Molecular Test Detects Active Tuberculosis in HIV Patients
|
By LabMedica International staff writers Posted on 11 Nov 2013 |
Image: PAXgene blood ribonucleic acid (RNA) kits (Photo courtesy of PreAnalytiX).
A major impediment to tuberculosis control in Africa is the difficulty in diagnosing active tuberculosis (TB), particularly in the context of human immunodeficiency virus (HIV) infections.
A set of RNA transcriptional signatures expressed in the blood of patients might provide the basis of a diagnostic test that can distinguish active TB from latent TB (LTBI), including from other diseases that have similar clinical symptoms and signs.
An international team of scientists led by those at Imperial College London (UK) recruited 584 adult patients with suspected TB in South Africa and Malawi who were eventually diagnosed with either TB, latent TB infection or another disease, where TB was considered in the differential diagnosis, but then excluded. They took blood samples from these patients and analyzed their blood transcriptional profiles to identify signatures that could be used to calculate a disease risk score capable of differentiating TB from other conditions prevalent in HIV-infected and -uninfected African adults.
The whole blood that was collected before or within 24 hours of commencing TB treatment in suspected patients at the time of recruitment, in PAXgene blood ribonucleic acid (RNA) tubes (PreAnalytiX; Hombrechtikon, Switzerland), frozen within three hours of collection, and later extracted using PAXgene blood RNA kits. RNA was shipped frozen to Singapore for analysis on HumanHT-12 v.4 expression Beadarrays (Illumina; San Diego, CA).
A 27-transcript signature distinguished TB from LTBI and a 44-transcript signature distinguished TB from other diseases. To evaluate the signatures, they used a novel computational method to calculate a disease risk score (DRS) for each patient. The classification based on this score was first evaluated in the test cohort, and then validated in an independent publically available dataset. In the independent validation cohort, the investigators found that patients with TB could be distinguished from patients with latent TB, with a sensitivity of 95% and a specificity of 94%, and from patients with other diseases where the sensitivity was 100% and specificity was 96%.
The authors concludes that from a clinical perspective a simple transcriptome-based test that reliably diagnoses or excludes TB in the majority of patients undergoing investigation for suspected TB. The use of a single blood sample would be of great value, allowing scarce hospital resources to be focused on the small proportion of patients where the result was indeterminate. The challenge is to develop innovative methods to translate multitranscript signatures into simple, cheap tests for TB suitable for use in African health facilities. The study was published on October 22, 2013, in the journal Public Library of Science Medicine.
Related Links:
Imperial College London
PreAnalytiX
Illumina
A set of RNA transcriptional signatures expressed in the blood of patients might provide the basis of a diagnostic test that can distinguish active TB from latent TB (LTBI), including from other diseases that have similar clinical symptoms and signs.
An international team of scientists led by those at Imperial College London (UK) recruited 584 adult patients with suspected TB in South Africa and Malawi who were eventually diagnosed with either TB, latent TB infection or another disease, where TB was considered in the differential diagnosis, but then excluded. They took blood samples from these patients and analyzed their blood transcriptional profiles to identify signatures that could be used to calculate a disease risk score capable of differentiating TB from other conditions prevalent in HIV-infected and -uninfected African adults.
The whole blood that was collected before or within 24 hours of commencing TB treatment in suspected patients at the time of recruitment, in PAXgene blood ribonucleic acid (RNA) tubes (PreAnalytiX; Hombrechtikon, Switzerland), frozen within three hours of collection, and later extracted using PAXgene blood RNA kits. RNA was shipped frozen to Singapore for analysis on HumanHT-12 v.4 expression Beadarrays (Illumina; San Diego, CA).
A 27-transcript signature distinguished TB from LTBI and a 44-transcript signature distinguished TB from other diseases. To evaluate the signatures, they used a novel computational method to calculate a disease risk score (DRS) for each patient. The classification based on this score was first evaluated in the test cohort, and then validated in an independent publically available dataset. In the independent validation cohort, the investigators found that patients with TB could be distinguished from patients with latent TB, with a sensitivity of 95% and a specificity of 94%, and from patients with other diseases where the sensitivity was 100% and specificity was 96%.
