Rapid Lateral Flow Assay Detects Several Viral Fevers Simultaneously
|
By LabMedica International staff writers Posted on 10 Mar 2015 |

Image: The diagnostic system is based on triangular silver nanoparticles that display different colors depending on their size (Photo courtesy of the Massachusetts Institute of Technology).
A team of biomedical engineers has developed a rapid diagnostic test that is able to detect several viruses such as Ebola, dengue fever, and yellow fever simultaneously from a single sample of patient's serum.
Investigators at the Massachusetts Institute of Technology (Cambridge, MA, USA) and Harvard Medical School (Boston, MA, USA) used a novel class of silver nanoparticles to drive a paper-based lateral flow assay. Lateral flow assays are commonly used to detect such pathogens as Rotavirus, adenovirus, Streptococcus A and B, and gonorrhea and in rapid pregnancy tests.
The investigators extended the usefulness of the lateral flow approach by incorporating antibodies bound to different colored nanoparticles. In the current version of the assay, following a 10 minute incubation, an orange band indicates the presence of yellow fever virus, a red band indicates Ebolavirus, and a green band indicates dengue fever virus.
The investigators expect that their new rapid test will complement more definitive diagnostic technologies, such as PCR. "If you are in a situation in the field with no power and no special technologies, if you want to know if a patient has Ebola, this test can tell you very quickly that you might not want to put that patient in a waiting room with other people who might not be infected," said senior author Dr. Lee Gehrke, professor of microbiology and immunology at Harvard Medical School. "That initial triage can be very important from a public health standpoint, and there could be a follow-up test later with PCR or something to confirm."
This type of diagnostic testing device is readily customizable to detect other infectious diseases, by linking the silver nanoparticles to different antibodies. "Thankfully the Ebola outbreak is dying off, which is a good thing, but what we are thinking about is what is coming next," said Dr. Gehrke. "There will undoubtedly be other viral outbreaks. It might be Sudan virus, or it might be another hemorrhagic fever. What we are trying to do is develop the antibodies needed to be ready for the next outbreak that is going to happen."
The rapid diagnostic test for multiple viral fevers was described in the February 12, 2015, online edition of the journal Lab on a Chip.
Related Links:
Massachusetts Institute of Technology
Harvard Medical School
Investigators at the Massachusetts Institute of Technology (Cambridge, MA, USA) and Harvard Medical School (Boston, MA, USA) used a novel class of silver nanoparticles to drive a paper-based lateral flow assay. Lateral flow assays are commonly used to detect such pathogens as Rotavirus, adenovirus, Streptococcus A and B, and gonorrhea and in rapid pregnancy tests.
The investigators extended the usefulness of the lateral flow approach by incorporating antibodies bound to different colored nanoparticles. In the current version of the assay, following a 10 minute incubation, an orange band indicates the presence of yellow fever virus, a red band indicates Ebolavirus, and a green band indicates dengue fever virus.
The investigators expect that their new rapid test will complement more definitive diagnostic technologies, such as PCR. "If you are in a situation in the field with no power and no special technologies, if you want to know if a patient has Ebola, this test can tell you very quickly that you might not want to put that patient in a waiting room with other people who might not be infected," said senior author Dr. Lee Gehrke, professor of microbiology and immunology at Harvard Medical School. "That initial triage can be very important from a public health standpoint, and there could be a follow-up test later with PCR or something to confirm."
