New System Breaks Technology and Cost Barriers for High-Throughput Large-Genome Sequencing
|
By LabMedica International staff writers Posted on 28 Jan 2014 |

Image: The HiSeq X Ten, composed of 10 HiSeq X Systems, breaks the USD 1000 barrier for a 30x human genome, enabling population-scale projects on genotypic variation to understand and improve human health (Photo courtesy of Illumina).
A new DNA sequencing system utilizes advanced design features to generate massive throughput and enable the world's first USD 1000 human genome sequence.
This achievement has been reached with the new HiSeq X Ten Sequencing System from Illumina (San Diego, CA, USA). The platform includes technology breakthroughs that enable researchers to undertake population and disease studies of unprecedented scale by providing the throughput to sequence tens of thousands of human whole genomes in a single year in a single lab. The HiSeq X Ten is the world’s first platform to deliver high quality, high-coverage human genome sequences for less than USD 1,000—inclusive of typical instrument depreciation, DNA extraction, library preparation, and estimated labor.
Purpose-built for population-scale human whole genome sequencing, the HiSeq X Ten is a platform especially suitable for scientists and institutions focused on the discovery of genotypic variation to enable a deeper understanding of biology and disease. It can deliver a comprehensive catalog of human variation within and outside of coding regions. “The ability to explore the human genome on this scale will bring the study of cancer and complex diseases to a new level. Breaking the ‘sound barrier’ of human genetics not only pushes us through a psychological milestone, it enables projects of unprecedented scale. We are excited to see what lies on the other side,” said Jay Flatley, CEO, Illumina.
Building on the proven performance of Illumina sequencing-by-synthesis (SBS) technology, HiSeq X Ten utilizes a number of advanced design features to generate massive throughput. Patterned flow cells (which contain billions of nanowells at fixed locations) combined with a new clustering chemistry deliver a significant increase in data density (6 billion clusters per run). Using state-of-the art optics and faster chemistry, HiSeq X Ten can process sequencing flow cells more quickly than ever before – generating a 10x increase in daily throughput when compared to current HiSeq 2500 performance. The HiSeq X Ten is sold as a set of 10 or more ultra-high throughput sequencing systems, each generating up to 1.8 terabases (Tb) of sequencing data in less than 3 days or up to 600 gigabases (Gb) per day, per system.
Initial users of the transformative HiSeq X Ten System include Macrogen (Seoul, Republic of Korea) and its CLIA laboratory (Rockville, MD, USA), the Broad Institute (Cambridge, MA, USA), and the Garvan Institute of Medical Research (Sydney, Australia).
“The sequencing capacity and economies of scale of the HiSeq X Ten facility will also allow Garvan to accelerate the introduction of clinical genomics and next-generation medicine in Australia,” said Prof. John Mattick, Executive Director of the Garvan Institute of Medical Research.
Eric Lander, founding director of the Broad Institute and professor of biology at MIT, said, “The HiSeq X Ten should give us the ability to analyze complete genomic information from huge sample populations. Over the next few years, we have an opportunity to learn as much about the genetics of human disease as we have learned in the history of medicine.”
“Macrogen will deploy this groundbreaking technology to open a new era of large-scale, whole genome sequencing in our certified CLIA laboratory,” said Dr. Jeong-Sun Seo, Chairman of Macrogen; “Additionally, we will use the HiSeq X Ten to continue our collaboration with the Genomic Medicine Institute of Seoul National University focused on sequencing Asian populations in order to build a genomics database for use in medical research and healthcare applications.”
Related Links:
Illumina
This achievement has been reached with the new HiSeq X Ten Sequencing System from Illumina (San Diego, CA, USA). The platform includes technology breakthroughs that enable researchers to undertake population and disease studies of unprecedented scale by providing the throughput to sequence tens of thousands of human whole genomes in a single year in a single lab. The HiSeq X Ten is the world’s first platform to deliver high quality, high-coverage human genome sequences for less than USD 1,000—inclusive of typical instrument depreciation, DNA extraction, library preparation, and estimated labor.