The authors concludes that from a clinical perspective a simple transcriptome-based test that reliably diagnoses or excludes TB in the majority of patients undergoing investigation for suspected TB. The use of a single blood sample would be of great value, allowing scarce hospital resources to be focused on the small proportion of patients where the result was indeterminate. The challenge is to develop innovative methods to translate multitranscript signatures into simple, cheap tests for TB suitable for use in African health facilities. The study was published on October 22, 2013, in the journal Public Library of Science Medicine.
Related Links:
Imperial College London
PreAnalytiX
Illumina
Latest Microbiology News
- Comprehensive Review Identifies Gut Microbiome Signatures Associated With Alzheimer’s Disease
- AI-Powered Platform Enables Rapid Detection of Drug-Resistant C. Auris Pathogens
- New Test Measures How Effectively Antibiotics Kill Bacteria
- New Antimicrobial Stewardship Standards for TB Care to Optimize Diagnostics
- New UTI Diagnosis Method Delivers Antibiotic Resistance Results 24 Hours Earlier
- Breakthroughs in Microbial Analysis to Enhance Disease Prediction
- Blood-Based Diagnostic Method Could Identify Pediatric LRTIs
- Rapid Diagnostic Test Matches Gold Standard for Sepsis Detection
- Rapid POC Tuberculosis Test Provides Results Within 15 Minutes
- Rapid Assay Identifies Bloodstream Infection Pathogens Directly from Patient Samples
- Blood-Based Molecular Signatures to Enable Rapid EPTB Diagnosis
- 15-Minute Blood Test Diagnoses Life-Threatening Infections in Children
- High-Throughput Enteric Panels Detect Multiple GI Bacterial Infections from Single Stool Swab Sample
- Fast Noninvasive Bedside Test Uses Sugar Fingerprint to Detect Fungal Infections
- Rapid Sepsis Diagnostic Device to Enable Personalized Critical Care for ICU Patients
- Microfluidic Platform Assesses Neutrophil Function in Sepsis Patients
Channels
Clinical Chemistry
view channel
New PSA-Based Prognostic Model Improves Prostate Cancer Risk Assessment
Prostate cancer is the second-leading cause of cancer death among American men, and about one in eight will be diagnosed in their lifetime. Screening relies on blood levels of prostate-specific antigen... Read more
Extracellular Vesicles Linked to Heart Failure Risk in CKD Patients
Chronic kidney disease (CKD) affects more than 1 in 7 Americans and is strongly associated with cardiovascular complications, which account for more than half of deaths among people with CKD.... Read moreMolecular Diagnostics
view channel
Diagnostic Device Predicts Treatment Response for Brain Tumors Via Blood Test
Glioblastoma is one of the deadliest forms of brain cancer, largely because doctors have no reliable way to determine whether treatments are working in real time. Assessing therapeutic response currently... Read more
