FDA Clears Hand-Held Test for Bordetella Pertussis
|
By LabMedica International staff writers Posted on 04 Jan 2015 |
The United States Food and Drug Administration (FDA) have cleared an accurate, easy-to-use molecular diagnostic assay for Bordetella Pertussis that employs a unique hand-held platform and requires no pre-extraction of DNA.
The incidence of whooping cough, the very contagious disease caused by B. pertussis attaching to cilia that line part of the upper respiratory tract, has risen over the last few years. Whooping cough can cause serious illness in children and adults, and is most dangerous for infants and babies. Quidel Corporation (San Diego, CA, USA), a developer and provider of new diagnostic solutions, has received the FDA clearance for its AmpliVue Bordetella Assay to detect B. pertussis nucleic acids isolated from nasopharyngeal swab specimens obtained from patients suspected of having a respiratory tract infection attributable to B. pertussis.
AmpliVue Bordetella is an easy-to-use, self-contained, disposable molecular diagnostic test with superb clinical accuracy. The assay requires no upfront extraction of DNA and generates the result in approximately 75 minutes. The AmpliVue platform is based on Quidel’s novel Helicase Dependent Amplification (HDA) technology that enables rapid nucleic acid amplification without a thermal cycler. So, like all FDA-cleared AmpliVue assays, AmpliVue Bordetella is CLIA-classified as moderately complex and does not require investment in expensive thermocycling equipment. This benefit, plus less laboratory space requirements, can significantly lower cost when adopting new molecular testing methods.
"We are pleased to receive 510(k) clearance for our AmpliVue Bordetella Assay—our 5th assay in the AmpliVue format. We've shown that we can harness our proprietary HDA technology to develop fast, accurate molecular diagnostic assays with a menu that matters to our customers," said Douglas Bryant, president and CEO of Quidel, "Longer-term, we intend to leverage this technology for other molecular diagnostic applications that will be appealing to significant market segments and potentially to public health agencies worldwide."
Quidel's novel AmpliVue platform now enables laboratories of all sizes to perform highly sensitive and specific molecular tests, and currently has FDA clearance for B. Pertussis, C. difficile, Group A Strep, Group B Strep, HSV1 and HSV2.
Related Links:
Quidel Corporation
The incidence of whooping cough, the very contagious disease caused by B. pertussis attaching to cilia that line part of the upper respiratory tract, has risen over the last few years. Whooping cough can cause serious illness in children and adults, and is most dangerous for infants and babies. Quidel Corporation (San Diego, CA, USA), a developer and provider of new diagnostic solutions, has received the FDA clearance for its AmpliVue Bordetella Assay to detect B. pertussis nucleic acids isolated from nasopharyngeal swab specimens obtained from patients suspected of having a respiratory tract infection attributable to B. pertussis.
AmpliVue Bordetella is an easy-to-use, self-contained, disposable molecular diagnostic test with superb clinical accuracy. The assay requires no upfront extraction of DNA and generates the result in approximately 75 minutes. The AmpliVue platform is based on Quidel’s novel Helicase Dependent Amplification (HDA) technology that enables rapid nucleic acid amplification without a thermal cycler. So, like all FDA-cleared AmpliVue assays, AmpliVue Bordetella is CLIA-classified as moderately complex and does not require investment in expensive thermocycling equipment. This benefit, plus less laboratory space requirements, can significantly lower cost when adopting new molecular testing methods.
"We are pleased to receive 510(k) clearance for our AmpliVue Bordetella Assay—our 5th assay in the AmpliVue format. We've shown that we can harness our proprietary HDA technology to develop fast, accurate molecular diagnostic assays with a menu that matters to our customers," said Douglas Bryant, president and CEO of Quidel, "Longer-term, we intend to leverage this technology for other molecular diagnostic applications that will be appealing to significant market segments and potentially to public health agencies worldwide."
Quidel's novel AmpliVue platform now enables laboratories of all sizes to perform highly sensitive and specific molecular tests, and currently has FDA clearance for B. Pertussis, C. difficile, Group A Strep, Group B Strep, HSV1 and HSV2.
Related Links:
Quidel Corporation
Read the full article by registering today, it's FREE! 

Register now for FREE to LabMedica.com and get access to news and events that shape the world of Clinical Laboratory Medicine. 
- Free digital version edition of LabMedica International sent by email on regular basis
- Free print version of LabMedica International magazine (available only outside USA and Canada).
- Free and unlimited access to back issues of LabMedica International in digital format
- Free LabMedica International Newsletter sent every week containing the latest news
- Free breaking news sent via email
- Free access to Events Calendar
- Free access to LinkXpress new product services
- REGISTRATION IS FREE AND EASY!