Purpose-built for population-scale human whole genome sequencing, the HiSeq X Ten is a platform especially suitable for scientists and institutions focused on the discovery of genotypic variation to enable a deeper understanding of biology and disease. It can deliver a comprehensive catalog of human variation within and outside of coding regions. “The ability to explore the human genome on this scale will bring the study of cancer and complex diseases to a new level. Breaking the ‘sound barrier’ of human genetics not only pushes us through a psychological milestone, it enables projects of unprecedented scale. We are excited to see what lies on the other side,” said Jay Flatley, CEO, Illumina.
Building on the proven performance of Illumina sequencing-by-synthesis (SBS) technology, HiSeq X Ten utilizes a number of advanced design features to generate massive throughput. Patterned flow cells (which contain billions of nanowells at fixed locations) combined with a new clustering chemistry deliver a significant increase in data density (6 billion clusters per run). Using state-of-the art optics and faster chemistry, HiSeq X Ten can process sequencing flow cells more quickly than ever before – generating a 10x increase in daily throughput when compared to current HiSeq 2500 performance. The HiSeq X Ten is sold as a set of 10 or more ultra-high throughput sequencing systems, each generating up to 1.8 terabases (Tb) of sequencing data in less than 3 days or up to 600 gigabases (Gb) per day, per system.
Initial users of the transformative HiSeq X Ten System include Macrogen (Seoul, Republic of Korea) and its CLIA laboratory (Rockville, MD, USA), the Broad Institute (Cambridge, MA, USA), and the Garvan Institute of Medical Research (Sydney, Australia).
“The sequencing capacity and economies of scale of the HiSeq X Ten facility will also allow Garvan to accelerate the introduction of clinical genomics and next-generation medicine in Australia,” said Prof. John Mattick, Executive Director of the Garvan Institute of Medical Research.
Eric Lander, founding director of the Broad Institute and professor of biology at MIT, said, “The HiSeq X Ten should give us the ability to analyze complete genomic information from huge sample populations. Over the next few years, we have an opportunity to learn as much about the genetics of human disease as we have learned in the history of medicine.”