Blood Test Detects Early-Stage Cancers by Measuring Epigenetic Instability
Early-stage cancers are notoriously difficult to detect because molecular changes are subtle and often missed by existing screening tools. Many liquid biopsies rely on measuring absolute DNA methylation... Read more
“Lab-On-A-Disc” Device Paves Way for More Automated Liquid Biopsies
Extracellular vesicles (EVs) are tiny particles released by cells into the bloodstream that carry molecular information about a cell’s condition, including whether it is cancerous. However, EVs are highly... Read more
Blood Test Identifies Inflammatory Breast Cancer Patients at Increased Risk of Brain Metastasis
Brain metastasis is a frequent and devastating complication in patients with inflammatory breast cancer, an aggressive subtype with limited treatment options. Despite its high incidence, the biological... Read moreHematology
view channel
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read more
Fast and Easy Test Could Revolutionize Blood Transfusions
Blood transfusions are a cornerstone of modern medicine, yet red blood cells can deteriorate quietly while sitting in cold storage for weeks. Although blood units have a fixed expiration date, cells from... Read more
Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
High-volume hemostasis sections must sustain rapid turnaround while managing reruns and reflex testing. Manual tube handling and preanalytical checks can strain staff time and increase opportunities for error.... Read more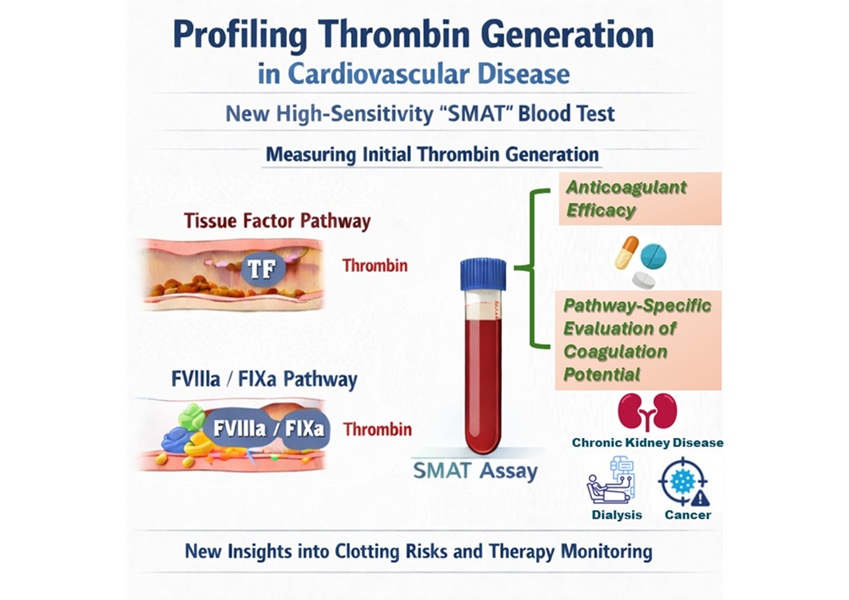
High-Sensitivity Blood Test Improves Assessment of Clotting Risk in Heart Disease Patients
Blood clotting is essential for preventing bleeding, but even small imbalances can lead to serious conditions such as thrombosis or dangerous hemorrhage. In cardiovascular disease, clinicians often struggle... Read moreImmunology
view channelBlood Test Identifies Lung Cancer Patients Who Can Benefit from Immunotherapy Drug
Small cell lung cancer (SCLC) is an aggressive disease with limited treatment options, and even newly approved immunotherapies do not benefit all patients. While immunotherapy can extend survival for some,... Read more
Whole-Genome Sequencing Approach Identifies Cancer Patients Benefitting From PARP-Inhibitor Treatment
Targeted cancer therapies such as PARP inhibitors can be highly effective, but only for patients whose tumors carry specific DNA repair defects. Identifying these patients accurately remains challenging,... Read more
Ultrasensitive Liquid Biopsy Demonstrates Efficacy in Predicting Immunotherapy Response
Immunotherapy has transformed cancer treatment, but only a small proportion of patients experience lasting benefit, with response rates often remaining between 10% and 20%. Clinicians currently lack reliable... Read morePathology