This type of diagnostic testing device is readily customizable to detect other infectious diseases, by linking the silver nanoparticles to different antibodies. "Thankfully the Ebola outbreak is dying off, which is a good thing, but what we are thinking about is what is coming next," said Dr. Gehrke. "There will undoubtedly be other viral outbreaks. It might be Sudan virus, or it might be another hemorrhagic fever. What we are trying to do is develop the antibodies needed to be ready for the next outbreak that is going to happen."
The rapid diagnostic test for multiple viral fevers was described in the February 12, 2015, online edition of the journal Lab on a Chip.
Related Links:
Massachusetts Institute of Technology
Harvard Medical School
Latest Microbiology News
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Methodd Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Channels
Clinical Chemistry
view channel
Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
Serum proteins underpin many routine tests used to detect inflammation, hormonal imbalance, cardiovascular disease, and metabolic disorders. Yet pediatric interpretation often relies on adult reference... Read more
Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
Alzheimer’s disease and related dementias develop over years, making it difficult to identify at-risk patients before symptoms appear. Clinicians therefore need widely available laboratory markers that... Read more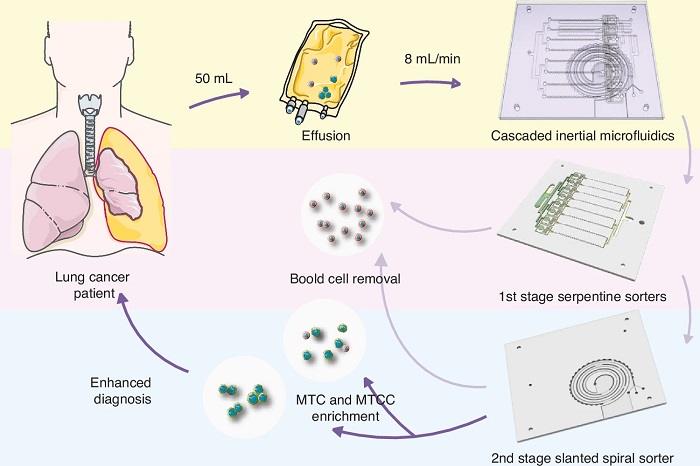
Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
Diagnosing malignancy from pleural effusion remains challenging because tumor cells are rare and clusters are easily disrupted during processing. Conventional cytology can miss malignant tumor cells and... Read moreMolecular Diagnostics
view channel
Genetic Signature Predicts Myeloid Leukemia Risk in Down Syndrome
Children with Down syndrome face a markedly increased risk of myeloid leukemia, yet early lesions and pre-cancerous cells can appear indistinguishable under the microscope. Many are born with a transient... Read more
Gene Expression Model Guides Neoadjuvant Therapy Selection in Breast Cancer
Predicting which patients with early breast cancer will benefit from chemotherapy before surgery remains difficult. Reliable upfront markers are lacking. In hormone-dependent, HER2-negative disease, overall... Read moreHematology
view channel
Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
CareDx (Brisbane, CA, USA) has introduced AlloSeq Nano, a nanopore‑based HLA (human leukocyte antigen) and ABO genotyping solution unveiled at the European Federation for Immunogenetics (EFI) Conference 2026.... Read more
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read morePathology
view channel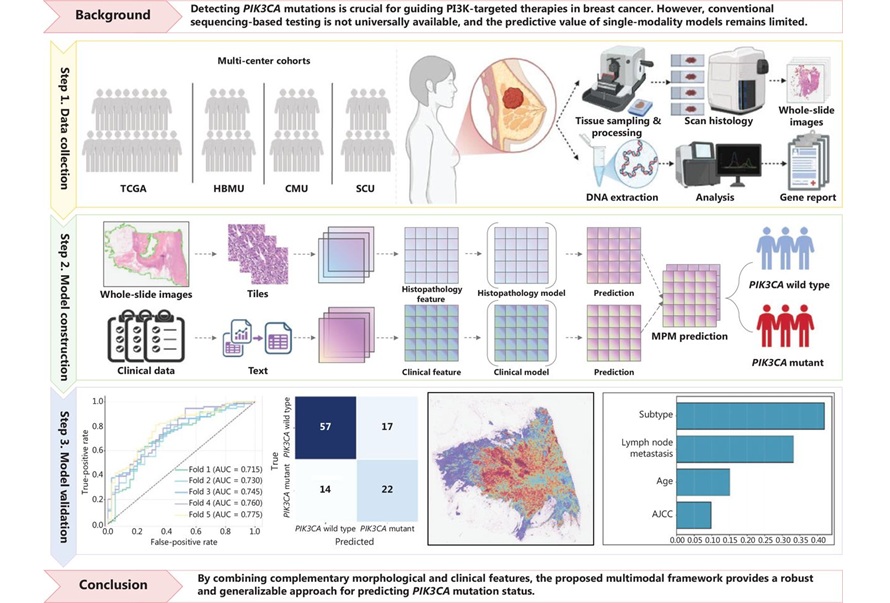
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read more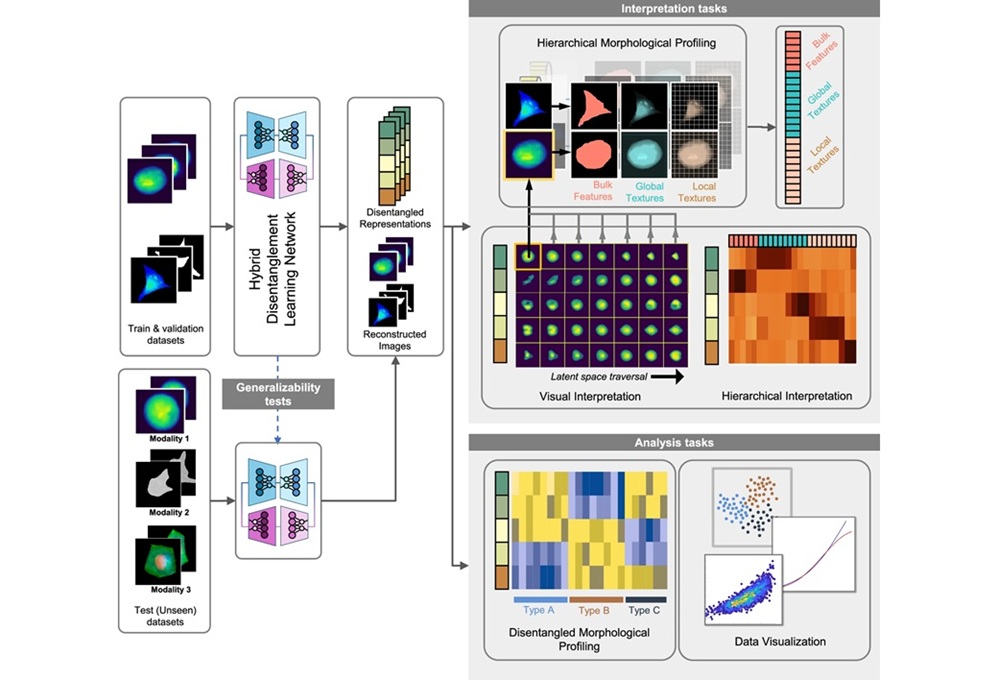
Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
Microscopy images contain rich clues about cell health, but many disease-relevant morphological differences are too subtle to see and difficult to quantify consistently. Artificial intelligence (AI) has... Read moreTechnology
view channel
Microfluidic Single-Cell Assay Predicts Breast Cancer Risk
Risk stratification for breast cancer remains imprecise, as population-based models and breast density can over- or underestimate individual risk, potentially leading to over- or under-screening.... Read more
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read moreIndustry
view channel
Collaboration Expands Access to Rapid Metagenomic Diagnostics for Complex Infections
Hospitals are seeing rising rates of complicated and healthcare-associated infections, especially in immunocompromised patients, intensifying the need for rapid, comprehensive pathogen detection.... Read more