“Macrogen will deploy this groundbreaking technology to open a new era of large-scale, whole genome sequencing in our certified CLIA laboratory,” said Dr. Jeong-Sun Seo, Chairman of Macrogen; “Additionally, we will use the HiSeq X Ten to continue our collaboration with the Genomic Medicine Institute of Seoul National University focused on sequencing Asian populations in order to build a genomics database for use in medical research and healthcare applications.”
Related Links:
Illumina
Latest Technology News
- AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
- Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
- Noninvasive Sputum Test Detects Early Lung Cancer
- New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
- Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
- Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
- Online Tool Supports Family Screening for Inherited Cancer Risk
- Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
- New Electronic Pipette Enhances Workflows with Touchscreen Control
- AI Model Outperforms Clinicians in Rare Disease Detection
- AI-Driven Diagnostic Demonstrates High Accuracy in Detecting Periprosthetic Joint Infection
- Blood Test “Clocks” Predict Start of Alzheimer’s Symptoms
- AI-Powered Biomarker Predicts Liver Cancer Risk
Channels
Clinical Chemistry
view channel
Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
Serum proteins underpin many routine tests used to detect inflammation, hormonal imbalance, cardiovascular disease, and metabolic disorders. Yet pediatric interpretation often relies on adult reference... Read more
Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
Alzheimer’s disease and related dementias develop over years, making it difficult to identify at-risk patients before symptoms appear. Clinicians therefore need widely available laboratory markers that... Read more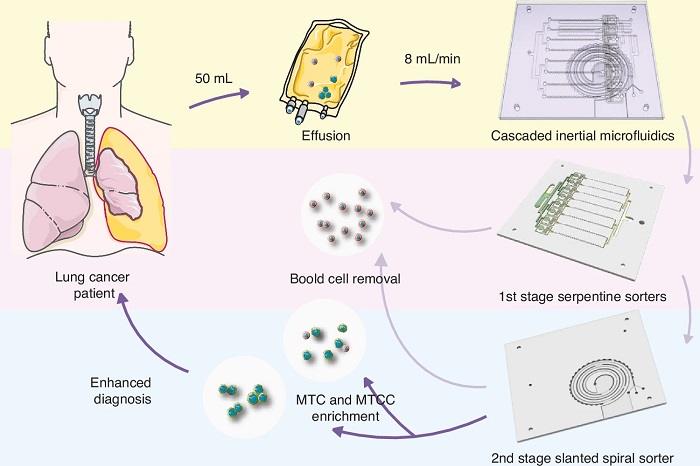
Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
Diagnosing malignancy from pleural effusion remains challenging because tumor cells are rare and clusters are easily disrupted during processing. Conventional cytology can miss malignant tumor cells and... Read moreMolecular Diagnostics
view channel
AI Blood Test Enhances Monitoring of Liver Cirrhosis Progression
Monitoring chronic liver disease remains difficult because clinicians rely on tools that can be inconsistent and may miss early progression. Standard approaches often combine ultrasound imaging with blood-based... Read more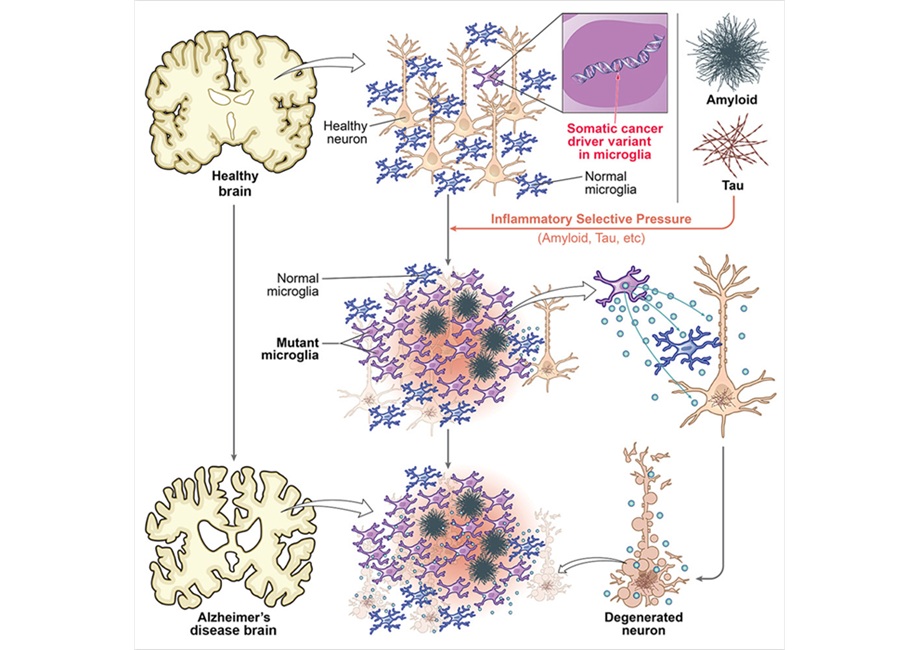