view channel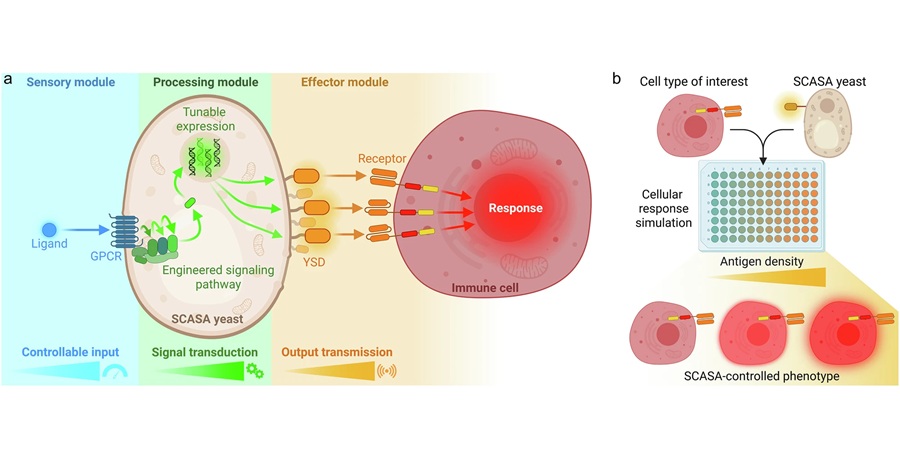
Engineered Yeast Cells Enable Rapid Testing of Cancer Immunotherapy
Developing new cancer immunotherapies is a slow, costly, and high-risk process, particularly for CAR T cell treatments that must precisely recognize cancer-specific antigens. Small differences in tumor... Read more
First-Of-Its-Kind Test Identifies Autism Risk at Birth
Autism spectrum disorder is treatable, and extensive research shows that early intervention can significantly improve cognitive, social, and behavioral outcomes. Yet in the United States, the average age... Read moreTechnology
view channel
Robotic Technology Unveiled for Automated Diagnostic Blood Draws
Routine diagnostic blood collection is a high‑volume task that can strain staffing and introduce human‑dependent variability, with downstream implications for sample quality and patient experience.... Read more
ADLM Launches First-of-Its-Kind Data Science Program for Laboratory Medicine Professionals
Clinical laboratories generate billions of test results each year, creating a treasure trove of data with the potential to support more personalized testing, improve operational efficiency, and enhance patient care.... Read moreAptamer Biosensor Technology to Transform Virus Detection
Rapid and reliable virus detection is essential for controlling outbreaks, from seasonal influenza to global pandemics such as COVID-19. Conventional diagnostic methods, including cell culture, antigen... Read more
AI Models Could Predict Pre-Eclampsia and Anemia Earlier Using Routine Blood Tests
Pre-eclampsia and anemia are major contributors to maternal and child mortality worldwide, together accounting for more than half a million deaths each year and leaving millions with long-term health complications.... Read moreIndustry
view channelNew Collaboration Brings Automated Mass Spectrometry to Routine Laboratory Testing
Mass spectrometry is a powerful analytical technique that identifies and quantifies molecules based on their mass and electrical charge. Its high selectivity, sensitivity, and accuracy make it indispensable... Read more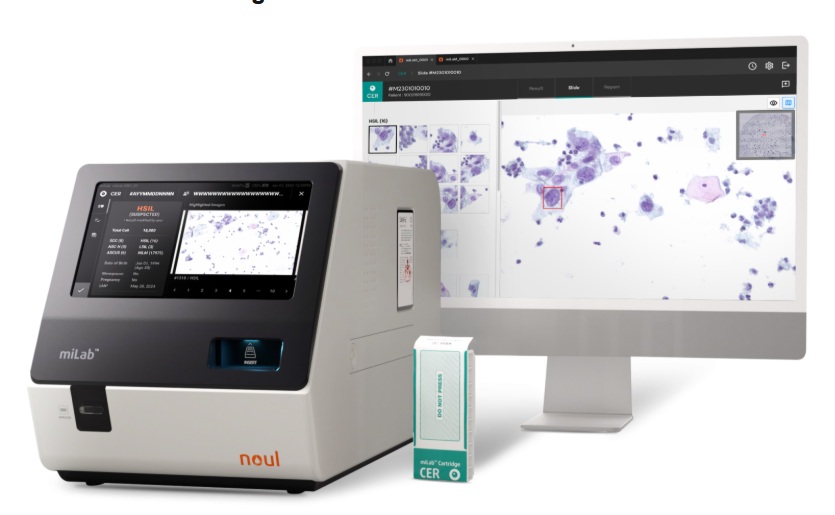
AI-Powered Cervical Cancer Test Set for Major Rollout in Latin America
Noul Co., a Korean company specializing in AI-based blood and cancer diagnostics, announced it will supply its intelligence (AI)-based miLab CER cervical cancer diagnostic solution to Mexico under a multi‑year... Read more
Diasorin and Fisher Scientific Enter into US Distribution Agreement for Molecular POC Platform
Diasorin (Saluggia, Italy) has entered into an exclusive distribution agreement with Fisher Scientific, part of Thermo Fisher Scientific (Waltham, MA, USA), for the LIAISON NES molecular point-of-care... Read more