New Multigene Test Improves Assessment of Hereditary Cancers
|
By LabMedica International staff writers Posted on 09 Jun 2014 |

Image: The Myriad myRisk Hereditary Cancer test uses next-generation sequencing to evaluate multiple clinically significant hereditary cancer genes associated with eight major hereditary cancers (Photo courtesy of Myriad Genetics).
New clinical data shows that a test covering multiple relevant genes not only detects more deleterious mutations than single cancer tests, but also helps solve the overlap-dilemma that exists among hereditary cancer syndromes.
Myriad Genetics, Inc. (Salt Lake City, UT, USA) has presented clinical studies on the "Myriad myRisk Hereditary Cancer" test at the 50th annual meeting of the American Society of Clinical Oncology (ASCO; Chicago, IL, USA), May 30–June 3, 2014. The test uses next-generation sequencing (NGS) to evaluate 25 clinically significant hereditary cancer genes associated with 8 major hereditary cancers including: breast, colon, ovarian, endometrial, pancreatic, prostate, gastric cancers, and melanoma. The myRisk test results are combined with a patient's personal and family history of cancer and medical society guidelines into a single comprehensive report, making it easier for physicians to tailor treatment plans for patients depending on their level of risk.
"There is robust evidence that hereditary cancers are caused by mutations in many genes and testing for only one hereditary cancer syndrome may lead to missed mutations," said Richard J. Wenstrup, MD, chief medical officer of Myriad, "The Myriad myRisk test solves this dilemma."
The following key studies were featured at ASCO: "Multigene panel testing in patients suspected to have Lynch syndrome," podium presentation by Matthew Yurgelun. This study, using myRisk to test 1,260 patients with a history of hereditary colon cancer, found that 27% of mutation carriers identified by myRisk had mutations in genes not normally associated with hereditary colon cancer. Also important is that more than 1/3 of the additional mutations found were in the BRCA1 and BRCA2 genes, which further demonstrates the overlap that exists between the hereditary breast and colon cancer syndromes.
"A study of ovarian cancer patients tested with a 25-gene panel of hereditary cancer genes," podium presentation by Lucy Langer. This study of 648 patients showed that 15.4% of patients with ovarian cancer had a mutation that was detected by the myRisk panel. Of these patients, 59.6% had mutations in BRCA1 and BRCA2, 34.6% had mutations in the other myRisk panel genes, including hereditary colon cancer genes. myRisk increased the number of positive test results in ovarian cancer patients by 63% over BRCA1 and BRCA2 testing alone.
"Analysis of patients with two hereditary cancers (breast/ovarian or colon/endometrial) who met NCCN genetic testing criteria after their first cancer," poster presentation by Jennifer Saam. In this study, patients with a history of 2 associated cancers were evaluated to determine what percentage of patients met National Comprehensive Cancer Network (NCCN) criteria for genetic testing after their first cancer diagnosis, but who did not receive a test result until after their second cancer diagnosis. The majority of patients had at least 5 years between their first and second cancers. For 9,982 patients with breast and ovarian cancer who were evaluated, the overall rate of BRCA1 and BRCA2 mutations was 22%. Of these, only 56% of patients diagnosed with breast and ovarian cancer met the NCCN criteria for genetic testing after their first cancer. Among 941 patients with colon and endometrial cancer who were evaluated, 28% had mutations in the genes associated with hereditary colon cancer. Of these, 65% of patients met NCCN criteria for genetic testing after their first cancer. These findings underscore the importance of diagnosing patients with hereditary cancer syndromes after their first cancer so that a second cancer can be prevented or identified early.
Related Links:
Myriad Genetics
Myriad Genetics, Inc. (Salt Lake City, UT, USA) has presented clinical studies on the "Myriad myRisk Hereditary Cancer" test at the 50th annual meeting of the American Society of Clinical Oncology (ASCO; Chicago, IL, USA), May 30–June 3, 2014. The test uses next-generation sequencing (NGS) to evaluate 25 clinically significant hereditary cancer genes associated with 8 major hereditary cancers including: breast, colon, ovarian, endometrial, pancreatic, prostate, gastric cancers, and melanoma. The myRisk test results are combined with a patient's personal and family history of cancer and medical society guidelines into a single comprehensive report, making it easier for physicians to tailor treatment plans for patients depending on their level of risk.
"There is robust evidence that hereditary cancers are caused by mutations in many genes and testing for only one hereditary cancer syndrome may lead to missed mutations," said Richard J. Wenstrup, MD, chief medical officer of Myriad, "The Myriad myRisk test solves this dilemma."
The following key studies were featured at ASCO: "Multigene panel testing in patients suspected to have Lynch syndrome," podium presentation by Matthew Yurgelun. This study, using myRisk to test 1,260 patients with a history of hereditary colon cancer, found that 27% of mutation carriers identified by myRisk had mutations in genes not normally associated with hereditary colon cancer. Also important is that more than 1/3 of the additional mutations found were in the BRCA1 and BRCA2 genes, which further demonstrates the overlap that exists between the hereditary breast and colon cancer syndromes.
"A study of ovarian cancer patients tested with a 25-gene panel of hereditary cancer genes," podium presentation by Lucy Langer. This study of 648 patients showed that 15.4% of patients with ovarian cancer had a mutation that was detected by the myRisk panel. Of these patients, 59.6% had mutations in BRCA1 and BRCA2, 34.6% had mutations in the other myRisk panel genes, including hereditary colon cancer genes. myRisk increased the number of positive test results in ovarian cancer patients by 63% over BRCA1 and BRCA2 testing alone.