Cancer-Related Mutations in Immune Cells Linked to Alzheimer’s
Alzheimer’s disease is marked by protein aggregation and inflammatory changes in the brain’s immune system, yet its molecular drivers remain incompletely understood. With aging, human cells accumulate... Read more
Composite Blood Biomarkers Enable Early Detection of Common Cancers
Early diagnosis of colorectal, lung, and ovarian cancers remains challenging, with many patients identified only after tumors have begun to spread. A scalable blood test could expand access to screening,... Read more
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read moreHematology
view channel
Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
CareDx (Brisbane, CA, USA) has introduced AlloSeq Nano, a nanopore‑based HLA (human leukocyte antigen) and ABO genotyping solution unveiled at the European Federation for Immunogenetics (EFI) Conference 2026.... Read more
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreMicrobiology
view channel
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read more
Label-Free Microscopy Methodd Enables Faster, Quantitative Detection of Malaria
Microscopy of blood smears remains a cornerstone for malaria diagnosis but can be slow, stain-dependent, and operator intensive. With more than 200 million infections and over 600,000 deaths annually,... Read more
Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
Melanoma remains prone to relapse even after surgery and adjuvant immunotherapy, with 25% to 40% of patients experiencing recurrence. Clinicians lack reliable pre-treatment indicators to identify those... Read more
Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
Gram-negative bloodstream infections and sepsis demand fast, precise antimicrobial therapy, yet conventional susceptibility workflows can delay targeted treatment. Clinical laboratories need platforms... Read morePathology
view channel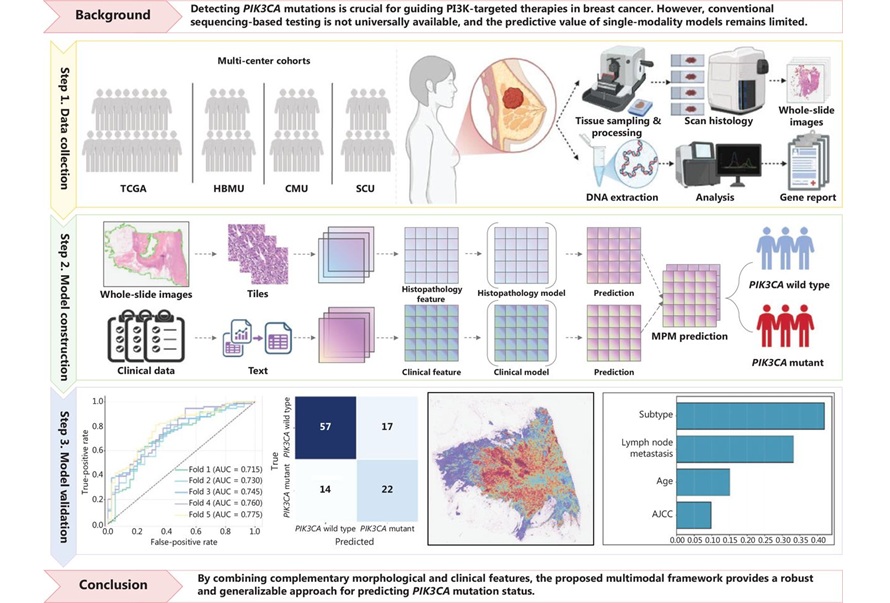
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read more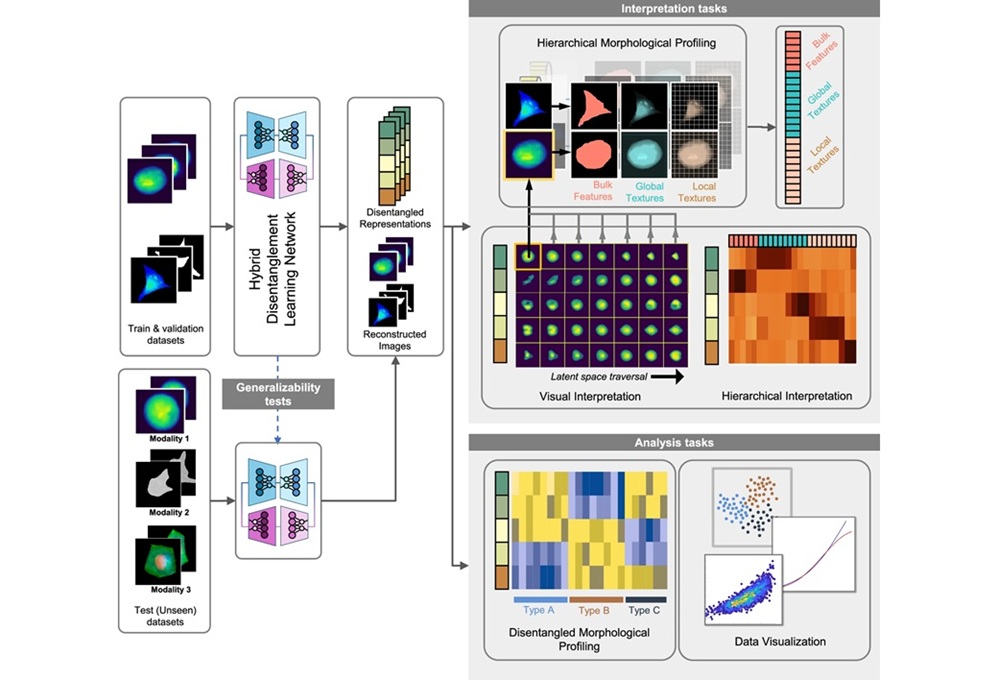
Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
Microscopy images contain rich clues about cell health, but many disease-relevant morphological differences are too subtle to see and difficult to quantify consistently. Artificial intelligence (AI) has... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more