Sign in: Registered website members
Sign in: Registered magazine subscribers
Latest Microbiology News
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
- Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
- Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
- WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
- New Imaging Approach Could Help Predict Dangerous Gut Infection
Channels
Clinical Chemistry
view channel
Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
Early detection across multiple cancers remains a major unmet need in population screening. Non-invasive approaches that can be delivered at scale may broaden access and shift diagnoses to earlier stages.... Read more
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read moreMolecular Diagnostics
view channel
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read more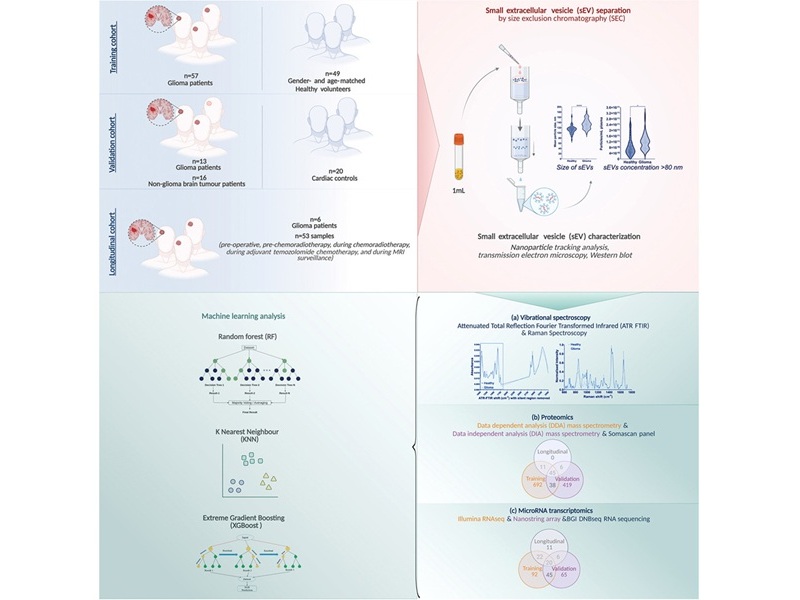
Blood Test Enables Early Detection and Classification of Glioma
High-grade gliomas, particularly glioblastoma, are fast-growing brain tumors that are often diagnosed late and typically require invasive procedures for confirmation. Current pathways rely on symptoms,... Read more
Multi-Biomarker Blood Test Detects Early-Stage Cancers Across Types
Abbott is showcasing its Cancerguard multi-cancer early detection (MCED) test at the American Association for Cancer Research (AACR) Annual Meeting 2026, where new data highlight continued progress in... Read more
New Sample-to-Answer PCR System Supports High-Throughput Infectious Disease Testing
Clinical laboratories face mounting demand for rapid, high‑volume molecular testing for infectious diseases, including routine monitoring in immunocompromised patients. Consolidated, sample‑to‑answer workflows... Read moreHematology
view channel
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read more
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read morePathology
view channel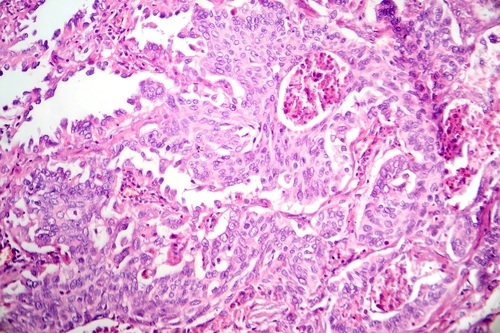
AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
Clinicians face persistent challenges identifying which patients with metastatic non-small cell lung cancer will benefit from immunotherapy, even as these agents transform oncology care.... Read more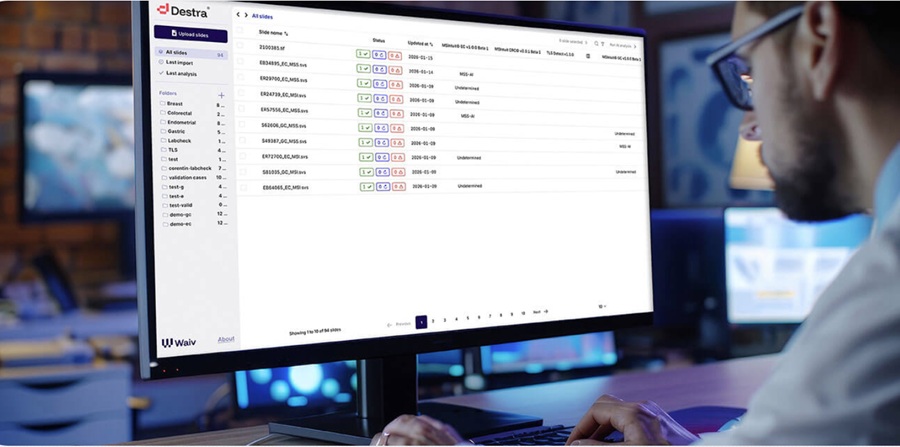
AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Reliable prognostic profiling and biomarker screening are essential to guide oncology treatment decisions, while laboratories must balance speed and resource constraints. Earlier identification of high‑risk... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more