"Analysis of patients with two hereditary cancers (breast/ovarian or colon/endometrial) who met NCCN genetic testing criteria after their first cancer," poster presentation by Jennifer Saam. In this study, patients with a history of 2 associated cancers were evaluated to determine what percentage of patients met National Comprehensive Cancer Network (NCCN) criteria for genetic testing after their first cancer diagnosis, but who did not receive a test result until after their second cancer diagnosis. The majority of patients had at least 5 years between their first and second cancers. For 9,982 patients with breast and ovarian cancer who were evaluated, the overall rate of BRCA1 and BRCA2 mutations was 22%. Of these, only 56% of patients diagnosed with breast and ovarian cancer met the NCCN criteria for genetic testing after their first cancer. Among 941 patients with colon and endometrial cancer who were evaluated, 28% had mutations in the genes associated with hereditary colon cancer. Of these, 65% of patients met NCCN criteria for genetic testing after their first cancer. These findings underscore the importance of diagnosing patients with hereditary cancer syndromes after their first cancer so that a second cancer can be prevented or identified early.
Related Links:
Myriad Genetics
Latest Molecular Diagnostics News
- Framework Guides Targeted Immunotherapy Selection in Liver Cancer
- Collaboration Brings Rapid At-Home STI Testing with Virtual Follow-Up
- Blood-Based Epigenetic Signals Enable Osteosarcoma Disease Monitoring
- Host–Virus Genetic Interactions Drive Nasopharyngeal Cancer Risk
- AI-Enabled Biochip Detects microRNA Biomarkers in Minutes
- Blood Test Detects Early Pancreatic Cancer in High-Risk Patients
- Long-Read RNA Sequencing Platform Improves Rare Disease Diagnosis
- Study Confirms Barrett’s Esophagus as Precursor to Esophageal Cancer
- Ultrasensitive Assay Reveals Previously Undetected Tuberculosis in Hospital Patients
- CE-Marked Blood Test Enables Monitoring of Neuroinflammation in Multiple Sclerosis
- Urine-Based Assay Predicts Severe Dengue Risk Early
- Ultrasensitive Assay Tracks Resistance Mutations MRD Monitoring
- FDA Clears At-Home HPV Test with Extended Genotyping for Cervical Screening
- Extracellular Vesicle RNA Biomarkers Enable Noninvasive IBD Diagnosis and Monitoring
- New Gene Signature Reveals Underdiagnosed Lung Cancer Subtype
- Genome Sequencing Identifies Noncoding Variants Causing Neonatal Diabetes
Channels
Clinical Chemistry
view channel
Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Alzheimer's disease often advances silently for years, making timely risk stratification difficult in routine practice. Current approaches to detect pathology can involve lumbar puncture or positron emission... Read more
Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
Routine blood tests that measure low-density lipoprotein (LDL), commonly known as “bad” cholesterol, are widely used to guide lipid-lowering therapy, but they do not always provide a complete picture of... Read more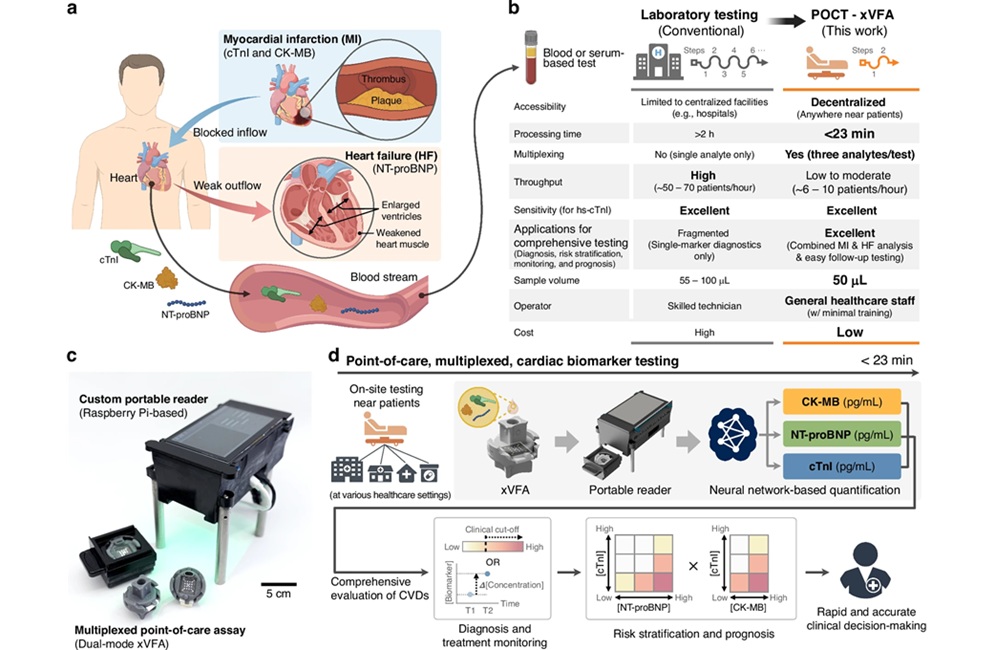
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreMicrobiology
view channel
mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
Central nervous system (CNS) infections such as encephalitis, ventriculitis, and brain abscess are among the most time-sensitive diagnostic challenges in neurology. Standard testing can return negative... Read more
Rapid Color Test Stratifies Virulent and Resistant Staph Strains
Staphylococcus aureus (golden staph) remains a leading cause of infection-related mortality worldwide, responsible for more than a million deaths each year. Rapidly distinguishing highly virulent or a... Read morePathology
view channel
AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
Reliable prognostic profiling and biomarker screening are essential to guide oncology treatment decisions, while laboratories must balance speed and resource constraints. Earlier identification of high‑risk... Read more
Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
Cutaneous squamous cell carcinoma (cSCC) is the world’s second most common skin cancer, and while many cases are treatable, a subset becomes highly aggressive and therapy‑resistant. Identifying molecular... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more