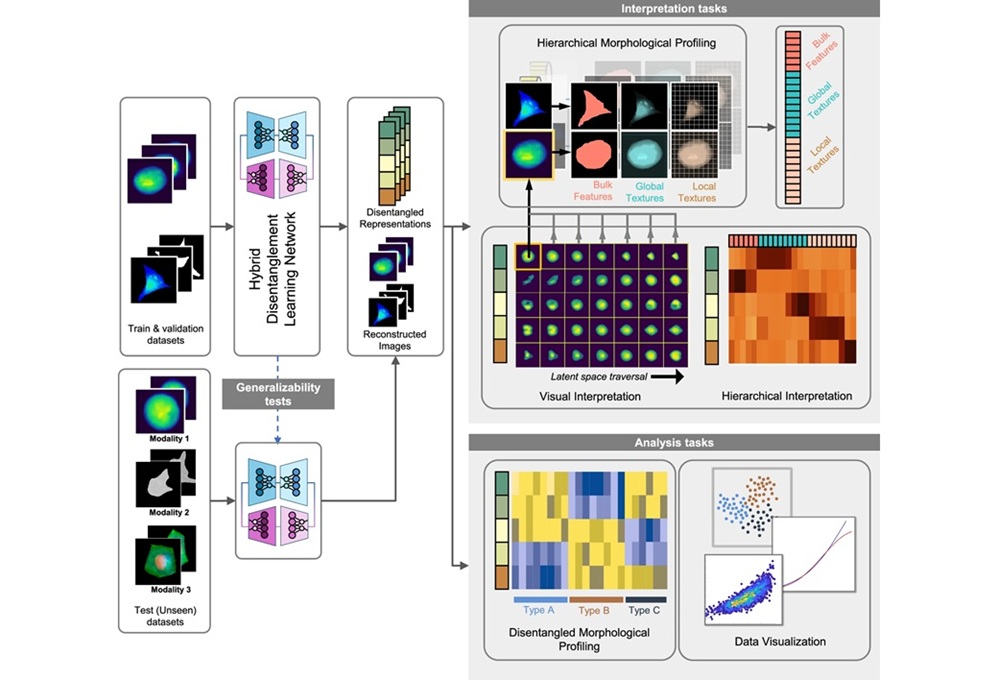
Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
Microscopy images contain rich clues about cell health, but many disease-relevant morphological differences are too subtle to see and difficult to quantify consistently. Artificial intelligence (AI) has... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more